

Synopsis
Synopsis
0
JDMF
0
VMF
0
FDF
0
FDA Orange Book

0
Australia

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
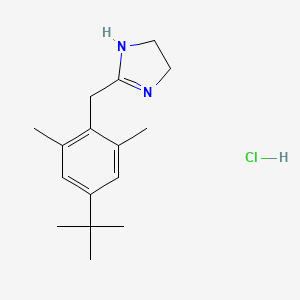

1. 2-(4'-tert-butyl-2',6'-dimethylphenylmethyl)imidazoline
2. Amidrin
3. Balkis
4. Chlorohist-la
5. Decongest
6. Espa-rhin
7. Gelonasal
8. Idasal
9. Idril N
10. Imidin
11. Nasan
12. Nasengel Al
13. Nasengel Ratiopharm
14. Nasenspray Al
15. Nasenspray Ratiopharm
16. Nasentropfen Al
17. Nasentropfen Ratiopharm
18. Novorin
19. Otradrops
20. Otraspray
21. Otriven
22. Otrivin
23. Otrivin Mentol
24. Rapako
25. Schnupfen Endrine
26. Snup
27. Stas
28. Xylometazoline
29. Xylometazoline Monohydrochloride
1. 1218-35-5
2. Xylometazoline Hcl
3. Xylometazoline (hydrochloride)
4. 2-(4-tert-butyl-2,6-dimethylbenzyl)-2-imidazoline Hydrochloride
5. Neo-synephrine Ii
6. Otrivin Hydrochloride
7. Nsc-757378
8. Mls000069695
9. Smr000058526
10. 2-(4-(tert-butyl)-2,6-dimethylbenzyl)-4,5-dihydro-1h-imidazole Hydrochloride
11. Dsstox_cid_25223
12. Dsstox_rid_80760
13. Dsstox_gsid_45223
14. 1h-imidazole, 2-[[4-(1,1-dimethylethyl)-2,6-dimethylphenyl]methyl]-4,5-dihydro-, Hydrochloride (1:1)
15. 2-(4-tert-butyl-2,6-dimethylbenzyl)-4,5-dihydro-1h-imidazole Hydrochloride
16. X5s84033nz
17. Xylometazoline Hydrochloride (usp)
18. 2-(4-tert-butyl-2,6-dimethylbenzyl)-2-imidazoline Monohydrochloride
19. 2-[(4-tert-butyl-2,6-dimethylphenyl)methyl]-4,5-dihydro-1h-imidazole;hydrochloride
20. Sr-01000000230
21. Ncgc00016101-02
22. Cas-1218-35-5
23. Mfcd00238707
24. Prestwick_574
25. Opera_id_1744
26. Neo-synephrine Ii (tn)
27. Schembl41837
28. Mls001076518
29. Spectrum1500614
30. Chembl1256400
31. Dtxsid3045223
32. Regid_for_cid_5282386
33. Hms1568n07
34. Hms1921k09
35. Pharmakon1600-01500614
36. Xylometazoline Hydrochloride ,(s)
37. Bcp22148
38. Hy-b0475
39. Tox21_110305
40. Tox21_501269
41. Ccg-39900
42. Nsc757378
43. S2576
44. Akos015895456
45. Tox21_110305_1
46. Ac-8301
47. Ks-5144
48. Lp01269
49. Ncgc00016101-10
50. Ncgc00094506-01
51. Ncgc00094506-02
52. Ncgc00094506-03
53. Ncgc00094506-04
54. Ncgc00094506-05
55. Ncgc00261954-01
56. Xylometazoline Hydrochloride [mi]
57. Xylometazoline Hydrochloride [mart.]
58. Xylometazoline Hydrochloride [vandf]
59. Eu-0101269
60. Ft-0603554
61. Sw196748-3
62. X0063
63. Xylometazoline Hydrochloride [usp-rs]
64. Xylometazoline Hydrochloride [who-dd]
65. D00757
66. X 6000
67. Xylometazoline Hydrochloride, Analytical Standard
68. 218x355
69. A804796
70. Xylometazoline Hydrochloride [ep Monograph]
71. J-004686
72. Sr-01000000230-2
73. Sr-01000000230-7
74. Xylometazoline Hydrochloride [usp Monograph]
75. Q27293585
76. Xylometazoline Hydrochloride, British Pharmacopoeia (bp) Reference Standard
77. Xylometazoline Hydrochloride, European Pharmacopoeia (ep) Reference Standard
78. Xylometazoline Hydrochloride, Pharmaceutical Secondary Standard; Certified Reference Material
79. Xylometazoline Hydrochloride, United States Pharmacopeia (usp) Reference Standard
| Molecular Weight | 280.83 g/mol |
|---|---|
| Molecular Formula | C16H25ClN2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 3 |
| Exact Mass | 280.1706265 g/mol |
| Monoisotopic Mass | 280.1706265 g/mol |
| Topological Polar Surface Area | 24.4 Ų |
| Heavy Atom Count | 19 |
| Formal Charge | 0 |
| Complexity | 302 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Nasal Decongestants
Drugs designed to treat inflammation of the nasal passages, generally the result of an infection (more often than not the common cold) or an allergy related condition, e.g., hay fever. The inflammation involves swelling of the mucous membrane that lines the nasal passages and results in inordinate mucus production. The primary class of nasal decongestants are vasoconstrictor agents. (From PharmAssist, The Family Guide to Health and Medicine, 1993) (See all compounds classified as Nasal Decongestants.)
 Polpharma is a Polish CDMO of APIs and a significant European API producer, delivering products to companies worldwide.
Polpharma is a Polish CDMO of APIs and a significant European API producer, delivering products to companies worldwide.

