
Synopsis
Synopsis
0
VMF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
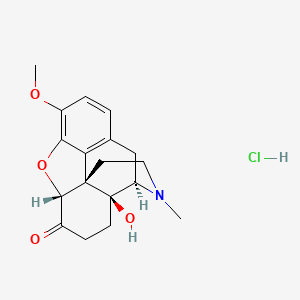

1. Dihydrohydroxycodeinone
2. Dihydrone
3. Dinarkon
4. Eucodal
5. Oxiconum
6. Oxycodeinon
7. Oxycodone
8. Oxycone
9. Oxycontin
10. Pancodine
11. Theocodin
1. 124-90-3
2. Oxycodone Hcl
3. Oxecta
4. Oxycontin
5. Endocodone
6. Roxicodone
7. Oxycodone Hydrochloride Cii
8. Chebi:7859
9. 4,5alpha-epoxy-14-hydroxy-3-methoxy-17-methylmorphinan-6-one Hydrochloride
10. (4r,4as,7ar,12bs)-4a-hydroxy-9-methoxy-3-methyl-2,4,5,6,7a,13-hexahydro-1h-4,12-methanobenzofuro[3,2-e]isoquinolin-7-one;hydrochloride
11. (4r,4as,7ar,12bs)-4a-hydroxy-9-methoxy-3-methyl-2,4,5,6,7a,13-hexahydro-1h-4,12-methanobenzofuro[3,2-e]isoquinolin-7-one;hydron;chloride
12. Roxicodone (tn)
13. Oxycontin (tn)
14. (4r,4as,7ar,12bs)-4a-hydroxy-9-methoxy-3-methyl-2,4,5,6,7a,13-hexahydro-1h-4,12-methanobenzofuro[3,2-e]isoquinoline-7-one;hydrochloride
15. Oxecta (tn)
16. Roxybond
17. Oxycodone Monohydrochloride
18. Schembl30095
19. Oxycodone Hydrochloride (usp)
20. Chembl1200890
21. Dtxsid80924674
22. Oxycodone Hydrochloride [mi]
23. Oxycodone Hydrochloride [usan]
24. Akos024457896
25. Oxycodone Hydrochloride [mart.]
26. Oxycodone Hydrochloride [vandf]
27. Anhydrous Oxycodone Hydrochloride
28. Oxycodone Hydrochloride [who-dd]
29. Oxycodone Hydrochloride, Analytical Standard
30. Oxycodone Hydrochloride [orange Book]
31. Oxycodone Hydrochloride Cii [usp-rs]
32. Oxycodone Hydrochloride [ep Monograph]
33. Tylox Component Oxycodone Hydrochloride
34. C08026
35. Codoxy Component Oxycodone Hydrochloride
36. D00847
37. Oxycet Component Oxycodone Hydrochloride
38. Oxycodone Hydrochloride [usp Monograph]
39. Roxicet Component Oxycodone Hydrochloride
40. Roxilox Component Oxycodone Hydrochloride
41. Troxyca Component Oxycodone Hydrochloride
42. Combunox Component Oxycodone Hydrochloride
43. Oxycodone Hydrochloride Component Of Tylox
44. Percocet Component Oxycodone Hydrochloride
45. Percodan Component Oxycodone Hydrochloride
46. Roxiprin Component Oxycodone Hydrochloride
47. Targiniq Component Oxycodone Hydrochloride
48. Xartemis Component Oxycodone Hydrochloride
49. Oxycodone Hydrochloride Component Of Codoxy
50. Oxycodone Hydrochloride Component Of Oxycet
51. Oxycodone Hydrochloride Component Of Roxicet
52. Oxycodone Hydrochloride Component Of Roxilox
53. Oxycodone Hydrochloride Component Of Troxyca
54. Oxycodone Hydrochloride Component Of Combunox
55. Oxycodone Hydrochloride Component Of Percocet
56. Oxycodone Hydrochloride Component Of Percodan
57. Oxycodone Hydrochloride Component Of Roxiprin
58. Oxycodone Hydrochloride Component Of Targiniq
59. Oxycodone Hydrochloride Component Of Xartemis
60. Percodan-demi Component Oxycodone Hydrochloride
61. Q27107601
62. Oxycodone Hydrochloride Component Of Percodan-demi
63. 14-hydroxy-3-methoxy-17-methyl-4,5alpha-epoxymorphinan-6-one Hydrochloride
64. Oxycodone Hydrochloride, European Pharmacopoeia (ep) Reference Standard
65. Oxycodone Hydrochloride, United States Pharmacopeia (usp) Reference Standard
66. (5?)-4,5-epoxy-14-hydroxy-3-methoxy-17-methylmorphinan-6-one Hydrochloride
67. (5alpha,17s)-14-hydroxy-3-methoxy-17-methyl-6-oxo-4,5-epoxymorphinan-17-ium Chloride
68. 4,5.alpha.-epoxy-14-hydroxy-3-methoxy-17-methylmorphinan-6-one Hydrochloride
69. Morphinan-6-one, 4,5-epoxy-14-hydroxy-3-methoxy-17-methyl-, Hydrochloride, (5.alpha.)-
70. Oxycodone Hydrochloride Solution, 1.0 Mg/ml In Methanol, Analytical Standard, For Drug Analysis
| Molecular Weight | 351.8 g/mol |
|---|---|
| Molecular Formula | C18H22ClNO4 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 1 |
