
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
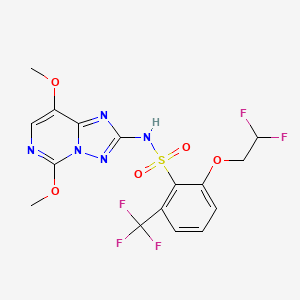

1. 219714-96-2
2. Granite
3. 2-(2,2-difluoroethoxy)-n-(5,8-dimethoxy-[1,2,4]triazolo[1,5-c]pyrimidin-2-yl)-6-(trifluoromethyl)benzenesulfonamide
4. Penoxsulam [iso]
5. 784elc1scz
6. Chebi:81776
7. 2-(2,2-difluoroethoxy)-n-(5,8-dimethoxy[1,2,4]triazolo[1,5-c]pyrimidin-2-yl)-6-(trifluoromethyl)benzenesulfonamide
8. 2-(2,2-difluoroethoxy)-n-(5,8-dimethoxy(1,2,4)triazolo(1,5-c)pyrimidin-2-yl)-6-(trifluoromethyl)benzenesulfonamide
9. Pxd
10. Viper
11. Penoxsulam [mi]
12. Unii-784elc1scz
13. Dsstox_cid_14803
14. Dsstox_rid_79204
15. Dsstox_gsid_34803
16. Schembl116968
17. Chembl1895913
18. Dtxsid0034803
19. Hsdb 7887
20. Amy12535
21. Bcp18718
22. Tox21_301010
23. De 638
24. De-638
25. Mfcd07363876
26. Zinc13827750
27. Akos025401685
28. Penoxsulam 100 Microg/ml In Methanol
29. Ncgc00163715-01
30. Ncgc00163715-02
31. Ncgc00163715-03
32. Ncgc00254912-01
33. Ac-24494
34. Penoxsulam 100 Microg/ml In Acetonitrile
35. Penoxsulam 1000 Microg/ml In Acetonitrile
36. Cas-219714-96-2
37. Ft-0696708
38. Penoxsulam, Pestanal(r), Analytical Standard
39. C13421
40. C18481
41. Q22808507
42. 2-(2,2-difluoroethoxy)-6-trifluoromethyl-n-(5, 8-dimethoxy[1,2,4]triazolo[1,5-c]pyrimidin-2-yl)benzenesulfonamide
43. 2-(2,2-difluoroethoxy)-n-(5,8-dimethoxy[1,2,4]-triazolo[1,5-c]pyrimidin-2-yl)-6-(trifluoromethyl)benzenesulfonamide
44. 2-(2,2-difluoroethoxy)-n-{5,8-dimethoxy-[1,2,4]triazolo[1,5-c]pyrimidin-2-yl}-6-(trifluoromethyl)benzene-1-sulfonamide
45. 2-(2,2-difluoroethyl)-n-(5,8-dimethoxy[1,2,4]triazolo[1,5-c]pyrimidin-2-yl)-6-(trifluoromethyl)benzenesulfonamide
46. 3-(2,2-difluoroethoxy)-n-(5,8-dimethoxy(1,2,4)triazolo(1,5-c)pyrimidin-2-yl)-.alpha.,.alpha.,.alpha.-trifluorotoluene-2-sulfonamide
47. Benzenesulfonamide, 2-(2,2-difluoroethoxy)-n-(5,8-dimethoxy(1,2,4)triazolo(1,5-c)pyrimidin-2-yl)-6-(trifluoromethyl)-
| Molecular Weight | 483.4 g/mol |
|---|---|
| Molecular Formula | C16H14F5N5O5S |
| XLogP3 | 2.9 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 14 |
| Rotatable Bond Count | 8 |
| Exact Mass | 483.06358055 g/mol |
| Monoisotopic Mass | 483.06358055 g/mol |
| Topological Polar Surface Area | 125 Ų |
| Heavy Atom Count | 32 |
| Formal Charge | 0 |
| Complexity | 727 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
All dosing was by gavage with labeled penoxsulam (XDE-638) (97.5% purity of unlabeled penoxsulam, triazole label purity > 99%, phenyl ring label purity 98.4%) in 0.5% Methocel suspension. Generally, groups of 4 rats/sex were used in each of 8 tests using triazole-labeled 14C-XDE-638. One additional set of 4 males was tested using phenyl-labeled 14C-XDE-638 to assess the extent of cleavage between triazole and phenyl moieties. Rats fitted with jugular vein cannulae were used primarily to determine the time course of residue concentrations in plasma, urine, and feces after single dosing with either 5 or 250 mg/kg penoxsulam. These rats were sacrificed on day 7 (as were the phenyl-labeled males), and tissue residues were determined at that time. Time to maximal plasma concentration (Cmax) was about 0.5 hr and 2 hr for both sexes at 5 and 250 mg/kg, respectively. Time to half of maximal concentration of radiolabel (1/2 Cmax) at 5 and 250 mg/kg were determined to be 2.6 hr and 3.0 hr for males, respectively, and 2.9 hr and 5.6 hr for females, respectively. Thus four sets of males and females were sacrificed following single oral dosing to achieve approximately Cmax and 1/2 Cmax plasma levels at the two dose levels, and to obtain tissue and excreta samples at early stages of exposure. Radiolabel was measured in each of 24 tissues at each sacrifice time. Quantities of major metabolites were assessed in pooled plasma at various intervals, and in liver and kidney tissue at sacrifice times. Three rats/sex were fitted with bile duct cannulae, and bile was collected at intervals over 24 hr to assess biliary excretion rate and metabolite analysis. Four additional rats/sex were dosed daily for 15 days with 5 mg/kg/day prior to treatment with triazole labeled 14C-XDE-638 on day 16. These rats were evaluated