Certificate Number : R1-CEP 2011-369 - Rev 01
Issue Date : 2020-03-03
Type : Chemical
Substance Number : 1162
Status : Valid

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 22045
Submission : 2008-09-19
Status : Active
Type : II

Certificate Number : R1-CEP 2008-255 - Rev 02
Issue Date : 2017-09-18
Type : Chemical
Substance Number : 1162
Status : Valid

Date of Issue : 2022-06-24
Valid Till : 2025-07-02
Written Confirmation Number : WC-0097
Address of the Firm : Block No. 251/P, 252/P, 253 to 255, 256/P,258/P, 276/P, 277/P, 278/P, 279 To 282,283/P, 284/P, GICD, Sachin, Surat, Gujrat

NDC Package Code : 42419-004
Start Marketing Date : 2008-09-19
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (25kg/25kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Sungwoo Chemical Co., Ltd.
Registration Date : 2020-03-11
Registration Number : 20140121-189-I-379-03(6)
Manufacturer Name : CTX Life Sciences Pvt. Ltd.
Manufacturer Address : Block No. 251/P, 252/P, GIDC, City : Sachin, Dist : Surat, India


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 9104
Submission : 1991-05-15
Status : Active
Type : II

Certificate Number : R1-CEP 2000-061 - Rev 03
Issue Date : 2015-12-10
Type : Chemical
Substance Number : 1162
Status : Valid

Registrant Name : CTC Bio Co., Ltd.
Registration Date : 2019-10-28
Registration Number : 20130806-189-I-304-02(1)
Manufacturer Name : Siegfried PharmaChemikalien Minden GmbH
Manufacturer Address : Karlstrasse 15-39, 42-44, 32423 Minden, North Rhine-Westphalia, Germany

USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 2691
Submission : 1976-05-27
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 2498
Submission : 1975-06-18
Status : Inactive
Type : II



Certificate Number : R0-CEP 2020-413 - Rev 00
Issue Date : 2022-12-20
Type : Chemical
Substance Number : 1162
Status : Valid


Certificate Number : R1-CEP 2006-286 - Rev 01
Issue Date : 2015-12-15
Type : Chemical
Substance Number : 1162
Status : Valid

Registrant Name : CTC Bio Co., Ltd.
Registration Date : 2019-10-28
Registration Number : 20130806-189-I-304-02(1)
Manufacturer Name : Siegfried PharmaChemikalien Minden GmbH
Manufacturer Address : Karlstrasse 15-39, 42-44, 32423 Minden, North Rhine-Westphalia, Germany

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name : XYLORIL PAED SPRA
Dosage Form : Nasal Spray
Dosage Strength : 0.05%/W/V
Packaging : 10 ml bottle
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : OTC
Registration Country : Canada
Brand Name : NASAL DECONGESTANT SPRAY WITH MOISTURIZERS
Dosage Form : SPRAY
Dosage Strength : 0.1%/W/V
Packaging : 30ML
Approval Date :
Application Number : 2452863
Regulatory Info : OTC
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Vietnam
Brand Name : Jazxylo
Dosage Form : Nasal Spray
Dosage Strength : 15MG/15ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Vietnam

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : OTC
Registration Country : Canada
Brand Name : OTRIVIN MEDICATED COMPLETE NASAL CARE
Dosage Form : SPRAY
Dosage Strength : 0.1%/W/V
Packaging : 20ML
Approval Date :
Application Number : 2331403
Regulatory Info : OTC
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Originator
Registration Country : South Africa
Brand Name : Sinutab
Dosage Form : SOL
Dosage Strength : 1mg/ml
Packaging : 1X1mg/ml
Approval Date :
Application Number :
Regulatory Info : Originator
Registration Country : South Africa

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : India
Brand Name : Nasomist-XP
Dosage Form : NASAL DROPS
Dosage Strength : 0.05G/100ML
Packaging :
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Rinosedin
Dosage Form : Gtt Nas
Dosage Strength : 0.05%
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Rinosedin
Dosage Form : Nasenspray
Dosage Strength : 0.10%
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : Zymelin Menthol
Dosage Form : NASAL SPRAY, SOLUTION
Dosage Strength : 1 MG / ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : Turkey
Xylometazoline Hydrochloride; Dexpanthenol
Brand Name :
Dosage Form : NASAL SPRAY
Dosage Strength : 1MG; 50MG
Packaging : 10 Ml/Bottle
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : Turkey

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : Klarimax
Dosage Form : NASAL SPRAY, SOLUTION
Dosage Strength : 1 MG / ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : Nasoferm
Dosage Form : NASAL SPRAY, SOLUTION
Dosage Strength : 1 MG / ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Xylometazoline hydrochloride; Dexpanthenol
Brand Name : Xylo Dexpanthenol Spirig HC
Dosage Form : Metered Spray
Dosage Strength :
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Rinosedin
Dosage Form : Gtt Nas
Dosage Strength : 0.05%
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Rinosedin
Dosage Form : Gtt Nas
Dosage Strength : 0.10%
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : Zymelin
Dosage Form : NASAL SPRAY, SOLUTION
Dosage Strength : 1 MG / ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : Zymelin Menthol
Dosage Form : NASAL SPRAY, SOLUTION
Dosage Strength : 1 MG / ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : Turkey
Xylometazoline Hydrochloride; Dexpanthenol
Brand Name :
Dosage Form : NASAL SPRAY
Dosage Strength : 1MG; 50MG
Packaging : 10 Ml/Bottle
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : Turkey

Regulatory Info :
Registration Country : Sweden
Brand Name : Teppix
Dosage Form : NASAL SPRAY, SOLUTION
Dosage Strength : 1 MG / ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Sweden
Brand Name : Teppix
Dosage Form : NASAL SPRAY, SOLUTION
Dosage Strength : 0.5 MG / ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