| Exact Mass | 351.1237359 g/mol |
| Monoisotopic Mass | 351.1237359 g/mol |
| Topological Polar Surface Area | 59 Ų |
| Heavy Atom Count | 24 |
| Formal Charge | 0 |
| Complexity | 553 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 4 | |
|---|---|
| Drug Name | Oxecta |
| Drug Label | OXECTA (oxycodone HCl, USP) tablets are an immediate-release opioid analgesic intended for oral administration only. OXECTA contains oxycodone HCl, USP as the active analgesic ingredient. The tablets are round, convex, white and debossed with the str... |
| Active Ingredient | Oxycodone hydrochloride |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 7.5mg; 5mg |
| Market Status | Prescription |
| Company | Acura Pharms |
| 2 of 4 | |
|---|---|
| Drug Name | Oxycodone hydrochloride |
| Drug Label | DESCRIPTIONOxyContin (oxycodone hydrochloride controlled-release) Tablets are an opioid analgesic supplied in 10 mg, 15 mg, 20 mg, 30 mg, 40 mg, 60 mg, 80 mg, and 160 mg tablet strengths for oral administration... |
| Active Ingredient | Oxycodone hydrochloride |
| Dosage Form | Tablet, extended release; Tablet; Capsule; Solution |
| Route | oral; Oral |
| Strength | 30mg; 100mg/5ml; 15mg; 5mg; 10mg; 5mg/5ml; 80mg; 40mg; 20mg |
| Market Status | Tentative Approval; Prescription |
| Company | Corepharma; Vintage Pharms; Amneal Pharms; Impax Pharms; Aurolife Pharma; Avanthi; Teva; Endo Pharms; Sun Pharm Inds; Coastal Pharms; Rhodes Pharms; Mallinckrodt; Roxane; Lannett Holdings; Nesher Pharms; Vistapharm; Actavis Elizabeth; Lehigh Valley; Alvog |
| 3 of 4 | |
|---|---|
| Drug Name | Oxecta |
| Drug Label | OXECTA (oxycodone HCl, USP) tablets are an immediate-release opioid analgesic intended for oral administration only. OXECTA contains oxycodone HCl, USP as the active analgesic ingredient. The tablets are round, convex, white and debossed with the str... |
| Active Ingredient | Oxycodone hydrochloride |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 7.5mg; 5mg |
| Market Status | Prescription |
| Company | Acura Pharms |
| 4 of 4 | |
|---|---|
| Drug Name | Oxycodone hydrochloride |
| Drug Label | DESCRIPTIONOxyContin (oxycodone hydrochloride controlled-release) Tablets are an opioid analgesic supplied in 10 mg, 15 mg, 20 mg, 30 mg, 40 mg, 60 mg, 80 mg, and 160 mg tablet strengths for oral administration... |
| Active Ingredient | Oxycodone hydrochloride |
| Dosage Form | Tablet, extended release; Tablet; Capsule; Solution |
| Route | oral; Oral |
| Strength | 30mg; 100mg/5ml; 15mg; 5mg; 10mg; 5mg/5ml; 80mg; 40mg; 20mg |
| Market Status | Tentative Approval; Prescription |
| Company | Corepharma; Vintage Pharms; Amneal Pharms; Impax Pharms; Aurolife Pharma; Avanthi; Teva; Endo Pharms; Sun Pharm Inds; Coastal Pharms; Rhodes Pharms; Mallinckrodt; Roxane; Lannett Holdings; Nesher Pharms; Vistapharm; Actavis Elizabeth; Lehigh Valley; Alvog |
Analgesics, Opioid
Compounds with activity like OPIATE ALKALOIDS, acting at OPIOID RECEPTORS. Properties include induction of ANALGESIA or NARCOSIS. (See all compounds classified as Analgesics, Opioid.)
Narcotics
Agents that induce NARCOSIS. Narcotics include agents that cause somnolence or induced sleep (STUPOR); natural or synthetic derivatives of OPIUM or MORPHINE or any substance that has such effects. They are potent inducers of ANALGESIA and OPIOID-RELATED DISORDERS. (See all compounds classified as Narcotics.)
 Click Us!
Click Us! 
GDUFA
DMF Review : Reviewed
Rev. Date : 2014-06-21
Pay. Date : 2014-06-13
DMF Number : 18472
Submission : 2005-06-30
Status : Active
Type : II

