
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
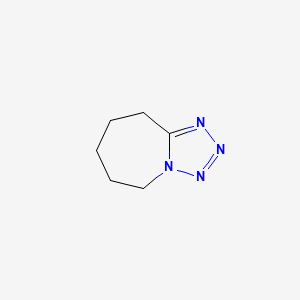

1. Cardiazol
2. Corasol
3. Corazol
4. Corazole
5. Korazol
6. Korazole
7. Leptazole
8. Metrazol
9. Metrazole
10. Pentamethylenetetrazole
11. Pentazol
12. Pentetrazole
13. Pentylenetetrazol
14. Pentylenetetrazole
1. Pentylenetetrazol
2. Pentylenetetrazole
3. 54-95-5
4. Metrazole
5. Cardiazol
6. 1,5-pentamethylenetetrazole
7. Cardiazole
8. Corazole
9. Korazol
10. Metrazol
11. Pentetrazole
12. Coranormol
13. Korazole
14. Leptazol
15. Leptazole
16. Cardiol
17. Cenazol
18. Corazol
19. Coryvet
20. Pentazol
21. Pentamethylenetetrazole
22. Cardiotonicum
23. Pentamethazol
24. Pentamethylenetetrazol
25. Cardifortan
26. Cardosal
27. Cardosan
28. Coratoline
29. Corvasol
30. Deumacard
31. Diovascole
32. Kardiazol
33. Phrenazol
34. Ventrazol
35. Corasol
36. Gewazol
37. 6,7,8,9-tetrahydro-5h-tetrazolo[1,5-a]azepine
38. Pentamethazolum
39. Angiazol
40. Centrazole
41. Coranormal
42. Corsedrol
43. Deamocard
44. Nauranzol
45. Naurazol
46. Nedcardol
47. Neocardol
48. Neurazol
49. Pemetesan
50. Penetrasol
51. Pentacard
52. Pentazolum
53. Pentemesan
54. Pentrazol
55. Pentrolone
56. Pentrozol
57. Petrazole
58. Phrenazone
59. Tetracor
60. Tetrazol
61. Yetrazol
62. Corisan
63. Cortis
64. Corvis
65. Opticor
66. Vasorex
67. Cerebro-nicin
68. Novo Cora-vinco
69. Cenalene-m
70. Metalex-p
71. Corazole (analeptic)
72. Delzol-w
73. Pentamethylene-1,5-tetrazole
74. Tetrazole, Pentamethylene-
75. Tt87
76. Penetratsol
77. Pentetrazolum
78. Stellacardiol
79. Stillcardiol
80. Angioton
81. Lepazol
82. Penetiazol
83. Pentacor
84. Petazol
85. Petezol
86. Alpha,beta-cyclopentamethylenetetrazole
87. Ptz
88. 6,7,8,9-tetrahydro-5-azepotetrazole
89. Tetrazole, (1,5-pentanediyl)-
90. Pentametilentetrazolo
91. Pentetrazol [inn]
92. 6,7,8,9-tetrahydro-5h-tetrazoloazepine
93. 5h-tetrazolo[1,5-a]azepine, 6,7,8,9-tetrahydro-
94. Cardiozol
95. Su-zol
96. Pentamethylenetetrazal
97. Nsc-5729
98. Nsc-66489
99. 1,2,3,3a-tetrazacyclohepta-8a,2-cyclopentadiene
100. 5h-tetrazolo(1,5-a)azepine, 6,7,8,9-tetrahydro-
101. .alpha.,.beta.-cyclopentamethylenetetrazole
102. Wm5z385k7t
103. Chebi:34910
104. Nsc5729
105. 6,7,8,9-tetrahydro-5h-tetrazolo(1,5-a)azepine
106. Nsc66489
107. Pentetrazol (inn)
108. 7,8,9,10-tetrazabicyclo(5.3.0)-8,10-decadiene
109. 7,8,9,10-tetrazabicyclo[5.3.0]-8,10-decadiene
110. Cas-54-95-5
111. Ncgc00015827-06
112. Pentetratsol
113. Corazolum
114. Pentrazole
115. Tetrazole,5-pentanediyl)-
116. Wln: T57 Annnn&tj
117. Pmt
118. Pentetrazolo [dcit]
119. Pentetrazolo
120. 6,8,9-tetrahydro-5-azepotetrazole
121. 6,8,9-tetrahydro-5h-tetrazoloazepine
122. Pentetrazolum [inn-latin]
123. 5h-tetrazoloazepine,7,8,9-tetrahydro-
124. 5h-tetrazolo[1, 6,7,8,9-tetrahydro-
125. Pentametilentetrazolo [italian]
126. 1,3,3a-tetrazacyclohepta-8a,2-cyclopentadiene
127. Pentylenetetrazol [nf]
