

Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
DRUG PRODUCT COMPOSITIONS
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
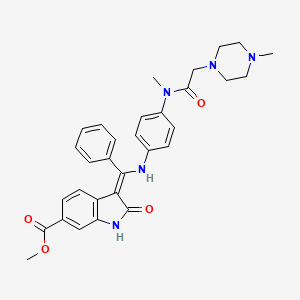

| Molecular Weight | 539.6 g/mol |
|---|---|
| Molecular Formula | C31H33N5O4 |
| XLogP3 | 3.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 8 |
| Exact Mass | 539.25325455 g/mol |
| Monoisotopic Mass | 539.25325455 g/mol |
| Topological Polar Surface Area | 94.2 A^2 |
| Heavy Atom Count | 40 |
| Formal Charge | 0 |
| Complexity | 947 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Ofev |
| Drug Label | OFEV capsules contain nintedanib, a kinase inhibitor [see Mechanism of Action (12.1)]. Nintedanib is presented as the ethanesulfonate salt (esylate), with the chemical name 1H-Indole-6-carboxylic acid, 2,3-dihydro-3-[[[4-[methyl[(4-methyl-1-piperazin... |
| Active Ingredient | Nintedanib |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 150mg; 100mg |
| Market Status | Prescription |
| Company | Boehringer Ingelheim |
| 2 of 2 | |
|---|---|
| Drug Name | Ofev |
| Drug Label | OFEV capsules contain nintedanib, a kinase inhibitor [see Mechanism of Action (12.1)]. Nintedanib is presented as the ethanesulfonate salt (esylate), with the chemical name 1H-Indole-6-carboxylic acid, 2,3-dihydro-3-[[[4-[methyl[(4-methyl-1-piperazin... |
| Active Ingredient | Nintedanib |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 150mg; 100mg |
| Market Status | Prescription |
| Company | Boehringer Ingelheim |
 Click Us!
Click Us! 
GDUFA
DMF Review : Reviewed
Rev. Date : 2019-03-26
Pay. Date : 2018-12-20
DMF Number : 33361
Submission : 2019-01-02
Status : Active
Type : II

Registrant Name : Masung LS Co., Ltd.
Registration Date : 2022-01-12
Registration Number : Wed163-23-ND
Manufacturer Name : Fermion Oy
Manufacturer Address : Lääketehtaantie 2, Oulu, FI-90660, Finland

| Available Reg Filing : ASMF |
 Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
 Midas Pharma provides expertise in developing and supplying APIs, finished products, and intermediates.
Midas Pharma provides expertise in developing and supplying APIs, finished products, and intermediates.

GDUFA
DMF Review : Reviewed
Rev. Date : 2018-04-27
Pay. Date : 2018-03-30
DMF Number : 32511
Submission : 2018-03-29
Status : Active
Type : II
 IKF/Pharmasynthese have been with fine chemicals market and APIs performance for more than 40 years.
IKF/Pharmasynthese have been with fine chemicals market and APIs performance for more than 40 years.
 Hubei Gedian Humanwell focuses on R&D, production, & sales of fertility regulation drugs & steroidal APIs.
Hubei Gedian Humanwell focuses on R&D, production, & sales of fertility regulation drugs & steroidal APIs.

GDUFA
DMF Review : Reviewed
Rev. Date : 2018-12-03
Pay. Date : 2018-09-20
DMF Number : 33152
Submission : 2018-09-28
Status : Active
Type : II

NDC Package Code : 82758-505
Start Marketing Date : 2022-05-30
End Marketing Date : 2025-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
 Jinan Tantu Chemicals offers customized R&D services & production of small molecule APIs & pharmaceutical intermediates.
Jinan Tantu Chemicals offers customized R&D services & production of small molecule APIs & pharmaceutical intermediates.
 Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.
Omgene: R&D-based biopharmaceutical company with GMP facilities, focused on innovation and high-quality, affordable medicines.

GDUFA
DMF Review : Reviewed
Rev. Date : 2023-05-08
Pay. Date : 2023-03-28
DMF Number : 37986
Submission : 2023-03-01
Status : Active
Type : II


GDUFA
DMF Review : Reviewed
Rev. Date : 2019-11-05
Pay. Date : 2019-10-10
DMF Number : 32152
Submission : 2017-12-28
Status : Active
Type : II
Date of Issue : 2021-01-08
Valid Till : 2023-11-23
Written Confirmation Number : WC-0310
Address of the Firm :

Registrant Name : Mpeak Korea Co., Ltd.
Registration Date : 2023-09-26
Registration Number : Wed812-21-ND
Manufacturer Name : Glenmark Life Sciences Ltd
Manufacturer Address : Plot No.Z-103/ I, DAHEJ SEZ Phase II, City : Dahej, District : Bharuch, Gujarat State, India

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
