

Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
FDA Orange Book

0
Canada

0
South Africa

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Weekly News Recap #Phispers
US Medicaid
NA
Finished Drug Prices
NA
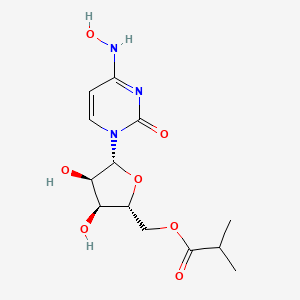

1. ((2r,3s,4r,5r)-3,4-dihydroxy-5-((4z)-4-(hydroxyimino)-2-oxo-3,4- Dihydropyrimidin-1(2h)-yl)oxolan-2-yl)methyl 2-methylpropanoate
2. Lagevrio
3. Mk-4482
4. Molnupiravir
1. Eidd 2801
2. Eidd2801
3. Molnupiravir
4. 2349386-89-4
5. Mk-4482
6. Molnupiravir [inn]
7. 2492423-29-5
8. Molnupiravir [usan]
9. Molnupiravir [who-dd]
10. Ya84ki1vew
11. Eidd 1931-isopropyl Ester
12. Uridine, 4-oxime, 5'-(2-methylpropanoate), (4z)-
13. N4-hydroxycytidine, 5'-isopropyl Ester
14. ((2r,3s,4r,5r)-3,4-dihydroxy-5-((4z)-4-(hydroxyimino)-2-oxo-3,4- Dihydropyrimidin-1(2h)-yl)oxolan-2-yl)methyl 2-methylpropanoate
15. ((2r,3s,4r,5r)-3,4-dihydroxy-5-(4-(hydroxyamino)-2-oxopyrimidin-1(2h)-yl)tetrahydrofuran-2-yl)methyl Isobutyrate
16. [(2r,3s,4r,5r)-3,4-dihydroxy-5-[4-(hydroxyamino)-2-oxopyrimidin-1-yl]oxolan-2-yl]methyl 2-methylpropanoate
17. Lagevrio
18. Who 11853
19. ((2r,3s,4r,5r)-3,4-dihydroxy-5-(4-(hydroxyimino)-2-oxo-3,4-dihydropyrimidin-1(2h)-yl)tetrahydrofuran-2-yl)methyl Isobutyrate
20. Molnupiravirum
21. Unii-ya84ki1vew
22. Pro-eidd-1931
23. Molnupiravir [jan]
24. Eidd-2801(molnupiravir)
25. Chembl4650320
26. Eidd 1931 5'-isopropylester
27. Gtpl10737
28. Med.21724, Compound 182
29. Chebi:180653
30. Bdbm429508
31. Dtxsid501028058
32. Bcp32744
33. Eidd-1931 Isopropyl Ester
34. Ex-a3432
35. Mfcd32663515
36. Mk4482
37. S8969
38. At13078
39. N(4)-hydroxycytidine 5'-isopropylester
40. Ac-35171
41. As-84465
42. Beta-d-n4 Hydroxycytidine-5'-isopropyl Ester
43. Beta-d-n(4)-hydroxycytidine-5'-isopropyl Ester
44. A936190
45. .beta.-d-n4 Hydroxycytidine-5'-isopropyl Ester
46. N-hydroxy-5'-o-(2-methylpropanoyl)-3,4-dihydrocytidine
47. Eidd 2801; Eidd2801; Uridine, 4-oxime, 5'-(2-methylpropanoate
48. ((2r,3s,4r,5r)-3,4-dihydroxy-5-((e)-4-(hydroxyimino)-2-oxo-3,4-dihydropyrimidin-1(2h)-yl)tetrahydrofuran-2-yl)methyl Isobutyrate
49. ((2r,3s,4r,5r)-3,4-dihydroxy-5-((z)-4-(hydroxyimino)-2-oxo-3,4-dihydropyrimidin-1(2h)-yl)tetrahydrofuran-2-yl)methyl Isobutyrate
50. [(2r,3s,4r,5r)-3,4-dihydroxy-5-[(4e)-4-(hydroxyimino)-2-oxo-1,2,3,4-tetrahydropyrimidin-1-yl]oxolan-2-yl]methyl 2-methylpropanoate
