
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz

1. Maidimeisu
2. Midecamin
3. Midecamycin Acetate
4. Midecamycin Diacetate
5. Midekamycin
6. Midekamycin Acetate
7. Mosil
8. Mydecamycin
9. Myoxam
10. Neoisomidecamycin
11. Normicina
12. Sf 837
13. Sf-837
1. Rubimycin
2. Espinomycin A
3. Platenomycin B1
4. Turimycin P3
5. Medecamycin A1
6. Macropen
7. Mydecamycin
8. Normicina
9. Myoxam
10. 35457-80-8
11. Midecamycin A1
12. Medemycin A1
13. Momicine
14. Medemycin
15. Mydecamycin A1
16. Leucomycin V, 3,4b-dipropanoate
17. Antibiotic Sf 837
18. N34z0y5uh7
19. Sf 837
20. Madecacine
21. Midecamicina
22. Midecamycine
23. Midecamycinum
24. Yl 704 B1
25. Macro-dil
26. Antibiotic Sf-837
27. Turimycin P(sub 3)
28. Midecamycin A(sub 1)
29. Antibiotic Sf 837 A1
30. Antibiotic Yl 704 B1
31. Midecamycine [inn-french]
32. Midecamycinum [inn-latin]
33. Nsc 154011
34. Midecamicina [inn-spanish]
35. Unii-n34z0y5uh7
36. Midecamycin [inn:dcf:jan]
37. Midecamycin,(s)
38. Nsc-154011
39. Medemycin (tn)
40. Ncgc00016830-01
41. Einecs 252-578-0
42. Cas-35457-80-8
43. Midecamycin [inn]
44. Midecamycin [jan]
45. Midecamycin (jp17/inn)
46. Dsstox_cid_25463
47. Dsstox_rid_80894
48. Midecamycin [mart.]
49. Dsstox_gsid_45463
50. Midecamycin [who-dd]
51. Midecamycin A1 [mi]
52. Schembl141581
53. Chembl444963
54. Dtxsid5045463
55. Chebi:31845
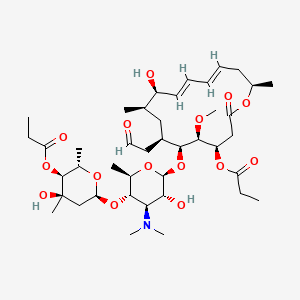
56. (4r,5s,6s,7r,9r,10r,11e,13e,16r)-6-(((2s,3r,4r,5s,6r)-4-(dimethylamino)-3-hydroxy-5-(((2s,4r,5s,6s)-4-hydroxy-4,6-dimethyl-5-(propionyloxy)tetrahydro-2h-pyran-2-yl)oxy)-6-methyltetrahydro-2h-pyran-2-yl)oxy)-10-hydroxy-5-methoxy-9,16-dimethyl-2-oxo-7-(2-oxoethyl)oxacyclohexadeca-11,13-dien-4-yl Propionate
57. Act02621
58. Hy-b1908
59. Tox21_110635
60. S5560
61. Akos022185298
62. Zinc169368401
63. Ccg-270507
64. Cs-5909
65. Db13456
66. Leucomycin V, 3,4(sup B)-dipropanoate
67. D01339
68. H10500
69. Midecamycin), Antibiotic For Culture Media Use Only
70. Q2636110
71. W-106669
72. Espinomycin A;medecamycin A1;platenomycin B1;rubimycin;turimycin P3
73. 7-(formylmethyl)-4,10-dihydroxy-5-methoxy-9,16-dimethyl-2-oxooxacyclohexadeca-11,13-dien-6-yl 3,6-dideoxy-4-o-(2,6-dideoxy-3-c-methyl-alpha-l-ribo-hexopyranosyl)-3-(dimethylamino)-beta-d-glucopyranoside 4',4''-dipropionate (ester)
74. 7-(formylmethyl)-4,10-dihydroxy-5-methoxy-9,16-dimethyl-2-oxooxacyclohexadeca-11,13-dien-6-yl 3,6-dideoxy-4-o-(2,6-dideoxy-3-c-methyl-alpha-l-ribo-hexopyranosyl)-3-(dimethylamino)-beta-d-glucopyranoside 4',4'-dipropionate (ester)
| Molecular Weight | 814.0 g/mol |
|---|---|
| Molecular Formula | C41H67NO15 |
| XLogP3 | 2.6 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 16 |
| Rotatable Bond Count | 14 |
| Exact Mass | 813.45107043 g/mol |
| Monoisotopic Mass | 813.45107043 g/mol |
| Topological Polar Surface Area | 206 Ų |
| Heavy Atom Count | 57 |
| Formal Charge | 0 |
| Complexity | 1360 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 16 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 2 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Midecamycin was used for the treatment of infections in the oral cavity, upper and lower respiratory tracts and skin and soft tissue infections. The alone use of midecamycin was mainly used in Europe or Japan.
