Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
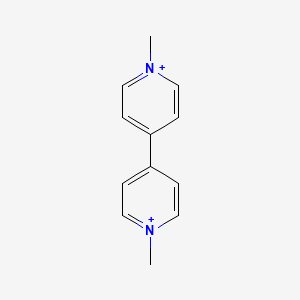

1. Gramoxone
2. Methyl Viologen
3. Paragreen A
4. Viologen, Methyl
1. 4685-14-7
2. 1,1'-dimethyl-4,4'-bipyridinium
3. Paraquat Ion
4. Paraquat Dication
5. Dimethyl Viologen
6. Starfire
7. Weedol
8. 4,4'-bipyridinium, 1,1'-dimethyl-
9. Spraytop-graze
10. Dextrone X
11. Methyl Viologen (2+)
12. Gramoxone
13. Methyl Viologen Ion(2+)
14. N,n'-dimethyl-4,4'-bipyridinium
15. Ccris 7731
16. 1,1'-dimethyl-4,4'-bipyridyldiylium
17. 1-methyl-4-(1-methylpyridin-1-ium-4-yl)pyridin-1-ium
18. Hsdb 1668
19. Esgram
20. 1,1'-dimethyl-4,4'-bipyridinium Salt
21. 1,1'-dimethyl-4,4'-bipyridinium Cation
22. Gramoxone S
23. N,n'-dimethyl-4,4'-bipyridinium Dication
24. 1,1'-dimethyl-4,4'-bipyridin-1-ium
25. Chebi:34905
26. 1,1'-dimethyl-4,4'-bipyridium
27. Plg39h7695
28. Mls001332595
29. Paraquat [ansi:bsi:iso]
30. Smr000875209
31. Einecs 225-141-7
32. N,n'-dimethyl-gamma,gamma'-dipyridylium
33. Unii-plg39h7695
34. Khj
35. Priglone
36. Paraquat Cation
37. Paraquat [hsdb]
38. Paraquat [iso]
39. Paraquat [mi]
40. Paraquat Aqueous Solution
41. Chemdiv3_000231
42. Paraquat [mart.]
43. Ncimech_000502
44. Nciopen2_005422
45. Schembl21652
46. Mls001304933
47. Bidd:er0480
48. Chembl74469
49. 1,1'-dimethyl-4,4'-bipyridyldiylium Ion (8ci)(9ci)
50. Dtxsid3034799
51. Bdbm96275
52. Cid_5351279
53. Hms1473k11
54. Zinc3861781
55. 1,1 '-dimethyl-4,4'-bipyridinium
56. Ccg-35984
57. Stk387391
58. Akos001483177
59. Idi1_019549
60. 1,1'-dimethyl-4,4'-bipyridyl
61. Ncgc00166161-02
62. Nci60_002105
63. Smr000752910
64. 1,1'-dimethyl-4,4'-bipyridyldiylium Ion
65. Ft-0606133
66. Ft-0652363
67. Bipyridinium, 1,1'-dimethyl-4,4'-
68. 1,1'-dimethyl-[4,4'-bipyridin]-1,1'-diium
69. 1,1'-dimethyl-4,4'-bipyridium Bromide (1:2)
70. 685p147
71. Ac-907/25005209
72. W-106083
73. Q26841324
74. 1-methyl-4-(1-methyl-4-pyridin-1-iumyl)pyridin-1-ium;chloride
75. 1-methyl-4-(1-methylpyridin-1-ium-4-yl)pyridin-1-ium;chloride
| Molecular Weight | 186.25 g/mol |
|---|---|
| Molecular Formula | C12H14N2+2 |
| XLogP3 | 1.7 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 0 |
| Rotatable Bond Count | 1 |
| Exact Mass | 186.115698455 g/mol |
| Monoisotopic Mass | 186.115698455 g/mol |
| Topological Polar Surface Area | 7.8 Ų |
| Heavy Atom Count | 14 |
| Formal Charge | 2 |
| Complexity | 145 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Toxic concentration in blood is 0.85 mg% (8.5 ug/mL); lethal concentration in blood is 3.5 mg% (35.0 ug/mL).
Winek, C.L. Drug and Chemical Blood-Level Data 1985. Pittsburgh, PA: Allied Fischer Scientific, 1985.
It has been estimated that a lethal dose in man is about 14 mL of a 40% solution of paraquat.
National Research Council. Drinking Water & Health Volume 1. Washington, DC: National Academy Press, 1977., p. 544
The minimum lethal dose of paraquat is stated to be about 35 mg/kg body weight for human beings.
WHO; Environ Health Criteria: Paraquat and Diquat p.76 (1984)
... High dose (>40 mg paraquat ion/ kg bw = 20 mL of 20-24% concentrate): toxicity is much more severe and death occurs early (24-48 hr) from multiple organ failure. ...