51. {(2r,3s,4r,5r)-3,4-dihydroxy-5-[4-(hydroxyimino)-2-oxo-3,4-dihydropyrimidin-1(2h)-yl]tetrahydrofuran-2-yl}methyl 2-methylpropanoate
| Molecular Weight | 329.31 g/mol |
|---|---|
| Molecular Formula | C13H19N3O7 |
| XLogP3 | -0.8 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Exact Mass | 329.12229995 g/mol |
| Monoisotopic Mass | 329.12229995 g/mol |
| Topological Polar Surface Area | 141 Ų |
| Heavy Atom Count | 23 |
| Formal Charge | 0 |
| Complexity | 534 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
[N4-hydroxycytidine] and its prodrug molnupiravir are being studied for its activity against a number of viral infections including influenza, MERS-CoV, and SARS-CoV-2. Molnupiravir is approved in the UK for reducing the risk of hospitalization and death in mild to moderate COVID-19 cases for patients at increased risk of severe disease (eg. with obesity, diabetes mellitus, heart disease, or are over 60 years old).
Treatment of Coronavirus disease 2019 (COVID-19)
Antiviral Agents
Agents used in the prophylaxis or therapy of VIRUS DISEASES. Some of the ways they may act include preventing viral replication by inhibiting viral DNA polymerase; binding to specific cell-surface receptors and inhibiting viral penetration or uncoating; inhibiting viral protein synthesis; or blocking late stages of virus assembly. (See all compounds classified as Antiviral Agents.)
Absorption
After an 800 mg oral dose of molnupiravir every 12 hours, the active compound (N4-hydroxycytidine) reaches a Cmax of 2970 ng/mL, with a Tmax of 1.5 hours, and an AUC0-12h of 8360 h\*ng/mL.
Route of Elimination
3% of an oral molnupiravir dose is eliminated in the urine as the active metabolite N4-hydroxycytidine.
Molnupiravir is hydrolyzed to [N4-hydroxycytidine], which distributes into tissues. Once inside cells, N4-hydroxycytidine is phosphorylated to the 5'-triphosphate form.
The half life of the active metabolite, N4-hydroxycytidine, is 3.3 hours.
Molnupiravir is hydrolyzed _in vivo_ to N4-hydroxycytidine, which is phosphorylated in tissue to the active 5-triphosphate form, and incorporated into the genome of new virions, resulting in the accumulation of inactivating mutations, known as viral error catastrophe. A [remdesivir] resistant mutant mouse hepatitis virus has also been shown to have increased sensitivity to N4-hydroxycytidine.
 Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.

