

Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
DRUG PRODUCT COMPOSITIONS
0
EDQM
0
USP
0
JP
0
Others
0
Weekly News Recap #Phispers

1. Azd-9291
2. Azd-9291 Mesylate
3. Azd9291
4. Azd9291 Mesylate
5. Mereletinib
6. Mereletinib Mesilate
7. Mereletinib Mesylate
8. N-(2-((2-(dimethylamino)ethyl)methylamino)-4-methoxy-5-((4-(1-methyl-1h-indol-3-yl)-2-pyrimidinyl)amino)phenyl)-2-propenamide
9. N-(2-((2-(dimethylamino)ethyl)methylamino)-4-methoxy-5-((4-(1-methyl-1h-indol-3-yl)-2-pyrimidinyl)amino)phenyl)-2-propenamide Methanesulfonate (1:1)
10. Osimertinib
11. Osimertinib Mesilate
12. Tagrisso
1. 1421373-66-1
2. Azd-9291 Mesylate
3. Azd9291 Mesylate
4. Azd-9291 (mesylate)
5. Mereletinib Mesylate
6. Tagrisso
7. Osimertinib Mesilate
8. Mereletinib Mesilate
9. Osimertinib Mesylate [usan]
10. Rdl94r2a16
11. N-[2-[2-(dimethylamino)ethyl-methylamino]-4-methoxy-5-[[4-(1-methylindol-3-yl)pyrimidin-2-yl]amino]phenyl]prop-2-enamide;methanesulfonic Acid
12. N-(2-((2-(dimethylamino)ethyl)(methyl)amino)-4-methoxy-5-((4-(1-methyl-1h-indol-3-yl)pyrimidin-2-yl)amino)phenyl)acrylamide Methanesulfonate
13. Osimertinib Mesilate (jan)
14. Osimertinib Mesylate (usan)
15. 2-propenamide, N-(2-((2-(dimethylamino)ethyl)methylamino)-4-methoxy-5-((4-(1-methyl-1h-indol-3-yl)-2-pyrimidinyl)amino)phenyl)-, Methanesulfonate (1:1)
16. Osimertinib Mesilate [jan]
17. 2-propenamide, N-[2-[[2-(dimethylamino)ethyl]methylamino]-4-methoxy-5-[[4-(1-methyl-1h-indol-3-yl)-2-pyrimidinyl]amino]phenyl]-, Compd. With Methanesulfonate (1:2)
18. Unii-rdl94r2a16
19. Tagrisso (tn)
20. Osimertinib Monomesylate
21. Azd 9291 Mesylate
22. Osimertinib Methanesulfonate
23. Amy226
24. Chembl3545063
25. Schembl14661152
26. Chebi:90948
27. Osimertinib Mesylate [mi]
28. Dtxsid101027822
29. Bcp09934
30. Ex-a1577
31. Hy-15772a
32. Mfcd28137994
33. Osimertinib Mesilate [who-dd]
34. Akos026673944
35. Ds-9913
36. Sb22953
37. Ac-29022
38. Da-35303
39. Osimertinib Mesylate [orange Book]
40. Azd-9291 Mesylate (osimertinibmereletinib)
41. Ft-0699962
42. S5078
43. D10766
44. Q27162942
45. Azd9291 Ms Salt, Osimertinib Ms Salt; Mereletinib Ms Salt
46. N-(2-{[2-(dimethylamino)ethyl](methyl)amino}-4-methoxy-5-{[4-(1-methyl-1h-indol-3-yl)pyrimidin-2-yl]amino}phenyl)prop-2-enamide Methanesulfonate
47. N-[2-[[2-(dimethylamino)ethyl]methylamino]-4-methoxy-5-[[4-(1-methyl-1h-indol-3-yl)-2-pyrimidinyl]amino]phenyl]-2-propenamide Methanesulfonate
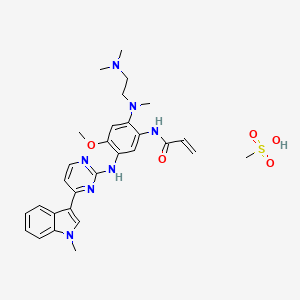
1. Osimertinib
| Molecular Weight | 595.7 g/mol |
|---|---|
| Molecular Formula | C29H37N7O5S |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 10 |
| Exact Mass | 595.25768848 g/mol |
| Monoisotopic Mass | 595.25768848 g/mol |
| Topological Polar Surface Area | 150 Ų |
| Heavy Atom Count | 42 |
| Formal Charge | 0 |
| Complexity | 845 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
TAGRISSO as monotherapy is indicated for:
-the adjuvant treatment after complete tumour resection in adult patients with stage IB-IIIA non-small cell lung cancer (NSCLC) whose tumours have epidermal growth factor receptor (EGFR) exon 19 deletions or exon 21 (L858R) substitution mutations
- the first-line treatment of adult patients NSCLC with activating EGFR mutations.
- the treatment of adult patients with locally advanced or metastatic EGFR T790M mutation-positive NSCLC.
TAGRISSO as monotherapy is indicated for:
- the adjuvant treatment after complete tumour resection in adult patients with stage IB-IIIA non-small cell lung cancer (NSCLC) whose tumours have epidermal growth factor receptor (EGFR) exon 19 deletions or exon 21 (L858R) substitution mutations.
- the first-line treatment of adult patients with locally advanced or metastatic NSCLC with activating EGFR mutations.
- the treatment of adult patients with locally advanced or metastatic EGFR T790M mutation-positive NSCLC.
Antineoplastic Agents
Substances that inhibit or prevent the proliferation of NEOPLASMS. (See all compounds classified as Antineoplastic Agents.)
Protein Kinase Inhibitors
Agents that inhibit PROTEIN KINASES. (See all compounds classified as Protein Kinase Inhibitors.)
L01XE