128. 7,9,10-tetrazabicyclo[5.3.0]-8,10-decadiene
129. Sr-01000076073
130. Nsc 5729
131. Einecs 200-219-3
132. Nsc 66489
133. Brn 0135492
134. Unii-wm5z385k7t
135. Pentavet
136. Pentylentetrazole
137. Ai3-23994
138. Cenalene M
139. Delzol W
140. Prestwick_382
141. Mfcd00005939
142. Pentamethylene Tetrazole
143. Spectrum_000393
144. Specplus_000787
145. 5h-tetrazoloazepine, 6,7,8,9-tetrahydro-
146. Prestwick0_000162
147. Prestwick1_000162
148. Prestwick2_000162
149. Prestwick3_000162
150. Spectrum2_001121
151. Spectrum3_001408
152. Spectrum4_000242
153. Spectrum5_001338
154. Lopac-p-6500
155. Biomol-nt_000269
156. Coryvet [veterinary] (tn)
157. Dsstox_cid_21091
158. Dsstox_rid_79624
159. Pentetrazol [mart.]
160. Dsstox_gsid_41091
161. Lopac0_000940
162. Schembl22674
163. 1, 5-pentamethylenetetrazole
164. 1,5-pentamethylene Tetrazole
165. Bspbio_000023
166. Bspbio_003056
167. Kbiogr_000644
168. Kbioss_000873
169. Pentetrazol [who-dd]
170. 4-26-00-01713 (beilstein Handbook Reference)
171. Divk1c_006883
172. Spectrum2300347
173. Spbio_000981
174. Spbio_001944
175. Pentylenetetrazole [mi]
176. Bpbio1_000027
177. Bpbio1_000860
178. Chembl116943
179. Zinc1905
180. Dtxsid7041091
181. Kbio1_001827
182. Kbio2_000873
183. Kbio2_003441
184. Kbio2_006009
185. Kbio3_002276
186. Pentylenetetrazol [vandf]
187. Hms1568b05
188. Hms2093p18
189. Hms2095b05
190. Hms3262l22
191. Hms3269n13
192. Hms3414p17
193. Hms3678p15
194. Hms3712b05
195. Hms3885p08
196. Pharmakon1600-02300347
197. Amy22325
198. Hy-b1242
199. Tox21_110234
200. Tox21_500940
201. Ccg-40268
202. Nsc759592
203. S4587
204. Stl356056
205. Akos005207166
206. Cs-4890
207. Db13415
208. Lp00940
209. Nsc-759592
210. Sb17494
211. Sdccgsbi-0050914.p004
212. Ncgc00015827-01
213. Ncgc00015827-02
214. Ncgc00015827-03
215. Ncgc00015827-04
216. Ncgc00015827-05
217. Ncgc00015827-07
218. Ncgc00015827-08
219. Ncgc00015827-09
220. Ncgc00015827-17
221. Ncgc00094246-01
222. Ncgc00094246-02
223. Ncgc00094246-03
224. Ncgc00094246-04
225. Ncgc00094246-05
226. Ncgc00261625-01
227. As-47604
228. 6,7,8, 9-tetrahydro-5h-tetrazoloazepine
229. Sbi-0050914.p003
230. Ab00052439
231. B7152
232. Eu-0100940
233. P0046
234. 5h-tetrazoloazepine, 6,7,8, 9-tetrahydro-
235. D07409
236. F14615
237. P 6500
238. Ab00052439_04
239. Q412391
240. 1,2,3, 3a-tetrazacyclohepta-8a,2-cyclopentadiene
241. 1,8,9,10-tetrazabicyclo[5.3.0]deca-7,9-diene
242. 6,7,8,9-tetrahydro-5h-tetraazolo[1,5-a]azepine
243. 6,7,8,9-tetrahydro-5h-tetrazolo[1,5 -a]azepine
244. Sr-01000076073-1
245. Sr-01000076073-3
246. Sr-01000076073-5
247. 6,7,8,9-tetrahydro-5h-tetraazolo[1,5-a]azepine #
248. 7,8,9,10-tetrazabicyclo[5.3.0]deca-8,10-diene
249. Brd-k57718010-001-01-0
250. Brd-k57718010-001-08-5
251. {5h-tetrazolo[1,5-a]azepine,} 6,7,8,9-tetrahydro-
252. 7,8,9, {10-tetrazabicyclo[5.3.0]-8,10-decadiene}
253. 20192-24-9
| Molecular Weight | 138.17 g/mol |