for residues in excreta and for tissue levels of metabolites, which were found to be comparable to non-pre-treated rats. Metabolite separation was by HPLC using C-18 stationary phase and a gradient program sufficient to separate most major peaks for detection by UV (254 nm) and by 14C-detector. Metabolite identification was by retention time and negative ion LC/MS. Estimated total absorption after 5 mg/kg was about 85% for either sex). Absorption was much lower after 250 mg/kg (17% in males, 32% in females). Unless otherwise stated, results below derive from single-dose administration of 5 mg/kg triazole-labeled penoxsulam. A comparison of distribution patterns of urinary and fecal metabolites following phenyl- or triazole-label indicated that most (at least 90%) of penoxsulam residues remained intact between the two labeled rings. Parent compound was the most abundant urinary residue, constituting about 31% and 19% of administered dose in urine samples of males dosed with triazole- or phenyl-labeled 14C, respectively. Females consistently excreted much larger percentages of label in urine than did males. In the 7-day urine collection in females, 66% of administered dose was parent penoxsulam. The most abundant fecal metabolite in either sex in the 7-day collection was an uncharacterized 'Metabolite Y,' comprising 14% to 19% of administered dose in males and 6% in females. Parent penoxsulam constituted 12% to 15% of administered label in feces of males and 3% in females. About 88% of 24-hr fecal label in males represented absorbed penoxsulam, based on percentage of label excreted in the bile. As expected, considering the proportionally higher excretion of label in females via the urine, females excreted much less in 24-hr bile collections than did males (14% of administered dose in females vs. 56% of administered dose in males). The largest single provisionally identified component of bile was the glutathione product of 5-hydroxy- or 8-hydroxy-penoxsulam (18% of administered penoxsulam). Two glucuronide products of hydroxylated penoxsulam (position of hydroxylation product unknown in either case) appeared to account together for about 15% of administered label in bile. Tissue concentration evaluations during times of 1/2 Cmax plasma concentrations revealed relatively high initial liver residue concentrations, whereas other tissues (except for 'GI/Ingesta') had much lower levels. Radiolabel in liver and in all other tissues were very low by day 7. Parent penoxsulam was much more abundant than any other residues in plasma, liver, and kidneys. Comparative concentrations of penoxsulam in ug-equivalents/g tissue at the time of plasma 1/2 Cmax for these tissues in males and females, respectively, were 10 and 9 for plasma, 44 and 50 for liver, and 3 and 4 for kidney. Originally, this study was unacceptable but upgradeable with identification of 'Metabolite Y,' which constituted up to 19% of fecal residues
California Environmental Protection Agency/Department of Pesticide Regulation; Summary of Toxicology Data on Penoxsulam p.11 (2005). Available from, as of February 7, 2011: https://www.cdpr.ca.gov/docs/risk/toxsums/toxsumlist.htm
In a metabolism study in rats, (14)C-penoxsulam was rapidly and nearly completely absorbed at the low dose of 5.0 mg/kg, but at the high dose of 250 mg/kg, there was evidence that absorption was largely incomplete (i.e. absorption was saturated). Both gender and dose affected the excretion pattern. At the low dose, the major route of excretion of radioactivity was via the feces in males and via the urine in females. At the high dose, radioactivity was predominantly excreted via the feces in both sexes. A significant enterohepatic circulation was observed, particularly in males. Most (>90%) of the administered dose was excreted within 36-48 hours. There was negligible radioactivity in tissues at 7 days and no evidence of accumulation in any tissue/organ. Although numerous metabolites were revealed in the urine, feces and bile, nearly all were <1% of the administered dose. Parent compound and a 2-hydroxyphenyl derivative were the major compounds in urine and feces.
