

Synopsis
Synopsis
0
JDMF
0
KDMF
0
VMF
0
FDF
0
Canada

0
Australia

0
South Africa

0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
Annual Reports
NA

1. Agyrax
2. Antivert
3. Bonamine
4. Bonine
5. Chiclida
6. D Vert
7. D-vert
8. Dihydrochloride, Meclizine
9. Dvert
10. Histametizyn
11. Hydrochloride, Meclizine
12. Meclizine
13. Meclizine Dihydrochloride
14. Meclizine Hydrochloride
15. Meclizine Monohydrochloride
16. Meclozine
17. Monohydrochloride, Meclizine
18. Parachloramine
19. Ru Vert M
20. Ru-vert-m
1. Meclizine Hydrochloride
2. Meclizine Dihydrochloride Monohydrate
3. 31884-77-2
4. Postafen
5. Agyrax
6. Bonamine
7. Hdp7w44cio
8. Meclozine Hydrochloride Monohydrate
9. Ucb-5062
10. 1-[(4-chlorophenyl)-phenylmethyl]-4-[(3-methylphenyl)methyl]piperazine;hydrate;dihydrochloride
11. Nsc-757094
12. Piperazine, 1-((4-chlorophenyl)phenylmethyl)-4-((3-methylphenyl)methyl)-, Dihydrochloride, Monohydrate
13. Mfcd00941489
14. Meclizine Hcl Hydrate
15. Nsc-28728
16. Unii-hdp7w44cio
17. Antivert (tn)
18. Chembl3989555
19. Chebi:31805
20. Dtxsid30953792
21. Meclizine Hydrochloride (jan/usp)
22. Meclizine Hydrochloride [usp:jan]
23. Bcp13363
24. Meclizine Hydrochloride [jan]
25. Meclizine Hydrochloride [vandf]
26. Nsc 757094
27. 1-(p-chloro-alpha-phenylbenzyl)-4-(m-methylbenzyl)piperazine Dihydrochloride Monohydrate
28. Meclizine Hydrochloride [usp-rs]
29. As-13740
30. Meclizine Hydrochloride [orange Book]
31. D01317
32. Meclizine Hydrochloride [usp Monograph]
33. A821043
34. Meclizine Dihydrochloride Monohydrate [mi]
35. (+/-)-meclizine Dihydrochloride Monohydrate
36. Meclozine Hydrochloride Monohydrate [who-dd]
37. Meclizine Dihydrochloride Monohydrate, (+/-)-
38. Q27279880
39. 1-((4-chlorophenyl)(phenyl)methyl)-4-(3-methylbenzyl)piperazine Dihydrochloride Hydrate
40. 1-((4-chlorophenyl)(phenyl)methyl)-4-(3-methylbenzyl)piperazinedihydrochloridehydrate
41. 1-(p-chloro-alpha-phenylbenzyl)-4-(m-methylbenzyl) Piperazine Dihydrochloride Monohydrate
42. 1-(p-chloro-.alpha.-phenylbenzyl)-4-(m-methylbenzyl)piperazine Dihydrochloride Monohydrate
43. 1-[(4-chlorophenyl)(phenyl)methyl]-4-[(3-methylphenyl)methyl]piperazine--hydrogen Chloride--water (1/2/1)
44. 1-[(4-chlorophenyl)-phenyl-methyl]-4-(m-tolylmethyl)piperazine Hydrate Dihydrochloride;meclizine Dihydrochloride Monohydrate
45. 1-[(4-chlorophenyl)phenylmethyl]-4-[(3-methylphenyl)methyl]piperazine Dihydrochloride Hydrate
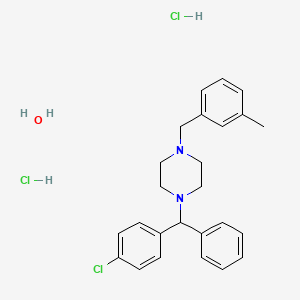
| Molecular Weight | 481.9 g/mol |
|---|---|
| Molecular Formula | C25H31Cl3N2O |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 5 |
| Exact Mass | 480.150197 g/mol |
| Monoisotopic Mass | 480.150197 g/mol |
| Topological Polar Surface Area | 7.5 Ų |
| Heavy Atom Count | 31 |
| Formal Charge | 0 |
| Complexity | 448 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 4 |
| 1 of 1 | |
|---|---|
| Drug Name | MECLIZINE HYDROCHLORIDE |
| Active Ingredient | MECLIZINE HYDROCHLORIDE |
| Company | AMNEAL PHARMS (Application Number: A201451); EPIC PHARMA LLC (Application Number: A200294); JUBILANT CADISTA (Application Number: A040659); MYLAN PHARMS INC (Application Number: A202640); PAR PHARM (Application Number: A087127); PAR PHARM (Application Number: A087128); SANDOZ (Application Number: A084092); SANDOZ (Application Number: A084843) |
Anti-Allergic Agents
Agents that are used to treat allergic reactions. Most of these drugs act by preventing the release of inflammatory mediators or inhibiting the actions of released mediators on their target cells. (From AMA Drug Evaluations Annual, 1994, p475) (See all compounds classified as Anti-Allergic Agents.)
Antiemetics
Drugs used to prevent NAUSEA or VOMITING. (See all compounds classified as Antiemetics.)
Histamine H1 Antagonists
Drugs that selectively bind to but do not activate histamine H1 receptors, thereby blocking the actions of endogenous histamine. Included here are the classical antihistaminics that antagonize or prevent the action of histamine mainly in immediate hypersensitivity. They act in the bronchi, capillaries, and some other smooth muscles, and are used to prevent or allay motion sickness, seasonal rhinitis, and allergic dermatitis and to induce somnolence. The effects of blocking central nervous system H1 receptors are not as well understood. (See all compounds classified as Histamine H1 Antagonists.)
Cholinergic Antagonists
Drugs that bind to but do not activate CHOLINERGIC RECEPTORS, thereby blocking the actions of ACETYLCHOLINE or cholinergic agonists. (See all compounds classified as Cholinergic Antagonists.)
Meclizine / Meclozine Hydrochloride USP
Date of Issue : 2020-06-18
Valid Till : 2022-08-13
Written Confirmation Number : WC-0229A2
Address of the Firm : Survey No. 270 Navabpet Village Shivampet Mandal Medak Dist, India

