
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
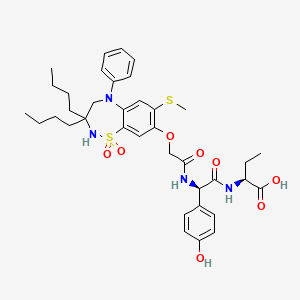

1. (2s)-2-(((2r)-2-((((3,3-dibutyl-7-(methylsulfanyl)-1,1-dioxido-5-phenyl-2,3,4,5-tetrahydro-1,2,5-benzothiadiazepin-8-yl)oxy)acetyl)amino)-2-(4-hydroxyphenyl)acetyl)amino)butanoic Acid
2. Bylvay
1. 501692-44-0
2. Azd8294
3. A4250
4. Ar-h064974
5. Odevixibat [usan]
6. A-4250
7. 2w150k0uuc
8. Azd-8294
9. Odevixibat (usan)
10. (2s)-2-[[(2r)-2-[[2-[(3,3-dibutyl-7-methylsulfanyl-1,1-dioxo-5-phenyl-2,4-dihydro-1lambda6,2,5-benzothiadiazepin-8-yl)oxy]acetyl]amino]-2-(4-hydroxyphenyl)acetyl]amino]butanoic Acid
11. (s)-2-((r)-2-(2-((3,3-dibutyl-7-(methylthio)-1,1-dioxido-5-phenyl-2,3,4,5-tetrahydrobenzo[f][1,2,5]thiadiazepin-8-yl)oxy)acetamido)-2-(4-hydroxyphenyl)acetamido)butanoic Acid
12. Butanoic Acid, 2-(((2r)-((((3,3-dibutyl-2,3,4,5-tetrahydro-7-(methylthio)-1,1-dioxido-5-phenyl-1,2,5-benzothiadiazepin-8-yl)oxy)acetyl)amino)(4-hydroxyphenyl)acetyl)amino)-, (2s)-
13. Butanoic Acid, 2-(((2r)-2-((2-((3,3-dibutyl-2,3,4,5-tetrahydro-7-(methylthio)-1,1-dioxido-5-phenyl-1,2,5-benzothiadiazepin-8-yl)oxy)acetyl)amino)-2-(4-hydroxyphenyl)acetyl)amino)-, (2s)-
14. Odevixibat [inn]
15. Odevixibat [who-dd]
16. Unii-2w150k0uuc
17. Schembl946468
18. Chembl4297588
19. Odevixibat [orange Book]
20. Bdbm77040
21. Gtpl11194
22. Dtxsid601336860
23. Ex-a5384
24. Example 5 [us9694018b1]
25. Who 10706
26. At28924
27. Bo181253
28. Hy-109120
29. Unk-d-ntyr-abu-oh (iupac Condensed Name)
30. Cs-0078340
31. D11716
32. Us9694018, 5
33. (2s)-2-(((2r)-2-((((3,3-dibutyl-7-(methylthio)-1,1-dioxido-5-phenyl-2,3,4,5-tetrahydro- 1,2,5-benzothiadiazepin-8-yl)oxy)acetyl)amino)-2-(4-hydroxyphenyl)acetyl)amino)butanoic Acid
| Molecular Weight | 740.9 g/mol |
|---|---|
| Molecular Formula | C37H48N4O8S2 |
| XLogP3 | 6.8 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 17 |
| Exact Mass | 740.29135685 g/mol |
| Monoisotopic Mass | 740.29135685 g/mol |
| Topological Polar Surface Area | 208 Ų |
| Heavy Atom Count | 51 |
| Formal Charge | 0 |
| Complexity | 1230 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Odevixibat is indicated for the treatment of pruritus in patients older than 3 months, with progressive familiar intrahepatic cholestasis (PFIC). It may not be effective in PFIC type 2 with ABCB11 variants. These patients lack a functional bile salt export pump.
Treatment of biliary atresia
Treatment of Progressive Familial Intrahepatic Cholestasis
Bylvay is indicated for the treatment of progressive familial intrahepatic cholestasis (PFIC) in patients aged 6 months or older (see sections 4. 4 and 5. 1).
Treatment of Alagille syndrome
Odevixibat, or A4250, is an ileal sodium/bile acid cotransporter inhibitor indicated for the treatment of pruritus in patients older than 3 months, with progressive familiar intrahepatic cholestasis (PFIC). It has a moderate duration of action as it is given once daily. Odevixibat has a wide therapeutic index as patients were given single doses up to 10 mg while the maximum therapeutic dose is 6 mg daily. Patients should be counselled regarding the risks of elevated liver function tests, diarrhea, and fat soluble vitamin defiencies.