Certificate Number : CEP 2005-036 - Rev 04
Issue Date : 2024-04-10
Type : Chemical
Substance Number : 1793
Status : Valid

NDC Package Code : 49812-0150
Start Marketing Date : 2010-06-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Samung Industry Co., Ltd.
Registration Date : 2019-12-23
Registration Number : 20191223-209-J-267
Manufacturer Name : Macfarlan Smith Ltd.
Manufacturer Address : 10 Wheatfield Road, Edinburgh, EH11 2QA, United Kingdom

| Available Reg Filing : ROW, CN |

Date of Issue : 2018-02-19
Valid Till : 2019-02-19
Written Confirmation Number : 665/149786
Address of the Firm :
 Noramco is a partner to the pharmaceutical industry for controlled substance development & manufacturing.
Noramco is a partner to the pharmaceutical industry for controlled substance development & manufacturing.

GDUFA
DMF Review : Reviewed
Rev. Date : 2013-04-16
Pay. Date : 2013-04-05
DMF Number : 16399
Submission : 2003-02-03
Status : Active
Type : II

Certificate Number : R1-CEP 2011-284 - Rev 02
Issue Date : 2021-02-24
Type : Chemical
Substance Number : 1793
Status : Valid

NDC Package Code : 51634-0003
Start Marketing Date : 2008-02-21
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Samung Industry Co., Ltd.
Registration Date : 2024-01-26
Registration Number : 20240126-209-J-1599
Manufacturer Name : Noramco, LLC
Manufacturer Address : 500 Swedes Landing Road Wilmington, Delaware 19801
 Siegfried – A global CDMO delivering integrated pharmaceutical development and manufacturing solutions.
Siegfried – A global CDMO delivering integrated pharmaceutical development and manufacturing solutions.

Certificate Number : R1-CEP 2006-001 - Rev 01
Issue Date : 2018-11-21
Type : Chemical
Substance Number : 1793
Status : Valid

NDC Package Code : 0792-2936
Start Marketing Date : 2010-02-04
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 32506
Submission : 2018-03-16
Status : Active
Type : II

Registrant Name : Nosa Chemical Co., Ltd.
Registration Date : 2019-12-23
Registration Number : 20191223-209-J-267(2)
Manufacturer Name : Macfarlan Smith Ltd.
Manufacturer Address : 10 Wheatfield Road, Edinburgh, EH11 2QA, United Kingdom

| Available Reg Filing : ROW, CN |


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 34028
Submission : 2019-10-14
Status : Active
Type : II

Registrant Name : Baeksu Pharmaceutical Co., Ltd.
Registration Date : 2019-12-23
Registration Number : 20191223-209-J-267(A)
Manufacturer Name : Macfarlan Smith Limited
Manufacturer Address : 10 Wheatfield Road, Edinburgh, EH11 2QA, United Kingdom

| Available Reg Filing : ROW, CN |

 Noramco is a partner to the pharmaceutical industry for controlled substance development & manufacturing.
Noramco is a partner to the pharmaceutical industry for controlled substance development & manufacturing.