Meclizine Hydrochloride Ph.Eur.
Date of Issue : 2022-09-01
Valid Till : 2024-11-04
Written Confirmation Number : WC-0092
Address of the Firm : Plot No. 18, 56, 57 & 58, K.I.A.D.B. Industrial Area\r\nNanjangud - 571 302, Mys...

Meclozine Hydrochloride(Ph. Eur/BP)
Date of Issue : 2019-09-03
Valid Till : 2022-09-02
Written Confirmation Number : WC-246
Address of the Firm : 17/24, MIDC, Taloja, Dist. Raigad, Maharashtra

Meclizine Hydrochloride (USP/EP/IP)
Date of Issue : 2019-10-07
Valid Till : 2022-09-08
Written Confirmation Number : WC-0072
Address of the Firm : Plot No. 25/B, Phase-III, I.D.A., Jeedimetla, Hyderabad-500 055. AP.

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
NDC Package Code : 17511-108
Start Marketing Date : 2020-02-10
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1mg/mg)
Marketing Category : BULK INGREDIENT
NDC Package Code : 66639-014
Start Marketing Date : 2019-01-18
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 65372-1137
Start Marketing Date : 2008-06-12
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

MECLIZINE HYDROCHLORIDE ANHYDROUS
NDC Package Code : 51927-0228
Start Marketing Date : 2022-02-08
End Marketing Date : 2025-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT FOR HUMAN P...