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Tagrisso (osimertinib) is a third-generation, irreversible EGFR-TKI, which is indicated for treatment of patients with unresectable, Stage III EGFR-mutated non small cell lung cancer.
Lead Product(s): Osimertinib Mesylate
Therapeutic Area: Oncology Brand Name: Tagrisso
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable September 26, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Osimertinib Mesylate
Therapeutic Area : Oncology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
TAGRISSO® Approved For Unresectable Stage III EGFR-Mutated Lung Cancer
Details : Tagrisso (osimertinib) is a third-generation, irreversible EGFR-TKI, which is indicated for treatment of patients with unresectable, Stage III EGFR-mutated non small cell lung cancer.
Brand Name : Tagrisso
Molecule Type : Small molecule
Upfront Cash : Not Applicable
September 26, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Tagrisso (osimertinib) is a third-generation, irreversible EGFR-TKI, which is being evaluated for patients with unresectable, Stage III EGFR-mutated non small cell lung cancer.
Lead Product(s): Osimertinib Mesylate
Therapeutic Area: Oncology Brand Name: Tagrisso
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable July 06, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Osimertinib Mesylate
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
EU Approves AstraZeneca’s Tagrisso with Chemotherapy for EGFR-Mutated Lung Cancer
Details : Tagrisso (osimertinib) is a third-generation, irreversible EGFR-TKI, which is being evaluated for patients with unresectable, Stage III EGFR-mutated non small cell lung cancer.
Brand Name : Tagrisso
Molecule Type : Small molecule
Upfront Cash : Not Applicable
July 06, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Tagrisso (osimertinib) is a third-generation, irreversible EGFR-TKI, which is being evaluated for patients with unresectable, Stage III EGFR-mutated non small cell lung cancer.
Lead Product(s): Osimertinib Mesylate
Therapeutic Area: Oncology Brand Name: Tagrisso
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable June 10, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Osimertinib Mesylate
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Tagrisso Granted Priority Review for Unresectable Stage III Lung Cancer
Details : Tagrisso (osimertinib) is a third-generation, irreversible EGFR-TKI, which is being evaluated for patients with unresectable, Stage III EGFR-mutated non small cell lung cancer.
Brand Name : Tagrisso
Molecule Type : Small molecule
Upfront Cash : Not Applicable
June 10, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Tagrisso (osimertinib) is a third-generation, irreversible EGFR-TKI, which is being evaluated for patients with unresectable, Stage III EGFR-mutated non small cell lung cancer.
Lead Product(s): Osimertinib Mesylate
Therapeutic Area: Oncology Brand Name: Tagrisso
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable June 02, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Osimertinib Mesylate
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
TAGRISSO® Reduces Disease Progression Risk by 84% in Stage III EGFR-Mutated Lung Cancer
Details : Tagrisso (osimertinib) is a third-generation, irreversible EGFR-TKI, which is being evaluated for patients with unresectable, Stage III EGFR-mutated non small cell lung cancer.
Brand Name : Tagrisso
Molecule Type : Small molecule
Upfront Cash : Not Applicable
June 02, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Tagrisso (osimertinib mesylate) is a third-generation, irreversible EGFR-TKI, small molecule drug candidate which is being evaluated for the treatment of EGFR-mutated advanced lung cancer.
Lead Product(s): Osimertinib Mesylate,Pemetrexed,Carboplatin
Therapeutic Area: Oncology Brand Name: Tagrisso
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable March 21, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Osimertinib Mesylate,Pemetrexed,Carboplatin
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Details : Tagrisso (osimertinib mesylate) is a third-generation, irreversible EGFR-TKI, small molecule drug candidate which is being evaluated for the treatment of EGFR-mutated advanced lung cancer.
Brand Name : Tagrisso
Molecule Type : Small molecule
Upfront Cash : Not Applicable
March 21, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Tagrisso (osimertinib) is a third-generation, irreversible EGFR-TKI, which is being evaluated for patients with unresectable, Stage III EGFR-mutated non small cell lung cancer.
Lead Product(s): Osimertinib Mesylate
Therapeutic Area: Oncology Brand Name: Tagrisso
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable February 19, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Osimertinib Mesylate
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Details : Tagrisso (osimertinib) is a third-generation, irreversible EGFR-TKI, which is being evaluated for patients with unresectable, Stage III EGFR-mutated non small cell lung cancer.
Brand Name : Tagrisso
Molecule Type : Small molecule
Upfront Cash : Not Applicable
February 19, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Tagrisso (osimertinib) is a third-generation, irreversible EGFR-TKI which is approved in combination with chemotherapy for the treatment of EGFR-mutated advanced non-small cell lung cancer.
Lead Product(s): Osimertinib Mesylate,Pemetrexed,Carboplatin
Therapeutic Area: Oncology Brand Name: Tagrisso
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable February 16, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Osimertinib Mesylate,Pemetrexed,Carboplatin
Therapeutic Area : Oncology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
TAGRISSO® (osimertinib) with the Addition of Chemotherapy Approved in the US for Patients with EG...
Details : Tagrisso (osimertinib) is a third-generation, irreversible EGFR-TKI which is approved in combination with chemotherapy for the treatment of EGFR-mutated advanced non-small cell lung cancer.
Brand Name : Tagrisso
Molecule Type : Small molecule
Upfront Cash : Not Applicable
February 16, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
ENV105 (carotuximab) is a novel ENG inhibitor monoclonal antibody. It is currently being evaluated in phase 1 clinical trials in combination with osimertinib for the treatment of non-small cell lung cancer.
Lead Product(s): Carotuximab,Osimertinib Mesylate
Therapeutic Area: Oncology Brand Name: ENV105
Study Phase: Phase IProduct Type: Large molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable January 16, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Carotuximab,Osimertinib Mesylate
Therapeutic Area : Oncology
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Details : ENV105 (carotuximab) is a novel ENG inhibitor monoclonal antibody. It is currently being evaluated in phase 1 clinical trials in combination with osimertinib for the treatment of non-small cell lung cancer.
Brand Name : ENV105
Molecule Type : Large molecule
Upfront Cash : Not Applicable
January 16, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Tagrisso (osimertinib) is a third-generation, irreversible EGFR-TKI which is being evaluated in combination with chemotherapy in phase 3 clinical trials for the treatment of EGFR-mutated advanced lung cancer and brain metastases.
Lead Product(s): Osimertinib Mesylate,Pemetrexed,Cisplatin
Therapeutic Area: Oncology Brand Name: Tagrisso
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable October 21, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Osimertinib Mesylate,Pemetrexed,Cisplatin
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Details : Tagrisso (osimertinib) is a third-generation, irreversible EGFR-TKI which is being evaluated in combination with chemotherapy in phase 3 clinical trials for the treatment of EGFR-mutated advanced lung cancer and brain metastases.
Brand Name : Tagrisso
Molecule Type : Small molecule
Upfront Cash : Not Applicable
October 21, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Tagrisso (osimertinib) is kinase inhibitor of the epidermal growth factor receptor (EGFR), which is investigated in combination with chemotherapy for patients with EGFR-mutated advanced lung cancer.
Lead Product(s): Osimertinib Mesylate,Pemetrexed,Carboplatin
Therapeutic Area: Oncology Brand Name: Tagrisso
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable October 16, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Osimertinib Mesylate,Pemetrexed,Carboplatin
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Details : Tagrisso (osimertinib) is kinase inhibitor of the epidermal growth factor receptor (EGFR), which is investigated in combination with chemotherapy for patients with EGFR-mutated advanced lung cancer.
Brand Name : Tagrisso
Molecule Type : Small molecule
Upfront Cash : Not Applicable
October 16, 2023

