

Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
FDF
0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
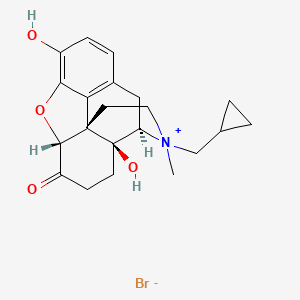

1. (5alpha)-17-(cyclopropylmethyl)-3,14-dihydroxy-17-methyl-4,5-epoxymorphinan-17-ium-6-one
2. 17-(cyclopropylmethyl)-4,5-epoxy-3,14-dihydroxymorphinanium-6-one
3. Methyl-naltrexone Hydrobromide
4. Methylnaltrexone
5. Morphinan-17-ium-6-one, 17-(cyclopropylmethyl)-4,5-epoxy-3,14-dihydroxy-17-methyl-, (5alpha)-
6. Morphinan-17-ium-6-one, 17-(cyclopropylmethyl)-4,5-epoxy-3,14-dihydroxy-17-methyl-, Bromide, (5alpha,17r)-
7. Morphinanium, 17-(cyclopropylmethyl)-4,5-epoxy-3,14-dihydroxy-17-methyl-6-oxo-, (5alpha)-
8. Mrz 2663br
9. Mrz-2663
10. N-methylnaltrexone Bromide
11. Naltrexone Mb
12. Naltrexone Methobromide
13. Naltrexone Methylbromide
14. Naltrexonium Methiodide
15. Quaternary Ammonium Naltrexone
16. Relistor
1. Naltrexone Methobromide
2. 73232-52-7
3. Relistor
4. Methylnaltrexone (bromide)
5. Methylnaltrexone
6. Moa-728
7. N-methylnaltrexone Bromide
8. Mrz-2663br
9. Mntx
10. Moa 728
11. Relistor (tn)
12. N-cyclopropylmethyl-noroxymorphone Methobromide
13. Relistormoa-728
14. Mntx-302
15. Moa728
16. Schembl2798188
17. Chembl1201770
18. Dtxsid30868236
19. Ono-3849
20. Methylnaltrexone Bromide (jan/usan)
21. Akos015896214
22. Cs-0762
23. (4r,4as,7ar,12bs)-3-(cyclopropylmethyl)-4a,9-dihydroxy-3-methyl-2,4,5,6,7a,13-hexahydro-1h-4,12-methanobenzofuro[3,2-e]isoquinoline-3-ium-7-one,bromide
24. Ac-32482
25. Hy-75766
26. Methylnaltrexone Bromide, >=97% (hplc)
27. D06618
28. Methylnaltrexone Bromide, United States Pharmacopeia (usp) Reference Standard
29. (4r,4as,7ar,12bs)-3-(cyclopropylmethyl)-4a,9-dihydroxy-3-methyl-2,4,5,6,7a,13-hexahydro-1h-4,12-methanobenzofuro[3,2-e]isoquinolin-3-ium-7-one;bromide
| Molecular Weight | 436.3 g/mol |
|---|---|
| Molecular Formula | C21H26BrNO4 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Exact Mass | 435.10452 g/mol |
| Monoisotopic Mass | 435.10452 g/mol |
| Topological Polar Surface Area | 66.8 Ų |
| Heavy Atom Count | 27 |
| Formal Charge | 0 |
| Complexity | 664 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Relistor |
| PubMed Health | Methylnaltrexone (Injection) |
| Drug Classes | Gastrointestinal Agent |
| Drug Label | RELISTOR (methylnaltrexone bromide) injection, a peripherally-acting mu-opioid receptor antagonist, is a sterile, clear and colorless to pale yellow aqueous solution. The chemical name for methylnaltrexone bromide is (R)-N-(cyclopropylmethyl) noroxym... |
| Active Ingredient | Methylnaltrexone bromide |
| Dosage Form | Injectable; Solution |
| Route | Subcutaneous |
| Strength | 8mg/0.4ml; 12mg/0.6ml (12mg/0.6ml) |
| Market Status | Prescription |
| Company | Salix Pharms |
| 2 of 2 | |
|---|---|
| Drug Name | Relistor |
| PubMed Health | Methylnaltrexone (Injection) |
| Drug Classes | Gastrointestinal Agent |
| Drug Label | RELISTOR (methylnaltrexone bromide) injection, a peripherally-acting mu-opioid receptor antagonist, is a sterile, clear and colorless to pale yellow aqueous solution. The chemical name for methylnaltrexone bromide is (R)-N-(cyclopropylmethyl) noroxym... |
| Active Ingredient | Methylnaltrexone bromide |
| Dosage Form | Injectable; Solution |
| Route | Subcutaneous |
| Strength | 8mg/0.4ml; 12mg/0.6ml (12mg/0.6ml) |
| Market Status | Prescription |
| Company | Salix Pharms |
Treatment of opioid-induced constipation in advanced-illness patients who are receiving palliative care when response to usual laxative therapy has not been sufficient.
Narcotic Antagonists
Agents inhibiting the effect of narcotics on the central nervous system. (See all compounds classified as Narcotic Antagonists.)
A06AH01
A06AH01
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
A - Alimentary tract and metabolism
A06 - Drugs for constipation
A06A - Drugs for constipation
A06AH - Peripheral opioid receptor antagonists
A06AH01 - Methylnaltrexone bromide
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
 Faran Shimi: Leading producer of high-quality APIs & alkaloid opiates, serving major pharmaceutical companies across the Middle East.
Faran Shimi: Leading producer of high-quality APIs & alkaloid opiates, serving major pharmaceutical companies across the Middle East.
 Noramco is a partner to the pharmaceutical industry for controlled substance development & manufacturing.
Noramco is a partner to the pharmaceutical industry for controlled substance development & manufacturing.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 26528
Submission : 2012-09-14
Status : Active
Type : II

NDC Package Code : 51634-3248
Start Marketing Date : 2024-07-12
End Marketing Date : 2025-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 30173
Submission : 2016-12-30
Status : Active
Type : II


GDUFA
DMF Review : Reviewed
Rev. Date : 2014-12-08
Pay. Date : 2014-07-28
DMF Number : 22553
Submission : 2009-02-16
Status : Active
Type : II

NDC Package Code : 0406-0151
Start Marketing Date : 2013-12-19
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : Reviewed
Rev. Date : 2020-06-05
Pay. Date : 2020-03-05
DMF Number : 31803
Submission : 2020-03-02
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 8948
Submission : 1991-01-24
Status : Inactive
Type : II

NDC Package Code : 0406-0151
Start Marketing Date : 2013-12-19
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 15837
Submission : 2002-01-31
Status : Inactive
Type : II

NDC Package Code : 0406-0151
Start Marketing Date : 2013-12-19
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
We have 2 companies offering Naltrexone Methobromide
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