NDC Package Code : 73435-018
Start Marketing Date : 2021-12-06
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (20kg/20kg)
Marketing Category : BULK INGREDIENT
 Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
 Gonane has API manufacturing expertise in new-age Corticosteroids, Hormones and other pharma raw materials.
Gonane has API manufacturing expertise in new-age Corticosteroids, Hormones and other pharma raw materials.
 Century has been an API manufacturer for over 30 years & is the partner of choice for multipurpose custom manufacturing projects.
Century has been an API manufacturer for over 30 years & is the partner of choice for multipurpose custom manufacturing projects.
 Tagoor's product development expertise, backed by our comprehensive understanding of the processes, helps us offer high-quality APIs.
Tagoor's product development expertise, backed by our comprehensive understanding of the processes, helps us offer high-quality APIs.
 Tofigh Daru develops & synthesizes a diverse range of APIs in Anticancer, Narcotics, Cardiovascular to Immunomodulatory Segments.
Tofigh Daru develops & synthesizes a diverse range of APIs in Anticancer, Narcotics, Cardiovascular to Immunomodulatory Segments.
 Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.

GDUFA
DMF Review : Reviewed
Rev. Date : 2021-08-27
Pay. Date : 2021-06-28
DMF Number : 36078
Submission : 2021-06-30
Status : Active
Type : II

NDC Package Code : 59285-040
Start Marketing Date : 2021-08-23
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 37087
Submission : 2022-05-06
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 36329
Submission : 2021-09-30
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 37435
Submission : 2022-08-25
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 36282
Submission : 2021-10-13
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 35960
Submission : 2021-05-31
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2021-08-27
Pay. Date : 2021-06-28
DMF Number : 36078
Submission : 2021-06-30
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 37087
Submission : 2022-05-06
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 36770
Submission : 2022-07-15
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Date of Issue : 2022-09-19
Valid Till : 2025-07-02
Written Confirmation Number : WC-0119
Address of the Firm : Plot No. 35, 36, 38, 39,40,49, 50 & 51, Phase-IV, IDA, Jeedimetla, Medchal- Malk...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
About the Company : Biophore, founded in 2007, has established itself as a reliable partner in the development and manufacturing of niche and complex pharmaceutical products. With 4 USFDA and EU-appro...
 Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
About the Company : Established in 2004, Metrochem API is one of the fastest-growing APIs, pellets & intermediates manufacturers. It has 6 dedicated manufacturing facilities for its 3 core product ...
 Gonane has API manufacturing expertise in new-age Corticosteroids, Hormones and other pharma raw materials.
Gonane has API manufacturing expertise in new-age Corticosteroids, Hormones and other pharma raw materials.
About the Company : Gonane Pharma, is a contract pharmaceutical company located in Gujarat, India, specializing in the manufacturing and marketing of Corticosteroids, Hormones, Antivirals, and Oncolog...
 Century has been an API manufacturer for over 30 years & is the partner of choice for multipurpose custom manufacturing projects.
Century has been an API manufacturer for over 30 years & is the partner of choice for multipurpose custom manufacturing projects.
About the Company : Century Pharmaceuticals, established in 1982, has 40 years of experience in manufacturing APIs. It has been supplying APIs produced in-house to several major pharma companies in In...
About the Company : HRV Global is a leading global manufacturer, seller & exporter of a wide range of APIs, advanced intermediates, pellets, food grade chemicals, food additives & food ingredients. It...
 Tagoor's product development expertise, backed by our comprehensive understanding of the processes, helps us offer high-quality APIs.
Tagoor's product development expertise, backed by our comprehensive understanding of the processes, helps us offer high-quality APIs.
About the Company : Tagoor Laboratories, established in 2018, is a part of the Tagoor Group. It specializes in providing APIs, advanced intermediates and key starting materials for critical and high-g...
 Tofigh Daru develops & synthesizes a diverse range of APIs in Anticancer, Narcotics, Cardiovascular to Immunomodulatory Segments.
Tofigh Daru develops & synthesizes a diverse range of APIs in Anticancer, Narcotics, Cardiovascular to Immunomodulatory Segments.
About the Company : Tofigh Daru (TODACO) belongs to the Tamin Pharmaceutical Investment Company, the biggest pharmaceutical holding in Iran. TODACO develops novel APIs in therapeutic categories like A...
 Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
About the Company : Omgene Life Sciences Private Limited is an R&D-driven biopharmaceutical company specializing in biopharmaceuticals, peptides, semi-synthetic, and synthetic actives. As a vertically...
About the Company : Hiray Pharma Solutions is an international end-to-end CDMO, facilitating the development and manufacturing of important drug products and key intermediates around the world. We pro...