IPCS; Poisons Information Monograph 399: Paraquat (October 2000). Available from, as of March 5, 2010: https://www.inchem.org/pages/pims.html
Lethal concentration in blood is 3.5 mg% (35.0 ug/mL).
Winek, C.L. Drug and Chemical Blood-Level Data 1985. Pittsburgh, PA: Allied Fischer Scientific, 1985.
Herbicides
Pesticides used to destroy unwanted vegetation, especially various types of weeds, grasses (POACEAE), and woody plants. Some plants develop HERBICIDE RESISTANCE. (See all compounds classified as Herbicides.)
In a fatal case of paraquat poisoning in a pregnant woman, who developed the typical symptoms and signs of paraquat poisoning and at postmortem had the typical lung pathology of paraquat poisoning, the fetal lungs were normal. ...However, ...the details /as reported/ of nine pregnant women who deliberately ingested paraquat, stated that paraquat in one case was concentrated 4-6 times in the fetus. In another of the cases, the amniotic fluid contained paraquat at twice the concentration of that in the maternal blood.
FAO/WHO; Joint FAO/WHO Meeting on Pesticide Residues; Pesticide Residues in Food: Paraquat (4685-14-7) (2003). Available from, as of July 22, 2015: https://www.inchem.org/pages/jmpr.html
Paraquat was poorly absorbed after oral administration to rats, dogs and mice. Once absorbed, paraquat was rapidly distributed to most tissues but especially to lungs and kidneys. Tissues other than lungs did not retain paraquat.
USEPA/Office of Pesticide Programs; Reregistration Eligibility Decision Document - Paraquate dichloride p.33 EPA 738-F-96-018 (August 1997). Available from, as of March 2, 2010: https://www.epa.gov/pesticides/reregistration/status.htm
In a dermal absorption study with healthy adult male volunteers, 0.3% of the applied (14)C-paraquat dichloride was absorbed through the intact skin (forearms, and back of the hands and lower legs) during the 24-hour exposure period. /Paraquat dichloride/
USEPA/Office of Pesticide Programs; Reregistration Eligibility Decision Document - Paraquate dichloride p.33 EPA 738-F-96-018 (August 1997). Available from, as of March 2, 2010: https://www.epa.gov/pesticides/reregistration/status.htm
Absorbed paraquat is distributed via the bloodstream to practically all organs and tissues of the body, but no prolonged storage takes place in any tissue. The distribution of paraquat is best described by a three-compartment open model with input to and elimination from the central compartment ... The lung then selectively accumulates paraquat from the plasma, by an energy dependent process.
IPCS; Poisons Information Monograph 399: Paraquat (October 2000). Available from, as of March 5, 2010: https://www.inchem.org/pages/pims.html
For more Absorption, Distribution and Excretion (Complete) data for Paraquat (28 total), please visit the HSDB record page.
Paraquat was not metabolized by rats. After oral administration (gastric intubation) of single doses of paraquat dichloride or dimethylsulfate to Wistar strain male and female rats, most of the administered radioactivity (69-96%) was excreted in feces as unchanged paraquat. After subcutaneous injection of these compounds, unchanged paraquat appeared mostly in urine (73-96% of the administered radioactivity). Paraquat used in this study (radiochemical purity: 99-100%) was labeled with (14)C in the methyl groups. The doses used for gastric intubation ranged from 0.5 to 50 mg/kg and for subcutaneous injection, from 12.5 to 24 mg/kg. Most of the radioactivity was detected in feces within 2-3 days after dosing and in urine, within 1 day after dosing. Following oral administration of paraquat, up to 30% of the dose appeared in feces in a degraded form. This was due to the microbial degradation of paraquat in the gut. That microbial degradation of paraquat occurred in feces was shown in an in vitro experiment in which fecal homogenates were incubated with added paraquat for 24 hours. In that experiment, 40-50% of paraquat was destroyed. However, a similar experiment with sterilized fecal homogenates produced only minor loss (trace amounts) of added paraquat. /Paraquat dichloride/
USEPA/Office of Pesticide Programs; Reregistration Eligibility Decision Document - Paraquate dichloride p.32 EPA 738-F-96-018 (August 1997). Available from, as of March 2, 2010: https://www.epa.gov/pesticides/reregistration/status.htm
Three Warren hens were given gelatin capsules containing (14)C-ring-labelled paraquat (purity, 99.7%; specific activity, 1.216 x 105 dpm/mg) at a daily dose of 4.52 mg of paraquat ion (0.247 mCi) for 10 days. ...Residues in tissues were generally unchanged paraquat. A small amount of a metabolite, 1-methyl-(4'-pyridyl), was found in the livers and kidneys.