A05AX
A - Alimentary tract and metabolism
A05 - Bile and liver therapy
A05A - Bile therapy
A05AX - Other drugs for bile therapy
A05AX05 - Odevixibat
Absorption
A 7.2 mg single oral dose of odevixibat in adults reaches a Cmax of 0.47 ng/mL, with an AUC0-24h of 2.19 h\*ng/mL. The majority of adult and pediatric patients, given a therapeutic dose, do not have detectable plasma concentrations of odevixibat.
Route of Elimination
Odevixibat is 82.9% recovered in the feces and <0.002% recovered in the urine. The dose recovered in the feces is 97% unchanged parent compound.
Volume of Distribution
The majority of adult and pediatric patients, given a therapeutic dose, do not have detectable plasma concentrations of odevixibat. Therefore, a volume of distribution has not been calculated.
Clearance
The majority of adult and pediatric patients, given a therapeutic dose, do not have detectable plasma concentrations of odevixibat. Therefore, the clearance has not been calculated.
Odevixibat is largely unmetabolized, however a small amount is metabolized _in vitro_ by mono-hydroxylation. The exact structure of the metabolite has not been characterized as a primary endpoint of the clinical trial was to characterize the structure of metabolites accounting for >10% of the dose in plasma, urine, or feces. No metabolites have been identified at such a high concentration.
A 7.2 mg oral dose of odevixibat has a mean half life of 2.36 hours in adults.
Progressive familiar intrahepatic cholestasis (PFIC) is a group of autosomal recessive disorders leading to cholestasis, fibrosis, and eventually a need for liver transplantation. Patients with PFIC require liver transplants or develop hepatocellular carcinomas in their first few years of life. Many of these patients experience severe pruritus. The exact mechanism of pruritus is PFIC is not known, but lower concentrations of bile acids have been shown to reduce pruritus. Patients with certain forms of PFIC type 2, associated with a non-functional or absent bile salt export pump, are not expected to benefit from odevixibat treatment. The ileal sodium/bile acid cotransporter is a transport glycoprotein responsible for reabsorption of 95% of bile acids in the distal ileum. Odevixibat is a reversible inhibitor of the ileal sodium/bile acid contransporter. Patients taking odevixibat for a week experienced a 56% reduction in bile acid area under the curve with a 3 mg once daily dose. A 1.5 mg daily dose lead to a 43% reduction in bile acid area under the curve. The decreased reabsorption of bile acids, leads to reduced stimulation of FXR, which reduces expression of FGF19, reducing binding of FGF19 to FGF4R, decreasing inhibition of bile acid synthesis. Further synthesis of bile acids that will not be reabsorbed in the intestine contributes to lowering low density lipoprotein levels.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
88
PharmaCompass offers a list of Odevixibat API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Odevixibat manufacturer or Odevixibat supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Odevixibat manufacturer or Odevixibat supplier.
PharmaCompass also assists you with knowing the Odevixibat API Price utilized in the formulation of products. Odevixibat API Price is not always fixed or binding as the Odevixibat Price is obtained through a variety of data sources. The Odevixibat Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Odevixibat manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Odevixibat, including repackagers and relabelers. The FDA regulates Odevixibat manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Odevixibat API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Odevixibat manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Odevixibat supplier is an individual or a company that provides Odevixibat active pharmaceutical ingredient (API) or Odevixibat finished formulations upon request. The Odevixibat suppliers may include Odevixibat API manufacturers, exporters, distributors and traders.
click here to find a list of Odevixibat suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Odevixibat written confirmation (Odevixibat WC) is an official document issued by a regulatory agency to a Odevixibat manufacturer, verifying that the manufacturing facility of a Odevixibat active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Odevixibat APIs or Odevixibat finished pharmaceutical products to another nation, regulatory agencies frequently require a Odevixibat WC (written confirmation) as part of the regulatory process.
click here to find a list of Odevixibat suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Odevixibat as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Odevixibat API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Odevixibat as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Odevixibat and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Odevixibat NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Odevixibat suppliers with NDC on PharmaCompass.
Odevixibat Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Odevixibat GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Odevixibat GMP manufacturer or Odevixibat GMP API supplier for your needs.
A Odevixibat CoA (Certificate of Analysis) is a formal document that attests to Odevixibat's compliance with Odevixibat specifications and serves as a tool for batch-level quality control.
Odevixibat CoA mostly includes findings from lab analyses of a specific batch. For each Odevixibat CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Odevixibat may be tested according to a variety of international standards, such as European Pharmacopoeia (Odevixibat EP), Odevixibat JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Odevixibat USP).