GDUFA
DMF Review : Reviewed
Rev. Date : 2012-12-27
Pay. Date : 2012-11-23
DMF Number : 20975
Submission : 2007-10-26
Status : Active
Type : II

Certificate Number : R1-CEP 2009-058 - Rev 04
Issue Date : 2022-05-12
Type : Chemical
Substance Number : 1793
Status : Valid

NDC Package Code : 51634-0002
Start Marketing Date : 2000-01-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Mundipharma Korea Co., Ltd.
Registration Date : 2022-01-13
Registration Number : 20220113-209-J-1209
Manufacturer Name : Noramco Coventry LLC
Manufacturer Address : 498 Washington Street Coventry, Rhode Island 02816 USA
 Noramco is a partner to the pharmaceutical industry for controlled substance development & manufacturing.
Noramco is a partner to the pharmaceutical industry for controlled substance development & manufacturing.

GDUFA
DMF Review : Reviewed
Rev. Date : 2013-02-15
Pay. Date : 2012-11-23
DMF Number : 21353
Submission : 2008-02-21
Status : Active
Type : II
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
PF614 (Oxycodone) is a controlled substance drug, which is currently being evaluated in Phase III clinical studies for the treatment of chronic pain.
Lead Product(s): Oxycodone Hydrochloride,Inapplicable
Therapeutic Area: Neurology Brand Name: PF614
Study Phase: Phase IIIProduct Type: Controlled Substance
Sponsor: Undisclosed
Deal Size: $20.0 million Upfront Cash: Undisclosed
Deal Type: Financing April 07, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Oxycodone Hydrochloride,Inapplicable
Therapeutic Area : Neurology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : $20.0 million
Deal Type : Financing
Ensysce Biosciences Secures Second Financing to Advance Pain Programs from 2025 Agreement
Details : PF614 (Oxycodone) is a controlled substance drug, which is currently being evaluated in Phase III clinical studies for the treatment of chronic pain.
Product Name : PF614
Product Type : Controlled Substance
Upfront Cash : Undisclosed
April 07, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Oxycodone HCl is a Small Molecule drug candidate drug candidate, which is currently being evaluated in Phase IV clinical studies for the treatment of Pain, Postoperative.
Lead Product(s): Tramadol Hydrochloride,Oxycodone Hydrochloride
Therapeutic Area: Neurology Brand Name: Undisclosed
Study Phase: Phase IVProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable September 03, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Tramadol Hydrochloride,Oxycodone Hydrochloride
Therapeutic Area : Neurology
Highest Development Status : Phase IV
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Oxycodone HCl is a Small Molecule drug candidate drug candidate, which is currently being evaluated in Phase IV clinical studies for the treatment of Pain, Postoperative.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
September 03, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Roxybond (oxycodone HCl) is an opioid agonist indicated for the management of pain severe enough to require an opioid analgesic and for which alternative treatments are inadequate.
Lead Product(s): Oxycodone Hydrochloride,Inapplicable
Therapeutic Area: Neurology Brand Name: Roxybond
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable October 29, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Oxycodone Hydrochloride,Inapplicable
Therapeutic Area : Neurology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Protega’s ROXYBOND™ Gets FDA Approval As Abuse-Deterrent Pain Management Option
Details : Roxybond (oxycodone HCl) is an opioid agonist indicated for the management of pain severe enough to require an opioid analgesic and for which alternative treatments are inadequate.
Product Name : Roxybond
Product Type : Miscellaneous
Upfront Cash : Inapplicable
October 29, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Gabapentin Enacarbil is a Other Small Molecule drug candidate, which is currently being evaluated in phase IV clinical studies for the treatment of Substance-Related Disorders.
Lead Product(s): Gabapentin Enacarbil,Oxycodone Hydrochloride
Therapeutic Area: Psychiatry/Psychology Brand Name: Undisclosed
Study Phase: Phase IVProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable February 08, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Gabapentin Enacarbil,Oxycodone Hydrochloride
Therapeutic Area : Psychiatry/Psychology
Highest Development Status : Phase IV
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Gabapentin Enacarbil is a Other Small Molecule drug candidate, which is currently being evaluated in phase IV clinical studies for the treatment of Substance-Related Disorders.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
February 08, 2024