FAO/WHO; Pesticide Residues in Food- Joint FAO/WHO Meeting on Pesticide Residues: Paraquat (2003). Available from, as of March 11, 2010: https://www.inchem.org/documents/jmpr/jmpmono/v2003pr08.htm
An English white nanny goat was dosed with (14)C-ring-labelled paraquat (purity, 99.7%; specific activity, 2.28 x 104 dpm/ug) in the diet at a dose equivalent to 100 ug of paraquat ion/g of diet ... for 7 days. ... Residues in tissues were generally unchanged paraquat. In the liver, small amounts of 4-(1,2-dihydro-1-methyl-2-oxo-4-pyridyl)-1-methyl pyridinium ion and 1-methyl-4-(4'-pyridyl) pyridinium ion were found. The latter compound was also found in peritoneal fat.
FAO/WHO; Pesticide Residues in Food- Joint FAO/WHO Meeting on Pesticide Residues: Paraquat (2003). Available from, as of March 11, 2010: https://www.inchem.org/documents/jmpr/jmpmono/v2003pr08.htm
Assessment of accumulation of paraquat in the lungs: 3 week inhalation study in rats (15 exposures). ...Lung concentrations in the high dose group peaked on about day 4 (4.71 mg paraquat/l), dropping to about half that concentration with continued dosing. Mean lung weights increased sharply after the fifth exposure in the high dose group to about 170% of controls. The elimination half-life was about 2 days.
California Environmental Protection Agency/Department of Pesticide Regulation; Toxicology Data Review Summaries: Paraquat dichloride. Available from, as of March 9, 2010: https://www.cdpr.ca.gov/docs/risk/toxsums/toxsumlist.htm
... The concentration-time curve in 15 adult patients (not hemodialyzed) was best described by a bi-exponential curve, with the elimination half-lives of the early and late phase being 5 and 84 hours, respectively.
IPCS; Poisons Information Monograph 399: Paraquat (October 2000). Available from, as of March 5, 2010: https://www.inchem.org/pages/pims.html
A biexponential body burden curve was observed in hamsters following intratracheal instillation of (14)C paraquat. Elimination rate was slow, beta value of 0.014 hr which corresponds to a half-life of 50 hr.
Abou-Donia MB, Komeil AA; Toxicol Appl Pharmacol 45 (1): 280 (1978).
Half-life for absorption of radioactivity from lung of hamster administered dichloride salt intratracheally was 21 hr.
Abou-Donia MB, Komeil AA; Toxicol Appl Pharmacol 45 (1): 280 (1978)
For more Biological Half-Life (Complete) data for Paraquat (6 total), please visit the HSDB record page.
The innate immune response is important in paraquat-induced acute lung injury, but the exact pathways involved are not elucidated. The objectives of this study were to determine the specific role of the NLRP3 inflammasome in the process. Acute lung injury was induced by administering paraquat (PQ) intraperitoneally. NLRP3 inflammasome including NLRP3, ASC, and caspase-1 mRNA and protein expression in lung tissue and IL-1beta and IL-18 levels in BALF were detected at 4, 8, 24, and 72 hr after PQ administration in rats. Moreover, rats were pretreated with 10, 30, and 50 mg/kg NLRP3 inflammasome blocker glybenclamide, respectively, 1 h before PQ exposure. At 72 hr after PQ administration, lung histopathology changes, NLRP3, ASC, and caspase-1 protein expression, as well as secretion of cytokines including IL-1beta and IL-18 in BALF were investigated. The NLRP3 inflammasome including NLRP3, ASC, caspase-1 expression, and cytokines IL-1beta and IL-18 levels in PQ poisoning rats were significantly higher than that in the control group. NLRP3 inflammasome blocker glybenclamide pretreatment attenuated lung edema, inhibited the NLRP3, ASC, and caspase-1 activation, and reduced IL-1beta and IL-18 levels in BALF. In the in vitro experiments, IL-1beta and IL-18 secreted from RAW264.7 mouse macrophages treated with paraquat were attenuated by glybenclamide. In conclusion, paraquat can induce IL-1beta/IL-18 secretion via NLRP3-ASC-caspase-1 pathway, and the NLRP3 inflammasome is essential for paraquat-induced acute lung injury.