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : TAGRISSO
Dosage Form : FILM COATED PILL
Dosage Strength : 80 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : TAGRISSO
Dosage Form : FILM COATED PILL
Dosage Strength : 40 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Norway
Brand Name : Tagrisso
Dosage Form : Antic-calc Tablet, Film Coated
Dosage Strength : 40 mg
Packaging : Blister
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Norway

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Norway
Brand Name : Tagrisso
Dosage Form : Antic-calc Tablet, Film Coated
Dosage Strength : 80 mg
Packaging : Blister
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Norway

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Tagrisso
Dosage Form : Tabl
Dosage Strength : 40mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Tagrisso
Dosage Form : Tabl
Dosage Strength : 80mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Prescription
Registration Country : Canada
OSIMERTINIB (OSIMERTINIB MESYLATE)
Brand Name : TAGRISSO
Dosage Form : TABLET
Dosage Strength : 40MG
Packaging : 30
Approval Date :
Application Number : 2456214
Regulatory Info : Prescription
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Prescription
Registration Country : Canada
OSIMERTINIB (OSIMERTINIB MESYLATE)
Brand Name : TAGRISSO
Dosage Form : TABLET
Dosage Strength : 80MG
Packaging : 30
Approval Date :
Application Number : 2456222
Regulatory Info : Prescription
Registration Country : Canada

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Global Sales Information

Market Place

Reply
04 Mar 2021
Reply
23 Oct 2018
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Patents & EXCLUSIVITIES
ABOUT THIS PAGE
We have 5 companies offering Osimertinib Mesylate
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