Reports have indicated that midecamycin is active against both erythromycin-susceptible and efflux-mediated erythromycin-resistant strains. The diacetate form of this product reduces gastrointestinal side effects and improves its pharmacokinetic profile. Studies have proved that midecamycin is highly active against Gram-positive organisms. The activity of midecamycin in the form of acetate salt presents a better activity, which seems to be potentiated at pH 7-8, as well as a longer half-life.
Anti-Bacterial Agents
Substances that inhibit the growth or reproduction of BACTERIA. (See all compounds classified as Anti-Bacterial Agents.)
J - Antiinfectives for systemic use
J01 - Antibacterials for systemic use
J01F - Macrolides, lincosamides and streptogramins
J01FA - Macrolides
J01FA03 - Midecamycin
Absorption
Midecamycin is rapidly and almost completely absorbed when orally administered. It is mainly absorbed in the alkaline intestinal environment. This rapid absorption is due to its liposoluble property which allows for good penetration in the tissues, especially bronchial secretion, prostatic tissue, middle ear exudates and bone tissue. The tissue/serum ratio concentration is greater than 1 which indicates that this product does not stay long in the plasma. After oral administration of 600 mg of midecamycin, the peak serum concentration is 0.8 mg/L and it is attained 1 hour after oral administration. This concentration dereased significantly after 4-6 hours.
Route of Elimination
The major route of elimination of midecamycin is is the liver, followed by a low significance of renal elimination. Urinary concentrations accounts for about 3.3% of the administered dose after 6 hours.
Volume of Distribution
The reported apparent volume of distribution of midecamycin is 7.7 L/kg.
Clearance
Midecamycin presentas a low renal clearance value.
Midecamycin undergoes extensive biotransformation in the liver and its metabolites are characterized by presenting little to no antimicrobial activity. The main metabolite is formed by a 14-hydroxylation and it can be also detected in urine.
The half-life of midecamycin is longer than the first macrolide antibiotics. after intravenous administration, the half-life reported is of 54 minutes.
Midecamycin, as part of the macrolides, act by inhibiting bacterial protein synthesis. More specifically, midecamycin inhibits bacterial growth by targetting the 50S ribosomal subunit preventing peptide bond formation and translocation during protein synthesis. The presence of mutations in the 50S RNA can prevent midecamycin binding. Midecamycin is a broad spectrum antibiotic and thus, it can interact with different bacteria.
ABOUT THIS PAGE
26
PharmaCompass offers a list of Midecamycin API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Midecamycin manufacturer or Midecamycin supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Midecamycin manufacturer or Midecamycin supplier.
PharmaCompass also assists you with knowing the Midecamycin API Price utilized in the formulation of products. Midecamycin API Price is not always fixed or binding as the Midecamycin Price is obtained through a variety of data sources. The Midecamycin Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Midecamycin manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Midecamycin, including repackagers and relabelers. The FDA regulates Midecamycin manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Midecamycin API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Midecamycin supplier is an individual or a company that provides Midecamycin active pharmaceutical ingredient (API) or Midecamycin finished formulations upon request. The Midecamycin suppliers may include Midecamycin API manufacturers, exporters, distributors and traders.
click here to find a list of Midecamycin suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Midecamycin written confirmation (Midecamycin WC) is an official document issued by a regulatory agency to a Midecamycin manufacturer, verifying that the manufacturing facility of a Midecamycin active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Midecamycin APIs or Midecamycin finished pharmaceutical products to another nation, regulatory agencies frequently require a Midecamycin WC (written confirmation) as part of the regulatory process.
click here to find a list of Midecamycin suppliers with Written Confirmation (WC) on PharmaCompass.
Midecamycin Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Midecamycin GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Midecamycin GMP manufacturer or Midecamycin GMP API supplier for your needs.
A Midecamycin CoA (Certificate of Analysis) is a formal document that attests to Midecamycin's compliance with Midecamycin specifications and serves as a tool for batch-level quality control.
Midecamycin CoA mostly includes findings from lab analyses of a specific batch. For each Midecamycin CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Midecamycin may be tested according to a variety of international standards, such as European Pharmacopoeia (Midecamycin EP), Midecamycin JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Midecamycin USP).