PMID:25338942 Liu Z, et al; Inflammation. 2015 Feb;38(1):433-44 (2015)
BACKGROUND: Paraquat (PQ) poisoning is a lethal toxicological challenge that served as a disease model of acute lung injury and pulmonary fibrosis, but the mechanism is undetermined and no effective treatment has been discovered. METHODS AND FINDINGS: We demonstrated that PQ injures mitochondria and leads to mtDNA release. The mtDNA mediated PBMC recruitment and stimulated the alveolar epithelial cell production of TGF-beta1 in vitro. The levels of mtDNA in circulation and bronchial alveolar lavage fluid (BALF) were elevated in a mouse of PQ-induced lung injury. DNaseI could protect PQ-induced lung injury and significantly improved survival. Acute lung injury markers, such as TNFalpha, IL-1beta, and IL-6, and marker of fibrosis, collagen I, were downregulated in parallel with the elimination of mtDNA by DNaseI. These data indicate a possible mechanism for PQ-induced, mtDNA-mediated lung injury, which may be shared by other causes of lung injury, as suggested by the same protective effect of DNaseI in bleomycin-induced lung injury model. Interestingly, increased mtDNA in the BALF of patients with amyopathic dermatomyositis-interstitial lung disease can be appreciated. CONCLUSIONS: DNaseI targeting mtDNA may be a promising approach for the treatment of PQ-induced acute lung injury and pulmonary fibrosis that merits fast tracking through clinical trials.
PMID:25759818 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4339792 Li G, et al; Biomed Res Int 2015:386952 (2015).
Paraquat (PQ), one of the most widely used herbicides, has been used for several decades in agriculture. Some studies suggest that PQ has effects on the immune system. Moreover, previous studies have shown that PQ imparted some immunosuppressive effects. In the present study, cytotoxicity assays using splenic NK cells from mice treated for 28 days with PQ (at 0.2, 1, and 5 mg/kg) were performed to determine whether PQ altered the function of NK cells. Given that PQ was expected to induce an immunosuppressive effect, it was hypothesized that a gene involved in cellular metal ion homeostasis, metallothionein-1 (MT-1), could play an important role in this outcome. This belief was based on the fact that MT1 encodes a protein responsible for zinc ion homeostasis, and that a reduction in free zinc ion levels impairs NK cell function. The results showed that PQ treatments led to increased MT expression in several organs (liver, kidneys, testes) and in splenocytes, caused a reduction of both free zinc ions in sera and in free intracellular zinc, and reduced the expression of GATA-3, a zinc-finger transcription factor important for maturation and activity of T-cells and NK cells. These results provide a basis for a new molecular mechanism to describe potential immunosuppressive effects of PQ in vivo.
PMID:25496228 Lim JH, et al; J Immunotoxicol 15:1-8 (2014).
Paraquat (PQ) causes selective degeneration of dopaminergic neurons in the substantia nigra pars compacta, reproducing an important pathological feature of Parkinson disease. Oxidative stress, c-Jun N-terminal kinase activation, and alpha-synuclein aggregation are each induced by PQ, but details of the cell death mechanisms involved remain unclear ... A Bak-dependent cell death mechanism /has been identified/ that is required for PQ-induced neurotoxicity. PQ induced morphological and biochemical features that were consistent with apoptosis, including dose-dependent cytochrome c release, with subsequent caspase-3 and poly(ADP-ribose) polymerase cleavage. Changes in nuclear morphology and loss of viability were blocked by cycloheximide, caspase inhibitor, and Bcl-2 overexpression. Evaluation of Bcl-2 family members showed that PQ induced high levels of Bak, Bid, BNip3, and Noxa. Small interfering RNA-mediated knockdown of BNip3, Noxa, and Bak each protected cells from PQ, but Bax knockdown did not. Finally, ... the sensitivity of Bak-deficient C57BL mice /was tested/ and found ... to be resistant to PQ treatments that depleted tyrosine hydroxylase immuno-positive neurons in the substantia nigra pars compacta of wild-type mice.
PMID:18056701 Fei Q, et al; J Biol Chem 283 (6): 3357-64 (2008)
For more Mechanism of Action (Complete) data for Paraquat (10 total), please visit the HSDB record page.
ABOUT THIS PAGE
20
PharmaCompass offers a list of Paraquat API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Paraquat manufacturer or Paraquat supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Paraquat manufacturer or Paraquat supplier.
PharmaCompass also assists you with knowing the Paraquat API Price utilized in the formulation of products. Paraquat API Price is not always fixed or binding as the Paraquat Price is obtained through a variety of data sources. The Paraquat Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Paraquat manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Paraquat, including repackagers and relabelers. The FDA regulates Paraquat manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Paraquat API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Paraquat supplier is an individual or a company that provides Paraquat active pharmaceutical ingredient (API) or Paraquat finished formulations upon request. The Paraquat suppliers may include Paraquat API manufacturers, exporters, distributors and traders.
Paraquat Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Paraquat GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Paraquat GMP manufacturer or Paraquat GMP API supplier for your needs.
A Paraquat CoA (Certificate of Analysis) is a formal document that attests to Paraquat's compliance with Paraquat specifications and serves as a tool for batch-level quality control.
Paraquat CoA mostly includes findings from lab analyses of a specific batch. For each Paraquat CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Paraquat may be tested according to a variety of international standards, such as European Pharmacopoeia (Paraquat EP), Paraquat JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Paraquat USP).
