
Synopsis
Synopsis
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book

0
Canada

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
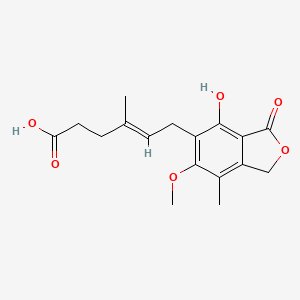

1. Cellcept
2. Mofetil Hydrochloride, Mycophenolate
3. Mofetil, Mycophenolate
4. Mycophenolate Mofetil
5. Mycophenolate Mofetil Hydrochloride
6. Mycophenolate Sodium
7. Mycophenolate, Sodium
8. Mycophenolic Acid Morpholinoethyl Ester
9. Myfortic
10. Rs 61443
11. Rs-61443
12. Rs61443
13. Sodium Mycophenolate
1. 24280-93-1
2. Mycophenolate
3. Myfortic
4. Melbex
5. Mycophenolsaeure
6. 483-60-3
7. Lilly-68618
8. 6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-5-phthalanyl)-4-methyl-4-hexenoic Acid
9. Nsc-129185
10. Acido Micofenolico
11. Micofenolico Acido
12. Acide Mycophenolique
13. Acidum Mycophenolicum
14. (4e)-6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-1,3-dihydro-2-benzofuran-5-yl)-4-methylhex-4-enoic Acid
15. Ccris 5565
16. Nsc 129185
17. Ly 68618
18. (e)-6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-5-phthalanyl)-4-methyl-4-hexenoic Acid
19. (e)-6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-1,3-dihydroisobenzofuran-5-yl)-4-methylhex-4-enoic Acid
20. (e)-6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-1h-2-benzofuran-5-yl)-4-methylhex-4-enoic Acid
21. Mfcd00036814
22. Lilly 68618
23. 4-hexenoic Acid, 6-(1,3-dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-, (e)-
24. Chembl866
25. Hu9dx48n0t
26. 6-(1,3-dihydro-7-hydroxy-5-methoxy-4-methyl-1-oxoisobenzofuran-6-yl)-4-methyl-4-hexanoic Acid
27. Chebi:168396
28. Tnp00198
29. Rs-61443 [as Mofetil]
30. Nsc129185
31. 4-hexenoic Acid, 6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-5-phthalanyl)-4-methyl-, (e)-
32. 4-methyl-5-methoxy-7-hydroxy-6-(5-carboxy-3-methylpent-2-en-1-yl)-phthalide (e)-
33. 6-(1,3-dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methylhex-4-enoic Acid
34. 6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-5-phthalanyl)-4-methyl-4-hexenoic Acid (e)-
35. 4-hexenoic Acid, 6-(1,3-dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-, (4e)-
36. Ncgc00016786-01
37. Cas-24280-93-1
38. Mycophenolic Acid 100 Microg/ml In Acetonitrile
39. Dsstox_cid_21070
40. Dsstox_rid_79619
41. Dsstox_gsid_41070
42. Mycophenolic Acid-d3 Solution
43. Mycophenoic Acid
44. 6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-1h-isobenzofuran-5-yl)-4-methyl-4-hexenoic Acid
45. Micofenolico Acido [spanish]
46. (4e)-6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-1,3-dihydroisobenzofuran-5-yl)-4-methylhex-4-enoic Acid (mycophenolic Acid)
47. (e)-6-(1,3-dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-4-hexenoic Acid
48. 4-hexenoic Acid,6-(1,3-dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-, (4e)-
49. 6-(1,3-dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-4-hexenoic Acid
50. 6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-1,3-dihydroisobenzofuran-5-yl)-4-methylhex-4-enoic Acid