Details:
Bupivacaine is a Other Small Molecule drug candidate, which is currently being evaluated in clinical studies for the treatment of Funnel Chest.
Lead Product(s): Bupivacaine,Oxycodone Hydrochloride
Therapeutic Area: Musculoskeletal Brand Name: Undisclosed
Study Phase: UndisclosedProduct Type: Miscellaneous
Sponsor: AtriCure
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable February 16, 2023

Lead Product(s) : Bupivacaine,Oxycodone Hydrochloride
Therapeutic Area : Musculoskeletal
Highest Development Status : Undisclosed
Partner/Sponsor/Collaborator : AtriCure
Deal Size : Inapplicable
Deal Type : Inapplicable
Intercostal Nerve Cryoablation Versus Epidural Analgesia for Nuss Repair of Pectus Excavatum
Details : Bupivacaine is a Other Small Molecule drug candidate, which is currently being evaluated in clinical studies for the treatment of Funnel Chest.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
February 16, 2023

Details:
NYX-783 is a Other Small Molecule drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of Opioid-Related Disorders.
Lead Product(s): NYX-783,Oxycodone Hydrochloride
Therapeutic Area: Psychiatry/Psychology Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable July 07, 2022

Lead Product(s) : NYX-783,Oxycodone Hydrochloride
Therapeutic Area : Psychiatry/Psychology
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Safety, Tolerability and Pharmacokinetics of NYX-783 and Oxycodone DDI Study
Details : NYX-783 is a Other Small Molecule drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of Opioid-Related Disorders.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
July 07, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Oxycodone is a Other Small Molecule drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Pain.
Lead Product(s): Oxycodone Hydrochloride,Ibuprofen
Therapeutic Area: Neurology Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable October 18, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Oxycodone Hydrochloride,Ibuprofen
Therapeutic Area : Neurology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Oxycodone is a Other Small Molecule drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Pain.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
October 18, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Delta 9 Tetrahydrocannabinol is a drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Cancer Pain.
Lead Product(s): Delta 9 Tetrahydrocannabinol,Cannabidiol,Oxycodone Hydrochloride
Therapeutic Area: Neurology Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Undisclosed
Sponsor: George Clinical | WriteSource Medical Pty Ltd
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable March 22, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Delta 9 Tetrahydrocannabinol,Cannabidiol,Oxycodone Hydrochloride
Therapeutic Area : Neurology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : George Clinical | WriteSource Medical Pty Ltd
Deal Size : Inapplicable
Deal Type : Inapplicable
NanaBisâ„¢ an Oro-buccal Administered delta9-Tetrahydrocannabinol (d9-THC) & Cannabidiol (CBD)...
Details : Delta 9 Tetrahydrocannabinol is a drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Cancer Pain.
Product Name : Undisclosed
Product Type : Undisclosed
Upfront Cash : Inapplicable
March 22, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Oxycodone is a Other Small Molecule drug candidate, which is currently being evaluated in phase IV clinical studies for the treatment of Pain, Postoperative.
Lead Product(s): Oxycodone Hydrochloride,Inapplicable
Therapeutic Area: Neurology Brand Name: Undisclosed
Study Phase: Phase IVProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable August 21, 2020

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Oxycodone Hydrochloride,Inapplicable
Therapeutic Area : Neurology
Highest Development Status : Phase IV
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Oxycodone is a Other Small Molecule drug candidate, which is currently being evaluated in phase IV clinical studies for the treatment of Pain, Postoperative.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
August 21, 2020

Details:
Oxycodone is a Other Small Molecule drug candidate, which is currently being evaluated in clinical studies for the treatment of Neoplasms.
Lead Product(s): Oxycodone Hydrochloride,Paracetamol
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: UndisclosedProduct Type: Miscellaneous
Sponsor: Proteus
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable December 11, 2019

