

Synopsis
Synopsis
0
JDMF
0
VMF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
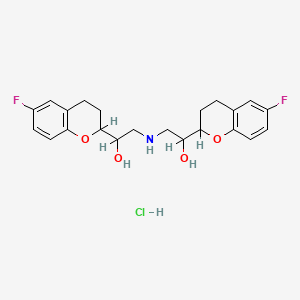

1. 67555, R
2. Alpha,alpha'-(iminobis(methylene))bis(6-fluoro-3,4-dihydro)-2h-1-benzopyran-2-methanol
3. Bystolic
4. Hydrochloride, Nebivolol
5. Lobivon
6. Nebilet
7. Nebivolol
8. R 67555
9. R-67555
10. R67555
11. Silostar
1. 169293-50-9
2. Nebivolol Hcl
3. 2,2'-azanediylbis(1-(6-fluorochroman-2-yl)ethanol) Hydrochloride
4. 152520-56-4
5. Rac Nebivolol Hydrochloride
6. R 065824 Hydrochloride
7. Nebilox
8. Rac-nebivolol Hcl
9. R-067555
10. Schembl514784
11. N-phenyliminodiaceticacid
12. Chembl1201731
13. Akos016006035
14. Ac-4233
15. Ac-5636
16. Ccg-214847
17. Ro67555
18. As-12100
19. Cs-0362720
20. Ft-0652243
21. Ft-0655833
22. 520n564
23. W-60374
24. J-010524
25. 1-(6-fluoro-3,4-dihydro-2h-chromen-2-yl)-2-[[2-(6-fluoro-3,4-dihydro-2h-chromen-2-yl)-2-hydroxyethyl]amino]ethanol;hydrochloride
26. 2h-1-benzopyran-2-methanol,a,a'-[iminobis(methylene)]bis[6-fluoro-3,4-dihydro-,hydrochloride(1:1)
27. Alpha,alpha'[iminobis(methylene)]bis[6-fluoro-3,4-dihydro-2h-1-benzopyran-2-methanol]hydrochloride
| Molecular Weight | 441.9 g/mol |
|---|---|
| Molecular Formula | C22H26ClF2NO4 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Exact Mass | 441.1518423 g/mol |
| Monoisotopic Mass | 441.1518423 g/mol |
| Topological Polar Surface Area | 71 Ų |
| Heavy Atom Count | 30 |
| Formal Charge | 0 |
| Complexity | 483 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 4 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Adrenergic beta-1 Receptor Agonists
Compounds that bind to and activate ADRENERGIC BETA-1 RECEPTORS. (See all compounds classified as Adrenergic beta-1 Receptor Agonists.)
Antihypertensive Agents
Drugs used in the treatment of acute or chronic vascular HYPERTENSION regardless of pharmacological mechanism. Among the antihypertensive agents are DIURETICS; (especially DIURETICS, THIAZIDE); ADRENERGIC BETA-ANTAGONISTS; ADRENERGIC ALPHA-ANTAGONISTS; ANGIOTENSIN-CONVERTING ENZYME INHIBITORS; CALCIUM CHANNEL BLOCKERS; GANGLIONIC BLOCKERS; and VASODILATOR AGENTS. (See all compounds classified as Antihypertensive Agents.)
Vasodilator Agents
Drugs used to cause dilation of the blood vessels. (See all compounds classified as Vasodilator Agents.)
GDUFA
DMF Review : Complete
Rev. Date : 2014-03-25
Pay. Date : 2013-09-11
DMF Number : 21152
Submission : 2007-12-08
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-10-13
Pay. Date : 2014-10-06
DMF Number : 22735
Submission : 2009-03-31
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2013-04-04
Pay. Date : 2012-12-05
DMF Number : 25296
Submission : 2011-09-12
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2015-01-05
Pay. Date : 2014-10-07
DMF Number : 28627
Submission : 2014-09-10
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 15417
Submission : 2001-05-07
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2016-10-21
Pay. Date : 2016-08-24
DMF Number : 29996
Submission : 2016-02-19
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-12-01
Pay. Date : 2014-11-20
DMF Number : 24916
Submission : 2011-05-04
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 26343
Submission : 2012-08-21
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 22906
Submission : 2009-06-29
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2016-04-13
Pay. Date : 2015-09-21
DMF Number : 29730
Submission : 2015-09-28
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R0-CEP 2022-095 - Rev 00
Status : Valid
Issue Date : 2022-08-31
Type : Chemical
Substance Number : 2775

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R0-CEP 2021-403 - Rev 00
Status : Valid
Issue Date : 2022-07-06
Type : Chemical
Substance Number : 2775

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : CEP 2023-063 - Rev 01
Status : Valid
Issue Date : 2024-07-30
Type : Chemical
Substance Number : 2775

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Nebivolol hydrochloride, Crystalline
Certificate Number : CEP 2022-193 - Rev 01
Status : Valid
Issue Date : 2024-03-13
Type : Chemical
Substance Number : 2775

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : CEP 2022-069 - Rev 00
Status : Valid
Issue Date : 2024-04-25
Type : Chemical
Substance Number : 2775

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : CEP 2023-122 - Rev 00
Status : Valid
Issue Date : 2024-07-19
Type : Chemical
Substance Number : 2775

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R0-CEP 2022-170 - Rev 00
Status : Valid
Issue Date : 2022-12-09
Type : Chemical
Substance Number : 2775

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : CEP 2022-140 - Rev 01
Status : Valid
Issue Date : 2023-12-20
Type : Chemical
Substance Number : 2775

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]  LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
About the Company : LGM Pharma is a global leader in sourcing hard-to-find APIs and intermediates for the pharmaceutical and biotech industries. LGM is also a full service CDMO providing formulation, ...
 Gonane has API manufacturing expertise in new-age Corticosteroids, Hormones and other pharma raw materials.
Gonane has API manufacturing expertise in new-age Corticosteroids, Hormones and other pharma raw materials.
About the Company : Gonane Pharma, is a contract pharmaceutical company located in Gujarat, India, specializing in the manufacturing and marketing of Corticosteroids, Hormones, Antivirals, and Oncolog...
About the Company : Jai Radhe Sales was founded in 1999 as an out-of-the-box distribution firm specializing in the global supply of high-quality pharmaceutical ingredients. The firm provides complete ...
 Octavius has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Octavius has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
About the Company : Octavius Pharma is a global leader in Directly Compressible Granules with over 40 years of experience in Formulation development, manufacturing and commercialization. It offers a w...
About the Company : We are a Pharmaceutical Company aligned across two business verticals viz. Products and Services. Our Products business comprises manufacture and sale of APIs and Intermediates to ...

About the Company : Established in the year 1988, we are a well named manufacturer, supplier, exporter and supplier of Active Pharmaceuticals Ingredients and its intermediates which includes Levocetir...

About the Company : Haorui, established in 1997, is a manufacturer and supplier of APIs, intermediates, veterinary and food additives. Meanwhile, it is assisting global pharmaceutical companies to get...

About the Company : Established in 2000, Shodhana Laboratories offers a wide range of APIs and intermediates and caters to the generic and custom requirements of some of the most prestigious pharma co...

About the Company : Suvan Lifesciences, an organization certified with GMP & ISO standards, stands out as a reputable producer and exporter of high-grade Active Pharmaceutical Ingredients. Our sophist...

About the Company : VASUDHA PHARMA CHEM LIMITED was incorporated, as a public limited company under the Companies Act, 1956 in 1994-95 at Hyderabad in the state of Telangana, India. The company is eng...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