MECLIZINE HYDROCHLORIDE ANHYDROUS
NDC Package Code : 71554-010
Start Marketing Date : 2019-04-27
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 49452-4390
Start Marketing Date : 2016-04-01
End Marketing Date : 2027-10-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT

NDC Package Code : 66326-809
Start Marketing Date : 2020-01-19
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 13746-102
Start Marketing Date : 2008-01-02
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 66022-0114
Start Marketing Date : 1982-01-20
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 66022-0114
Start Marketing Date : 1982-01-20
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
About the Company : LGM Pharma is a global leader in sourcing hard-to-find APIs and intermediates for the pharmaceutical and biotech industries. LGM is also a full service CDMO providing formulation, ...
 Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
About the Company : Cohance Lifesciences is a leading CDMO and API platform, offering products and services across all phases of a molecule’s lifecycle from development to commercialzation. With our...
About the Company : Jai Radhe Sales was founded in 1999 as an out-of-the-box distribution firm specializing in the global supply of high-quality pharmaceutical ingredients. The firm provides complete ...
About the Company : Pharm-Rx has earned an outstanding reputation since its establishment in 1991, serving as a reputable importer and distributor of active ingredients to the pharmaceutical, nutritio...
About the Company : Sigmak Lifesciences is a leading contract manufacturer, merchant exporter, & distributor of pharmaceutical APIs & Fine Chemicals. We excel in Contract Manufacturing, covering a wid...
About the Company : Our endeavours also lie in the import, and domestic sourcing of raw materials from reliable vendors of the market. We have an excellent infrastructure with world class cGMP facilit...
About the Company : Deliver excellent quality through continuous research and development to satisfy customer needs on time, offer highest value to our customers through a Professional, integrated and...

About the Company : With 33 years of CONSISTENCY, CREDIBILITY & CONTINUITY, D K Pharma has established its presence in the Global Market for Manufacturing Bulk Drugs, Speciality Fine Chemicals and Adv...

About the Company : Headquartered in Mumbai, Keshava Organics is a US FDA approved facility making select antihistamine active pharmaceutical ingredients.Our facility located 100 kms north of Mumbai i...

About the Company : RR LIFESCIENCES manufactures products purely by organic synthesis to offer its customers a choice of products in their areas. Our facilities are equipped with a range of reactor si...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Regulatory Info :
Registration Country : Sweden
meklozindihydrokloridmonohydrat
Brand Name : Postafen
Dosage Form : TABLET
Dosage Strength : 25 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength : 12.5MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Packaging :
Regulatory Info :
Dosage : Tablet
Dosage Strength : 12.5MG
Brand Name :
Approval Date :
Application Number :
Registration Country : India
 Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength : 25MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Packaging :
Regulatory Info :
Dosage : Tablet
Dosage Strength : 25MG
Brand Name :
Approval Date :
Application Number :
Registration Country : India
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength : 50MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
Packaging :
Regulatory Info :
Dosage : Tablet
Dosage Strength : 50MG
Brand Name :
Approval Date :
Application Number :
Registration Country : India
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Dosage Form : Tablet
Grade : Oral
Brand Name : Sodium Stearyl Fumarate G...
Application : Lubricants & Glidants
Pharmacopoeia Ref : USP, EP, ICH, Q7GMP
Technical Specs : NA
Ingredient(s) : Sodium Stearyl Fumarate
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Suspension
Grade : Oral
Brand Name : Acesulfame Potassium
Application : Taste Masking
Dosage Form : Tablet
Grade : Oral
Dosage Form : Syrup
Grade : Oral
Application : Fillers, Diluents & Binders
Dosage Form : Tablet
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Application : Taste Masking
Excipient Details : Mannitol is mainly used as a bulking agent in tableting. It is also used as taste masking agent in ODT formulations.
Dosage Form : Suspension
Grade : Oral
Dosage Form : Suspension
Grade : Oral
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Injectable / Parenteral
Grade : Parenteral
Brand Name : Expansorb® PLA / PEG
Application : Parenteral
Excipient Details : Expansorb® PLA?/PEG? polymers are used as functional excipients in injectable formulations for controlled & slow drug release with a single injection.
Pharmacopoeia Ref : N/A
Technical Specs : Classic and ultrapure LMP
Ingredient(s) : Poly lactic acid
Dosage Form : Injectable / Parenteral
Grade : Parenteral
Brand Name : Expansorb® PLGA / PEG
Application : Parenteral
Excipient Details : Expansorb® PLGA? / PEG copolymers are used as functional excipients in single dose injections for controlled and slow drug release (weeks to months).
Pharmacopoeia Ref : N/A
Technical Specs : Classic and ultrapure LMP
Ingredient(s) : Poly-DL-Lactic-co-Glycolic Acid
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
We have 21 companies offering Meclizine hydrochloride anhydrous
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