51. Moa
52. Smr000471887
53. Acido Micofenolico [inn-spanish]
54. Acide Mycophenolique [inn-french]
55. Acidum Mycophenolicum [inn-latin]
56. Sr-01000597602
57. Einecs 246-119-3
58. Unii-hu9dx48n0t
59. Mycophenolic-acid
60. Prestwick_817
61. Einecs 207-595-8
62. Mycophenolic Acid [usan:inn:ban]
63. Tocris-1505
64. (z)-mycophenolic Acid
65. 1jr1
66. Starbld0040186
67. Mycophenolic Acid (tn)
68. Prestwick2_000556
69. Prestwick3_000556
70. Spectrum5_001654
71. Mycophenolic Acid (mpa)
72. Mycophenolic Acid Solution
73. Upcmld-dp028
74. 4-hexenoic Acid, (e)-
75. Ec 246-119-3
76. Mycophenolate;rs-61443
77. Schembl4549
78. Mycophenolic (mycophenolate)
79. Mycophenolic Acid, >=98%
80. Bspbio_000631
81. Bspbio_002534
82. Mycophenolate [vandf]
83. Mls001074701
84. Mls002222265
85. Mls002695945
86. Bidd:gt0456
87. Spectrum1500674
88. Mycophenolic Acid [mi]
89. Bpbio1_000695
90. Gtpl6832
91. Megxm0_000120
92. Mycophenolic Acid (usan/inn)
93. Myfortic (mycophenolate Sodium)
94. Schembl2514376
95. Zinc1758
96. Mycophenolic Acid [inn]
97. Dtxsid4041070
98. Mycophenolic Acid [usan]
99. Upcmld-dp028:001
100. Acon0_000960
101. Acon1_000496
102. Bdbm19264
103. Chebi:92545
104. Hms500h08
105. Ex-a975
106. Mycophenolic Acid [vandf]
107. Mycophenolic Acid [mart.]
108. Hms1569p13
109. Hms1921a18
110. Hms2089a17
111. Hms2092g22
112. Hms2096p13
113. Hms2268h22
114. Hms3403p09
115. Hms3412f09
116. Hms3676f09
117. Pharmakon1600-01500674
118. Mycophenolic Acid [who-dd]
119. Act02623
120. Amy40494
121. Bcp05321
122. Hy-b0421
123. Tox21_110610
124. Bbl034696
125. Ccg-39914
126. Nsc757424
127. S2487
128. Stl419986
129. Mycophenolic Acid, Analytical Standard
130. Akos015888214
131. Tox21_110610_1
132. Ac-4491
133. Bcp9000970
134. Db01024
135. Ds-1638
136. Mycophenolic Acid [orange Book]
137. Nsc-757424
138. Sdccgmls-0066618.p001
139. (e)-6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-1h-isobenzofuran-5-yl)-4-methyl-hex-4-enoic Acid
140. Idi1_000146
141. Ncgc00016786-02
142. Ncgc00016786-03
143. Ncgc00016786-04
144. Ncgc00016786-05
145. Ncgc00016786-06
146. Ncgc00016786-07
147. Ncgc00016786-08
148. Ncgc00016786-09
149. Ncgc00016786-10
150. Ncgc00016786-11
151. Ncgc00016786-12
152. Ncgc00016786-15
153. Ncgc00025190-01
154. Ncgc00025190-02
155. Ncgc00025190-03
156. Ncgc00025190-04
157. Ncgc00025190-05
158. Ncgc00025190-07
159. Ncgc00025190-08
160. Ncgc00025190-09
161. Ncgc00025190-10
162. (4e)-6-(4-hydroxy-6-methoxy-7-methyl-3-oxohydroisobenzofuran-5-yl)-4-methylhex -4-enoic Acid
163. Bm164624
164. Mycophenolic Acid - Cas 24280-93-1
165. Sbi-0051945.p003
166. A-249
167. M2216
168. Sw196951-2
169. C20380
170. D05096
171. M 5255
172. M02087
173. Ab00052466-13
174. Ab00052466-14
175. Ab00052466_15
176. Ab00052466_16
177. 280m931
178. A817192
179. Mycophenolate Mofetil Impurity, Mycophenolic Acid-
180. Q420553
181. Sr-01000597602-1
182. Sr-01000597602-3
183. Sr-01000597602-4
184. Brd-k63750851-001-06-6
185. Brd-k63750851-001-13-2
186. Mycophenolate Mofetil Impurity F [ep Impurity]
187. Mycophenolate Mofetil Impurity, Mycophenolic Acid- [usp Impurity]
188. Mycophenolic Acid, Powder, Bioreagent, Suitable For Cell Culture, >=98%
189. 6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-5-phthalanyl)-4-methyl-4-hexenoic Acid;nsc 129185
190. 6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-5-phthanlanyl)-4-methyl-4-hexanoic Acid
191. (e)-6-(1,3-dihydro-4-hydroxy-6- Methoxy-7-methyl-3-oxoisobenzofuran-5-yl)-4-methyl-4-hexenoic Acid
192. (e)-6-(1,3-dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxoisobenzofuran-5-yl)-4-methyl-4-hexenoic Acid
193. 1162256-90-7
194. 4-hexenoic Acid, 6-(1,3-dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-, (4e)- (9ci)
195. 4-hexenoic Acid, 6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-5-phthalanyl)-4-methyl-, (e)- (8ci)
196. 4-hexenoic Acid,3-dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5- Isobenzofuranyl)-4-methyl-, (e)-
197. 4-methyl-6-[3-oxo-7-methyl-4-hydroxy-6-methoxy-1,3-dihydroisobenzofuran-5-yl]-4-hexenoic Acid
198. E-6-(1,3-dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-4-hexenoic Acid
199. Mycophenolic Acid Solution, 1.0 Mg/ml In Acetonitrile, Ampule Of 1 Ml, Certified Reference Material