Lead Product(s) : Oxycodone Hydrochloride,Paracetamol
Therapeutic Area : Oncology
Highest Development Status : Undisclosed
Partner/Sponsor/Collaborator : Proteus
Deal Size : Inapplicable
Deal Type : Inapplicable
S1916 Digital Medicine Program for Pain Control in Cancer Patients
Details : Oxycodone is a Other Small Molecule drug candidate, which is currently being evaluated in clinical studies for the treatment of Neoplasms.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
December 11, 2019

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Dosage Form : Cream / Lotion / Ointment, Suspension
Grade : Topical
Category : API Stability Enhancers, Thickeners and Stabilizers
Dosage Form : Cream / Lotion / Ointment, Capsule, Gel, Tablet
Grade : Oral, Topical
Category : API Stability Enhancers, Thickeners and Stabilizers
Brand Name : Citric Acid 1-Hydrate
Application : API Stability Enhancers
Excipient Details : Citric Acid 1-hydrate is used as a chelating and/or complexing agent in various oral liquid formulations.
Pharmacopoeia Ref : Ph. Eur.|USP
Technical Specs : Not Available
Ingredient(s) : Citric Acid Excipient
Dosage Form : Capsule, Solution, Tablet
Grade : Oral
Category : API Stability Enhancers, Lubricants & Glidants
Application : API Stability Enhancers, Lubricants & Glidants
Excipient Details : Sodium Benzoate is used as a antimicrobial preservative in oral liquid dosage forms.
Pharmacopoeia Ref : Ph. Eur.|NF
Technical Specs : Not Available
Ingredient(s) : Sodium Benzoate Excipient
Dosage Form : Cream / Lotion / Ointment, Solution
Grade : Oral
Category : API Stability Enhancers, Topical
Brand Name : Disodium Phosphate 2-Hydrate
Application : API Stability Enhancers
Excipient Details : Disodium Phosphate 2-hydrate is used as a pH-Modifier (Acidifying/Alkalizing Agent/Buffering Agent) in various oral liquid dosage forms.
Pharmacopoeia Ref : Ph. Eur.|USP
Technical Specs : Not Available
Ingredient(s) : Disodium Phosphate Dihydrate Excipient
Brand Name : Trisodium Citrate 5.5-Hydrate
Application : API Stability Enhancers
Excipient Details : Trisodium Citrate 5.5-hydrate is used as a pH-modifier in oral liquid dosage forms.
Pharmacopoeia Ref : Ph. Eur.|USP
Technical Specs : Not Available
Ingredient(s) : Trisodium Citrate Excipient
Dosage Form : Cream / Lotion / Ointment, Solution
Grade : Oral
Category : API Stability Enhancers, Topical
Brand Name : Trisodium Citrate 2-Hydrate
Application : API Stability Enhancers, Topical
Excipient Details : Trisodium Citrate 2-hydrate is used as a emollient & pH-modifier in oral liquid dosage forms.
Pharmacopoeia Ref : Ph. Eur.|USP
Technical Specs : Not Available
Ingredient(s) : Sodium Citrate Dihydrate Excipient
Application : API Stability Enhancers, Thickeners and Stabilizers
Excipient Details : Adsorbent, Moisture Protection, Stabilization of API
Pharmacopoeia Ref : USP-NF, JP, EP
Technical Specs : Also Available as FUJISIL-F
Ingredient(s) : Silicon Dioxide
Application : Fillers, Diluents & Binders
Excipient Details : It is used as an adhesive agent.
Pharmacopoeia Ref : Not Available
Technical Specs : Also Available as K17, K25, K90 and VA-64
Ingredient(s) : Povidone K30
Global Sales Information