|---|---|
| Molecular Formula | C6H10N4 |
| XLogP3 | 0.1 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Exact Mass | 138.090546336 g/mol |
| Monoisotopic Mass | 138.090546336 g/mol |
| Topological Polar Surface Area | 43.6 Ų |
| Heavy Atom Count | 10 |
| Formal Charge | 0 |
| Complexity | 116 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Convulsants
Substances that act in the brain stem or spinal cord to produce tonic or clonic convulsions, often by removing normal inhibitory tone. They were formerly used to stimulate respiration or as antidotes to barbiturate overdose. They are now most commonly used as experimental tools. (See all compounds classified as Convulsants.)
GABA Antagonists
Drugs that bind to but do not activate GABA RECEPTORS, thereby blocking the actions of endogenous GAMMA-AMINOBUTYRIC ACID and GABA RECEPTOR AGONISTS. (See all compounds classified as GABA Antagonists.)
R - Respiratory system
R07 - Other respiratory system products
R07A - Other respiratory system products
R07AB - Respiratory stimulants
R07AB03 - Pentetrazol
Global Sales Information

ABOUT THIS PAGE
46
PharmaCompass offers a list of Pentylenetetrazole API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Pentylenetetrazole manufacturer or Pentylenetetrazole supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Pentylenetetrazole manufacturer or Pentylenetetrazole supplier.
A Pentylenetetrazole manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Pentylenetetrazole, including repackagers and relabelers. The FDA regulates Pentylenetetrazole manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Pentylenetetrazole API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Pentylenetetrazole supplier is an individual or a company that provides Pentylenetetrazole active pharmaceutical ingredient (API) or Pentylenetetrazole finished formulations upon request. The Pentylenetetrazole suppliers may include Pentylenetetrazole API manufacturers, exporters, distributors and traders.
Pentylenetetrazole Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Pentylenetetrazole GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Pentylenetetrazole GMP manufacturer or Pentylenetetrazole GMP API supplier for your needs.
A Pentylenetetrazole CoA (Certificate of Analysis) is a formal document that attests to Pentylenetetrazole's compliance with Pentylenetetrazole specifications and serves as a tool for batch-level quality control.
Pentylenetetrazole CoA mostly includes findings from lab analyses of a specific batch. For each Pentylenetetrazole CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Pentylenetetrazole may be tested according to a variety of international standards, such as European Pharmacopoeia (Pentylenetetrazole EP), Pentylenetetrazole JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Pentylenetetrazole USP).