| Molecular Weight | 320.3 g/mol |
|---|---|
| Molecular Formula | C17H20O6 |
| XLogP3 | 3.2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 6 |
| Exact Mass | 320.12598835 g/mol |
| Monoisotopic Mass | 320.12598835 g/mol |
| Topological Polar Surface Area | 93.1 Ų |
| Heavy Atom Count | 23 |
| Formal Charge | 0 |
| Complexity | 486 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 4 | |
|---|---|
| Drug Name | Mycophenolic acid |
| PubMed Health | Mycophenolate |
| Drug Classes | Immune Suppressant |
| Drug Label | Mycophenolic acid delayed-release tablets are an enteric formulation of mycophenolate sodium that delivers the active moiety mycophenolic acid (MPA). Mycophenolic acid is an immunosuppressive agent. As the sodium salt, MPA is chemically designated as... |
| Active Ingredient | Mycophenolic acid |
| Dosage Form | Tablet, delayed release |
| Route | Oral |
| Strength | 180mg; 360mg |
| Market Status | Prescription |
| Company | Mylan Pharms; Apotex |
| 2 of 4 | |
|---|---|
| Drug Name | Myfortic |
| Active Ingredient | Mycophenolic acid |
| Dosage Form | Tablet, delayed release |
| Route | Oral |
| Strength | 180mg; 360mg |
| Market Status | Prescription |
| Company | Novartis |
| 3 of 4 | |
|---|---|
| Drug Name | Mycophenolic acid |
| PubMed Health | Mycophenolate |
| Drug Classes | Immune Suppressant |
| Drug Label | Mycophenolic acid delayed-release tablets are an enteric formulation of mycophenolate sodium that delivers the active moiety mycophenolic acid (MPA). Mycophenolic acid is an immunosuppressive agent. As the sodium salt, MPA is chemically designated as... |
| Active Ingredient | Mycophenolic acid |
| Dosage Form | Tablet, delayed release |
| Route | Oral |
| Strength | 180mg; 360mg |
| Market Status | Prescription |
| Company | Mylan Pharms; Apotex |
| 4 of 4 | |
|---|---|
| Drug Name | Myfortic |
| Active Ingredient | Mycophenolic acid |
| Dosage Form | Tablet, delayed release |
| Route | Oral |
| Strength | 180mg; 360mg |
| Market Status | Prescription |
| Company | Novartis |
For the prophylaxis of organ rejection in patients receiving allogeneic renal transplants, administered in combination with cyclosporine and corticosteroids.
Mycophenolic acid is an antibiotic substance derived from Penicillium stoloniferum. It blocks de novo biosynthesis of purine nucleotides by inhibition of the enzyme inosine monophosphate dehydrogenase. Mycophenolic acid is important because of its selective effects on the immune system. It prevents the proliferation of T-cells, lymphocytes, and the formation of antibodies from B-cells. It also may inhibit recruitment of leukocytes to inflammatory sites.
Antibiotics, Antitubercular
Substances obtained from various species of microorganisms that are, alone or in combination with other agents, of use in treating various forms of tuberculosis; most of these agents are merely bacteriostatic, induce resistance in the organisms, and may be toxic. (See all compounds classified as Antibiotics, Antitubercular.)
Antibiotics, Antineoplastic
Chemical substances, produced by microorganisms, inhibiting or preventing the proliferation of neoplasms. (See all compounds classified as Antibiotics, Antineoplastic.)
Enzyme Inhibitors
Compounds or agents that combine with an enzyme in such a manner as to prevent the normal substrate-enzyme combination and the catalytic reaction. (See all compounds classified as Enzyme Inhibitors.)
L - Antineoplastic and immunomodulating agents
L04 - Immunosuppressants
L04A - Immunosuppressants
L04AA - Selective immunosuppressants
L04AA06 - Mycophenolic acid
Absorption
Bioavailability following oral administration of Myfortic delayed-release tablet ranges from 70-95%
Volume of Distribution
54 25 L
Clearance
140 +/- 30 mL/min [Stable renal transplant patients]
Mycophenolic acid is metabolized mainly by glucuronyl transferase to glucuronidated metabolites, predominantly the phenolic glucuronide, mycophenolic acid glucuronide (MPAG). MPAG does not manifest pharmacological activity. The acyl glucuronide minor metabolite has pharmacological activity similar to mycophenolic acid. The AUC ratio of Mycophenolic acid:MPAG:acyl glucuronide is approximately 1:24:0.28 at steady state.
