
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
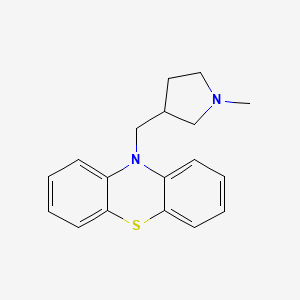

1. Methdilazine Hydrochloride
1. Tacaryl
2. 1982-37-2
3. Methdilazinum
4. Tacazyl
5. Methdilazinum [inn-latin]
6. Metodilazina [inn-spanish]
7. Nci-c60720
8. 10-[(1-methylpyrrolidin-3-yl)methyl]phenothiazine
9. Methdilazine (inn)
10. 10-[(1-methylpyrrolidin-3-yl)methyl]-10h-phenothiazine
11. 10-((1-methyl-3-pyrrolidinyl)methyl)phenothiazine
12. 4q13ly9z8x
13. Chebi:6823
14. Methodilazine
15. Metodilazina
16. 10h-phenothiazine, 10-((1-methyl-3-pyrrolidinyl)methyl)-
17. 10h-phenothiazine, 10-[(1-methyl-3-pyrrolidinyl)methyl]-
18. Tacryl
19. Methdilazine [inn]
20. Nsc169091
21. Mj 5022
22. 10-[(1-methyl-3-pyrrolidinyl)methyl]phenothiazine
23. Phenothiazine, 10-((1-methyl-3-pyrrolidinyl)methyl)-
24. Phenothiazine, 10-[(1-methyl-3-pyrrolidinyl)methyl]-
25. 10-[(1-methyl-3-pyrrolidinyl)methyl]phenothiazine; Mj 5022
26. Tacaryl (tn)
27. Product 5022
28. Einecs 217-841-6
29. Metdilazina
30. Unii-4q13ly9z8x
31. Methdilazine [usp:inn:ban]
32. 10-((1-methyl-3-pyrrolidinyl)methyl)phenothiazine Monohydrochloride
33. Dilosyn (salt/mix)
34. Disyncram (salt/mix)
35. Disyncran (salt/mix)
36. 10h-phenothiazine, 10-((1-methyl-3-pyrrolidinyl)methyl)-, Monohydrochloride
37. Methdilazine [mi]
38. Methdilazine [vandf]
39. Methdilazine [mart.]
40. Methdilazine(200mg)
41. Methdilazine [who-dd]
42. Schembl121507
43. Gtpl7231
44. 10-((1-methyl-3-pyrrolidinyl)methyl)-10h-phenothiazine
45. Chembl1200959
46. Dtxsid6023282
47. Bdbm81470
48. Methdilazine (200 Mg)
49. Methdilazine [orange Book]
50. Hy-b1690
51. 10h-phenothiazine, Tannate (5:1)
52. Cas_14677
53. Nsc_14677
54. Nsc244191
55. Pdsp1_000149
56. Pdsp2_000148
57. Db00902
58. Nsc-244191
59. Nci60_001351
60. Cs-0013673
61. C07175
62. D04979
63. L001130
64. Sr-01000944494
65. Q6665709
66. Sr-01000944494-1
67. 10-[(1-methyl-3-pyrrolidinyl)methyl]-10h-phenothiazine #
| Molecular Weight | 296.4 g/mol |
|---|---|
| Molecular Formula | C18H20N2S |
| XLogP3 | 5.2 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 2 |
| Exact Mass | 296.13471982 g/mol |
| Monoisotopic Mass | 296.13471982 g/mol |
| Topological Polar Surface Area | 31.8 Ų |
| Heavy Atom Count | 21 |
| Formal Charge | 0 |
| Complexity | 339 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Used for the symptomatic relief of hypersensitivity reactions and particularly for the control of pruritic skin disorders
In allergic reactions an allergen interacts with and cross-links surface IgE antibodies on mast cells and basophils. Once the mast cell-antibody-antigen complex is formed, a complex series of events occurs that eventually leads to cell-degranulation and the release of histamine (and other chemical mediators) from the mast cell or basophil. Once released, histamine can react with local or widespread tissues through histamine receptors. Histamine, acting on H1-receptors, produces pruritis, vasodilatation, hypotension, flushing, headache, tachycardia, and bronchoconstriction. Histamine also increases vascular permeability and potentiates pain. Methdilazine is a histamine H1 antagonist. It competes with histamine for the normal H1-receptor sites on effector cells of the gastrointestinal tract, blood vessels and respiratory tract. It provides effective, temporary relief of sneezing, watery and itchy eyes, and runny nose due to hay fever and other upper respiratory allergies.
R - Respiratory system
R06 - Antihistamines for systemic use
R06A - Antihistamines for systemic use
R06AD - Phenothiazine derivatives
R06AD04 - Methdilazine
Absorption
Well absorbed in the digestive tract.
Methdilazine binds to the histamine H1 receptor. This blocks the action of endogenous histamine, which subsequently leads to temporary relief of the negative symptoms brought on by histamine.
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
60
PharmaCompass offers a list of Methdilazine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Methdilazine manufacturer or Methdilazine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Methdilazine manufacturer or Methdilazine supplier.
PharmaCompass also assists you with knowing the Methdilazine API Price utilized in the formulation of products. Methdilazine API Price is not always fixed or binding as the Methdilazine Price is obtained through a variety of data sources. The Methdilazine Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Methdilazine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Methdilazine, including repackagers and relabelers. The FDA regulates Methdilazine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Methdilazine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Methdilazine supplier is an individual or a company that provides Methdilazine active pharmaceutical ingredient (API) or Methdilazine finished formulations upon request. The Methdilazine suppliers may include Methdilazine API manufacturers, exporters, distributors and traders.
click here to find a list of Methdilazine suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Methdilazine DMF (Drug Master File) is a document detailing the whole manufacturing process of Methdilazine active pharmaceutical ingredient (API) in detail. Different forms of Methdilazine DMFs exist exist since differing nations have different regulations, such as Methdilazine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Methdilazine DMF submitted to regulatory agencies in the US is known as a USDMF. Methdilazine USDMF includes data on Methdilazine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Methdilazine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Methdilazine suppliers with USDMF on PharmaCompass.
Methdilazine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Methdilazine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Methdilazine GMP manufacturer or Methdilazine GMP API supplier for your needs.
A Methdilazine CoA (Certificate of Analysis) is a formal document that attests to Methdilazine's compliance with Methdilazine specifications and serves as a tool for batch-level quality control.
Methdilazine CoA mostly includes findings from lab analyses of a specific batch. For each Methdilazine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Methdilazine may be tested according to a variety of international standards, such as European Pharmacopoeia (Methdilazine EP), Methdilazine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Methdilazine USP).