US EPA Office of Prevention, Pesticides and Toxic Substances, Pesticide Fact Sheet for Penoxsulam, p.5 (September 2004). Available from, as of February 4, 2011: https://www.epa.gov/opprd001/factsheets/
All dosing was by gavage with labeled penoxsulam (XDE-638) (97.5% purity of unlabeled penoxsulam, triazole label purity > 99%, phenyl ring label purity 98.4%) in 0.5% Methocel suspension. Generally, groups of 4 rats/sex were used in each of 8 tests using triazole-labeled 14C-XDE-638. One additional set of 4 males was tested using phenyl-labeled 14C-XDE-638 to assess the extent of cleavage between triazole and phenyl moieties. Rats fitted with jugular vein cannulae were used primarily to determine the time course of residue concentrations in plasma, urine, and feces after single dosing with either 5 or 250 mg/kg penoxsulam. ... Quantities of major metabolites were assessed in pooled plasma at various intervals, and in liver and kidney tissue at sacrifice times. Three rats/sex were fitted with bile duct cannulae, and bile was collected at intervals over 24 hr to assess biliary excretion rate and metabolite analysis. Four additional rats/sex were dosed daily for 15 days with 5 mg/kg/day prior to treatment with triazole labeled 14C-XDE-638 on day 16. These rats were evaluated for residues in excreta and for tissue levels of metabolites, which were found to be comparable to non-pre-treated rats. Metabolite separation was by HPLC using C-18 stationary phase and a gradient program sufficient to separate most major peaks for detection by UV (254 nm) and by 14C-detector. Metabolite identification was by retention time and negative ion LC/MS. ... A comparison of distribution patterns of urinary and fecal metabolites following phenyl- or triazole-label indicated that most (at least 90%) of penoxsulam residues remained intact between the two labeled rings. Parent compound was the most abundant urinary residue, constituting about 31% and 19% of administered dose in urine samples of males dosed with triazole- or phenyl-labeled 14C, respectively. ... The most abundant fecal metabolite in either sex in the 7-day collection was an uncharacterized 'Metabolite Y,' comprising 14% to 19% of administered dose in males and 6% in females. Parent penoxsulam constituted 12% to 15% of administered label in feces of males and 3% in females. ... The largest single provisionally identified component of bile was the glutathione product of 5-hydroxy- or 8-hydroxy-penoxsulam (18% of administered penoxsulam). Two glucuronide products of hydroxylated penoxsulam (position of hydroxylation product unknown in either case) appeared to account together for about 15% of administered label in bile. ...
California Environmental Protection Agency/Department of Pesticide Regulation; Summary of Toxicology Data on Penoxsulam p.11 (2005). Available from, as of February 7, 2011: https://www.cdpr.ca.gov/docs/risk/toxsums/toxsumlist.htm
Sulfonamide herbicides ... inhibit the enzyme acetolactate synthase (ALS)... /Sulfonamides/
PMID:19464188 Johnson TC et al; Bioorg Med Chem 17 (12): 4230-40 (2009)
ABOUT THIS PAGE
92
PharmaCompass offers a list of Penoxsulam API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Penoxsulam manufacturer or Penoxsulam supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Penoxsulam manufacturer or Penoxsulam supplier.
PharmaCompass also assists you with knowing the Penoxsulam API Price utilized in the formulation of products. Penoxsulam API Price is not always fixed or binding as the Penoxsulam Price is obtained through a variety of data sources. The Penoxsulam Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Penoxsulam manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Penoxsulam, including repackagers and relabelers. The FDA regulates Penoxsulam manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Penoxsulam API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Penoxsulam manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Penoxsulam supplier is an individual or a company that provides Penoxsulam active pharmaceutical ingredient (API) or Penoxsulam finished formulations upon request. The Penoxsulam suppliers may include Penoxsulam API manufacturers, exporters, distributors and traders.
click here to find a list of Penoxsulam suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
Penoxsulam Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Penoxsulam GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Penoxsulam GMP manufacturer or Penoxsulam GMP API supplier for your needs.
A Penoxsulam CoA (Certificate of Analysis) is a formal document that attests to Penoxsulam's compliance with Penoxsulam specifications and serves as a tool for batch-level quality control.
Penoxsulam CoA mostly includes findings from lab analyses of a specific batch. For each Penoxsulam CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Penoxsulam may be tested according to a variety of international standards, such as European Pharmacopoeia (Penoxsulam EP), Penoxsulam JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Penoxsulam USP).