About the Company : Mangalam Drugs & Organics Limited started manufacturing APIs and intermediates in 1977 in Gujarat, India. It has a multi-product manufacturing facility and an in-house R&D lab reco...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Details:
The licences are applicable for both molnupiravir (API) and finished product in capsules form. It enables the company to launch the product within India as well as export to 104 other countries around the world, Biophore India Pharmaceuticals said in a statement.
Lead Product(s): Molnupiravir
Therapeutic Area: Infections and Infectious Diseases Brand Name: MK-4482
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: The Medicines Patent Pool
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Licensing Agreement January 20, 2022
Lead Product(s) : Molnupiravir
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved
Partner/Sponsor/Collaborator : The Medicines Patent Pool
Deal Size : Undisclosed
Deal Type : Licensing Agreement
Biophore Receives Sub-License From MPP to Manufacture and Market Molnupiravir for Covid Treatment
Details : The licences are applicable for both molnupiravir (API) and finished product in capsules form. It enables the company to launch the product within India as well as export to 104 other countries around the world, Biophore India Pharmaceuticals said in a s...
Brand Name : MK-4482
Molecule Type : Small molecule
Upfront Cash : Undisclosed
January 20, 2022
Details:
Molflu a generic version of the Molnupiravir, is the first oral antiviral medication that helps treat mild to moderate COVID-19 in adults is expected to be available from next week in pharmacies throughout (India).
Lead Product(s): Molnupiravir
Therapeutic Area: Infections and Infectious Diseases Brand Name: Molflu
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable January 04, 2022
Lead Product(s) : Molnupiravir
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Merck's Anti- Covid Pill 'Molflu' 5-Day Course to Cost ₹ 1,400 In India
Details : Molflu a generic version of the Molnupiravir, is the first oral antiviral medication that helps treat mild to moderate COVID-19 in adults is expected to be available from next week in pharmacies throughout (India).
Brand Name : Molflu
Molecule Type : Small molecule
Upfront Cash : Not Applicable
January 04, 2022
Details:
Emergency-use authorisation recieved from DCGI to manufacture and market oral anti-viral drug Molflu (Molnupiravir) 200mg for treatment of adult patients with Covid19.
Lead Product(s): Molnupiravir
Therapeutic Area: Infections and Infectious Diseases Brand Name: Molflu
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable December 29, 2021
Lead Product(s) : Molnupiravir
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Dr Reddy’s Gets DCGI Nod for Molnupiravir, to Launch Soon
Details : Emergency-use authorisation recieved from DCGI to manufacture and market oral anti-viral drug Molflu (Molnupiravir) 200mg for treatment of adult patients with Covid19.
Brand Name : Molflu
Molecule Type : Small molecule
Upfront Cash : Not Applicable
December 29, 2021
Details:
Cipla Limited, Dr Reddy’s Laboratories Ltd, Emcure Pharmaceuticals, Sun Pharmaceutical Industries Limited and Torrent Pharmaceuticals will collaborate for the clinical trial of the oral anti-viral drug molnupiravir for mild Covid-19 in an outpatient setting in India.
Lead Product(s): Molnupiravir
Therapeutic Area: Infections and Infectious Diseases Brand Name: MK-4482
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Cipla
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Collaboration June 29, 2021
Lead Product(s) : Molnupiravir
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Cipla
Deal Size : Undisclosed
Deal Type : Collaboration
Details : Cipla Limited, Dr Reddy’s Laboratories Ltd, Emcure Pharmaceuticals, Sun Pharmaceutical Industries Limited and Torrent Pharmaceuticals will collaborate for the clinical trial of the oral anti-viral drug molnupiravir for mild Covid-19 in an outpatient se...
Brand Name : MK-4482
Molecule Type : Small molecule
Upfront Cash : Undisclosed
June 29, 2021
Details:
Fermenta Biotech will exclusively license its biocatalytic technology for synthesis of Molnupiravir (Molflu) and supply its enzyme Candida Antarctica B Lipase (CAL B) to Aurigene. Molnupiravir API manufactured by Aurigene will be utilised by Dr Reddy’s for its formulation.
Lead Product(s): Molnupiravir
Therapeutic Area: Infections and Infectious Diseases Brand Name: Molflu
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Fermenta Biotech Limited
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Licensing Agreement January 21, 2022
Lead Product(s) : Molnupiravir
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Fermenta Biotech Limited
Deal Size : Undisclosed
Deal Type : Licensing Agreement
Details : Fermenta Biotech will exclusively license its biocatalytic technology for synthesis of Molnupiravir (Molflu) and supply its enzyme Candida Antarctica B Lipase (CAL B) to Aurigene. Molnupiravir API manufactured by Aurigene will be utilised by Dr Reddy’s...
Brand Name : Molflu
Molecule Type : Small molecule