Market Place

Reply
07 Jul 2025
Reply
25 Mar 2025
Reply
06 Feb 2025
Reply
17 Aug 2024
Reply
16 Jul 2024
Reply
27 Apr 2024
Reply
07 Feb 2023
Reply
31 May 2022
Reply
16 Apr 2022
Reply
24 Sep 2020
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
64
PharmaCompass offers a list of Oxycodone Hydrochloride API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Oxycodone Hydrochloride manufacturer or Oxycodone Hydrochloride supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Oxycodone Hydrochloride manufacturer or Oxycodone Hydrochloride supplier.
A Oxycodone Hydrochloride manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Oxycodone Hydrochloride, including repackagers and relabelers. The FDA regulates Oxycodone Hydrochloride manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Oxycodone Hydrochloride API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Oxycodone Hydrochloride manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Oxycodone Hydrochloride supplier is an individual or a company that provides Oxycodone Hydrochloride active pharmaceutical ingredient (API) or Oxycodone Hydrochloride finished formulations upon request. The Oxycodone Hydrochloride suppliers may include Oxycodone Hydrochloride API manufacturers, exporters, distributors and traders.
click here to find a list of Oxycodone Hydrochloride suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Oxycodone Hydrochloride DMF (Drug Master File) is a document detailing the whole manufacturing process of Oxycodone Hydrochloride active pharmaceutical ingredient (API) in detail. Different forms of Oxycodone Hydrochloride DMFs exist exist since differing nations have different regulations, such as Oxycodone Hydrochloride USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Oxycodone Hydrochloride DMF submitted to regulatory agencies in the US is known as a USDMF. Oxycodone Hydrochloride USDMF includes data on Oxycodone Hydrochloride's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Oxycodone Hydrochloride USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Oxycodone Hydrochloride suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Oxycodone Hydrochloride Drug Master File in Japan (Oxycodone Hydrochloride JDMF) empowers Oxycodone Hydrochloride API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Oxycodone Hydrochloride JDMF during the approval evaluation for pharmaceutical products. At the time of Oxycodone Hydrochloride JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Oxycodone Hydrochloride suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Oxycodone Hydrochloride Drug Master File in Korea (Oxycodone Hydrochloride KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Oxycodone Hydrochloride. The MFDS reviews the Oxycodone Hydrochloride KDMF as part of the drug registration process and uses the information provided in the Oxycodone Hydrochloride KDMF to evaluate the safety and efficacy of the drug.
After submitting a Oxycodone Hydrochloride KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Oxycodone Hydrochloride API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Oxycodone Hydrochloride suppliers with KDMF on PharmaCompass.
A Oxycodone Hydrochloride CEP of the European Pharmacopoeia monograph is often referred to as a Oxycodone Hydrochloride Certificate of Suitability (COS). The purpose of a Oxycodone Hydrochloride CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Oxycodone Hydrochloride EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Oxycodone Hydrochloride to their clients by showing that a Oxycodone Hydrochloride CEP has been issued for it. The manufacturer submits a Oxycodone Hydrochloride CEP (COS) as part of the market authorization procedure, and it takes on the role of a Oxycodone Hydrochloride CEP holder for the record. Additionally, the data presented in the Oxycodone Hydrochloride CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Oxycodone Hydrochloride DMF.
A Oxycodone Hydrochloride CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Oxycodone Hydrochloride CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Oxycodone Hydrochloride suppliers with CEP (COS) on PharmaCompass.
A Oxycodone Hydrochloride written confirmation (Oxycodone Hydrochloride WC) is an official document issued by a regulatory agency to a Oxycodone Hydrochloride manufacturer, verifying that the manufacturing facility of a Oxycodone Hydrochloride active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Oxycodone Hydrochloride APIs or Oxycodone Hydrochloride finished pharmaceutical products to another nation, regulatory agencies frequently require a Oxycodone Hydrochloride WC (written confirmation) as part of the regulatory process.
click here to find a list of Oxycodone Hydrochloride suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Oxycodone Hydrochloride as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Oxycodone Hydrochloride API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Oxycodone Hydrochloride as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Oxycodone Hydrochloride and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Oxycodone Hydrochloride NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Oxycodone Hydrochloride suppliers with NDC on PharmaCompass.
Oxycodone Hydrochloride Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Oxycodone Hydrochloride GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Oxycodone Hydrochloride GMP manufacturer or Oxycodone Hydrochloride GMP API supplier for your needs.
A Oxycodone Hydrochloride CoA (Certificate of Analysis) is a formal document that attests to Oxycodone Hydrochloride's compliance with Oxycodone Hydrochloride specifications and serves as a tool for batch-level quality control.
Oxycodone Hydrochloride CoA mostly includes findings from lab analyses of a specific batch. For each Oxycodone Hydrochloride CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Oxycodone Hydrochloride may be tested according to a variety of international standards, such as European Pharmacopoeia (Oxycodone Hydrochloride EP), Oxycodone Hydrochloride JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Oxycodone Hydrochloride USP).