Mycophenolic acid has known human metabolites that include 6-O-desmethyl-MPA (DM-MPA) and Mycophenolic acid glucuronide.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
The mean elimination half-life for mycophenolic acid ranges from 8-16 hours, while that of the MPAG metabolite ranges from 13-17 hours.
Mycophenolic acid is a potent, selective, uncompetitive, and reversible inhibitor of inosine monophosphate dehydrogenase (IMPDH), and therefore inhibits the de novo pathway of guanosine nucleotide synthesis without incorporation into DNA. Because T- and B-lymphocytes are critically dependent for their proliferation on de novo synthesis of purines, whereas other cell types can utilize salvage pathways, mycophenolic acid has potent cytostatic effects on lymphocytes. Mycophenolic acid inhibits proliferative responses of T- and B-lymphocytes to both mitogenic and allospecific stimulation. Addition of guanosine or deoxyguanosine reverses the cytostatic effects of mycophenolic acid on lymphocytes. Mycophenolic acid also suppresses antibody formation by B-lymphocytes. Mycophenolic acid prevents the glycosylation of lymphocyte and monocyte glycoproteins that are involved in intercellular adhesion to endothelial cells and may inhibit recruitment of leukocytes into sites of inflammation and graft rejection.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

ABOUT THIS PAGE
74
PharmaCompass offers a list of Mycophenolic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Mycophenolic Acid manufacturer or Mycophenolic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Mycophenolic Acid manufacturer or Mycophenolic Acid supplier.
PharmaCompass also assists you with knowing the Mycophenolic Acid API Price utilized in the formulation of products. Mycophenolic Acid API Price is not always fixed or binding as the Mycophenolic Acid Price is obtained through a variety of data sources. The Mycophenolic Acid Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Mycophenolic Acid manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Mycophenolic Acid, including repackagers and relabelers. The FDA regulates Mycophenolic Acid manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Mycophenolic Acid API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Mycophenolic Acid manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Mycophenolic Acid supplier is an individual or a company that provides Mycophenolic Acid active pharmaceutical ingredient (API) or Mycophenolic Acid finished formulations upon request. The Mycophenolic Acid suppliers may include Mycophenolic Acid API manufacturers, exporters, distributors and traders.
click here to find a list of Mycophenolic Acid suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Mycophenolic Acid DMF (Drug Master File) is a document detailing the whole manufacturing process of Mycophenolic Acid active pharmaceutical ingredient (API) in detail. Different forms of Mycophenolic Acid DMFs exist exist since differing nations have different regulations, such as Mycophenolic Acid USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Mycophenolic Acid DMF submitted to regulatory agencies in the US is known as a USDMF. Mycophenolic Acid USDMF includes data on Mycophenolic Acid's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Mycophenolic Acid USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Mycophenolic Acid suppliers with USDMF on PharmaCompass.
A Mycophenolic Acid CEP of the European Pharmacopoeia monograph is often referred to as a Mycophenolic Acid Certificate of Suitability (COS). The purpose of a Mycophenolic Acid CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Mycophenolic Acid EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Mycophenolic Acid to their clients by showing that a Mycophenolic Acid CEP has been issued for it. The manufacturer submits a Mycophenolic Acid CEP (COS) as part of the market authorization procedure, and it takes on the role of a Mycophenolic Acid CEP holder for the record. Additionally, the data presented in the Mycophenolic Acid CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Mycophenolic Acid DMF.
A Mycophenolic Acid CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Mycophenolic Acid CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Mycophenolic Acid suppliers with CEP (COS) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Mycophenolic Acid as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Mycophenolic Acid API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Mycophenolic Acid as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Mycophenolic Acid and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Mycophenolic Acid NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Mycophenolic Acid suppliers with NDC on PharmaCompass.
Mycophenolic Acid Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Mycophenolic Acid GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Mycophenolic Acid GMP manufacturer or Mycophenolic Acid GMP API supplier for your needs.
A Mycophenolic Acid CoA (Certificate of Analysis) is a formal document that attests to Mycophenolic Acid's compliance with Mycophenolic Acid specifications and serves as a tool for batch-level quality control.
Mycophenolic Acid CoA mostly includes findings from lab analyses of a specific batch. For each Mycophenolic Acid CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Mycophenolic Acid may be tested according to a variety of international standards, such as European Pharmacopoeia (Mycophenolic Acid EP), Mycophenolic Acid JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Mycophenolic Acid USP).