Upfront Cash : Undisclosed
January 21, 2022
Details:
DCGI approved Stripiravir (molnupiravir) an orally administered form of a potent ribonucleoside analog that inhibits the replication of SARS‐CoV‐2 in COVID-19 infection.
Lead Product(s): Molnupiravir
Therapeutic Area: Infections and Infectious Diseases Brand Name: Stripiravir
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable December 28, 2021
Lead Product(s) : Molnupiravir
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Details : DCGI approved Stripiravir (molnupiravir) an orally administered form of a potent ribonucleoside analog that inhibits the replication of SARS‐CoV‐2 in COVID-19 infection.
Brand Name : Stripiravir
Molecule Type : Small molecule
Upfront Cash : Not Applicable
December 28, 2021
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Lagevrio (molnupiravir) is an investigational, orally administered nucleoside analog that accumulates errors in the viral genome leading in inhibition of SARS-CoV-2 replication, the causative agent of COVID-19.
Lead Product(s): Molnupiravir
Therapeutic Area: Infections and Infectious Diseases Brand Name: Lagevrio
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Merck Group
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable February 24, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Molnupiravir
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Merck Group
Deal Size : Not Applicable
Deal Type : Not Applicable
Merck and Ridgeback Provide Update on EU Marketing Authorization Application for LAGEVRIO™ (Moln...
Details : Lagevrio (molnupiravir) is an investigational, orally administered nucleoside analog that accumulates errors in the viral genome leading in inhibition of SARS-CoV-2 replication, the causative agent of COVID-19.
Brand Name : Lagevrio
Molecule Type : Small molecule
Upfront Cash : Not Applicable
February 24, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Lagevrio (molnupiravir) is an investigational, orally administered nucleoside analog that accumulates errors in the viral genome leading in inhibition of SARS-CoV-2 replication, the causative agent of COVID-19.
Lead Product(s): Molnupiravir
Therapeutic Area: Infections and Infectious Diseases Brand Name: Lagevrio
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable February 21, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Molnupiravir
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Merck Provides Update on Phase 3 MOVe-AHEAD Trial Evaluating LAGEVRIO™ (molnupiravir) for Post-e...
Details : Lagevrio (molnupiravir) is an investigational, orally administered nucleoside analog that accumulates errors in the viral genome leading in inhibition of SARS-CoV-2 replication, the causative agent of COVID-19.
Brand Name : Lagevrio
Molecule Type : Small molecule
Upfront Cash : Not Applicable
February 21, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
A preliminary analysis in highly-vaccinated adults mostly <65 years of age, showed no evidence of difference between LAGEVRIO (molnupiravir) added to usual care compared to usual care alone for reduction of hospitalizations and deaths through Day 28.
Lead Product(s): Molnupiravir
Therapeutic Area: Infections and Infectious Diseases Brand Name: Lagevrio
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Ridgeback Biotherapeutics
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable October 06, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Molnupiravir
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Ridgeback Biotherapeutics
Deal Size : Not Applicable
Deal Type : Not Applicable
Details : A preliminary analysis in highly-vaccinated adults mostly <65 years of age, showed no evidence of difference between LAGEVRIO (molnupiravir) added to usual care compared to usual care alone for reduction of hospitalizations and deaths through Day 28.
Brand Name : Lagevrio
Molecule Type : Small molecule
Upfront Cash : Not Applicable
October 06, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Analyses of pre-specied exploratory endpoints indicate that a lower proportion of LAGEVRIO (Molnupiravir)-treated participants in MTT population had an acute care visit or a COVID-19-related acute care visit versus placebo-treated participants in the MITT population.
Lead Product(s): Molnupiravir
Therapeutic Area: Infections and Infectious Diseases Brand Name: Lagevrio
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Ridgeback Biotherapeutics
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable June 07, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Molnupiravir
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Ridgeback Biotherapeutics
Deal Size : Not Applicable
Deal Type : Not Applicable
Merck and Ridgeback Announce New Data For Investigational LAGEVRIO™ (molnupiravir) From Phase 3 ...
Details : Analyses of pre-specied exploratory endpoints indicate that a lower proportion of LAGEVRIO (Molnupiravir)-treated participants in MTT population had an acute care visit or a COVID-19-related acute care visit versus placebo-treated participants in the MIT...
Brand Name : Lagevrio
Molecule Type : Small molecule
Upfront Cash : Not Applicable
June 07, 2022

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]((3aR,4R,6R,6aR)-6-(4-(hydroxyamino)-2-oxopyrimidi...
CAS Number : 2346620-55-9
End Use API : Molnupiravir
About The Company : Shandong Chenghui Shuangda Pharmaceutical Co., Ltd. is specialized in R&D and production of APIs and advanced intermediates. With 22 years of production experie...
CAS Number : 3258-02-4
End Use API : Molnupiravir
About The Company : Kimia Biosciences, established in 1993, is focused on manufacturing bulk drugs for various therapeutic segments. With a strong emphasis on excellence and advanc...

(((3aR,4R,6R,6aR)6-(( 4 hydroxyimino 2 oxo 3,4 dih...
CAS Number : 2346620-55-9
End Use API : Molnupiravir
About The Company : Synaptics Labs Private Limited is a young Pharmaceutical Company with a fully integrated and globally specialized in the development, production and marketing o...

((3aR,4R,6R,6aR)-6-(4-(hydroxyamino)-2-oxopyrimidi...
CAS Number : 2346620-55-9
End Use API : Molnupiravir
About The Company : VIDGAS aims to provide our pharmaceutical, Bio pharma, Agrochemical, Food and Fine chemical customers with the highest quality SFC and HPLC purification and iso...

((3aR,4R,6R,6aR)-6-(2,4-dioxo- 3,4-dihydropyrimidi...
CAS Number : 2346620-53-7
End Use API : Molnupiravir
About The Company : VIDGAS aims to provide our pharmaceutical, Bio pharma, Agrochemical, Food and Fine chemical customers with the highest quality SFC and HPLC purification and iso...

((3aR,4R,6R,6aR)-2,2-dimethyl-6-(2-oxo-4-(1H-1,2,4...
CAS Number : 2346620-54-8
End Use API : Molnupiravir
About The Company : VIDGAS aims to provide our pharmaceutical, Bio pharma, Agrochemical, Food and Fine chemical customers with the highest quality SFC and HPLC purification and iso...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Octavius has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Octavius has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Capsule
Dosage Strength : 200MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Octavius has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Octavius has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Capsule
Dosage Strength : 400MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Capsule
Dosage Strength : 200MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Australia
Brand Name : Lagevrio
Dosage Form :
Dosage Strength :
Packaging : 40
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Australia

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Australia
Brand Name : Lagevrio
Dosage Form :
Dosage Strength :
Packaging : 40
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Australia

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Egypt
Brand Name : Jeravir
Dosage Form : Capsule
Dosage Strength : 200MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Egypt

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Turkey
Brand Name : MOLNUVIR
Dosage Form : Capsule
Dosage Strength : 200MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Turkey

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Turkey
Brand Name : MOLNUVIR
Dosage Form : Capsule
Dosage Strength : 400MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Turkey

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Turkey
Brand Name : MOLNUVIR
Dosage Form : Capsule
Dosage Strength : 200MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Turkey

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Turkey
Brand Name : MOLNUVIR
Dosage Form : Capsule
Dosage Strength : 400MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Turkey

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
We have 33 companies offering Molnupiravir
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
