
Synopsis
Synopsis
0
JDMF
0
NDC API
0
VMF
0
FDA Orange Book

0
Canada

0
Australia

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
US Medicaid
NA
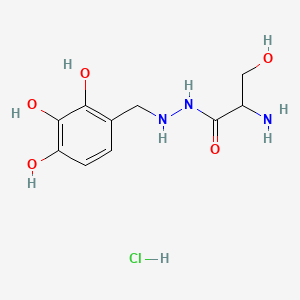

1. 14919-77-8
2. Benserazide Hcl
3. 2-amino-3-hydroxy-n'-(2,3,4-trihydroxybenzyl)propanehydrazide Hydrochloride
4. Benzerazide Hydrochloride
5. Benserazide (hydrochloride)
6. 2'-(2,3,4-trihydroxybenzyl)-dl-serinohydrazide Monohydrochloride
7. Ro 4-4602/001
8. Benserazide Hydrochloride [jan]
9. 14046-64-1
10. Dl-serine 2-(2,3,4-trihydroxybenzyl)hydrazine Hydrochloride
11. 2-amino-3-hydroxy-n'-[(2,3,4-trihydroxyphenyl)methyl]propanehydrazide Hydrochloride
12. Mls000028424
13. B66e5rk36q
14. Dl-serine 2-(2,3,4-trihydroxybenzyl)hydrazide Hydrochloride
15. Chebi:31262
16. (s)-2-amino-3-hydroxy-n'-(2,3,4-trihydroxybenzyl)propanehydrazide Hydrochloride
17. 2-amino-3-hydroxy-n'-[(2,3,4-trihydroxyphenyl)methyl]propanehydrazide;hydrochloride
18. Nsc-755907
19. Smr000058421
20. Ro-4-4602
21. Dsstox_cid_25412
22. Dsstox_rid_80862
23. Dsstox_gsid_45412
24. Benserazide (as Hydrochloride)
25. 3-hydroxy-1-oxo-1-[2-(2,3,4-trihydroxybenzyl)hydrazinyl]propan-2-aminium Chloride
26. Ccris 5092
27. Sr-01000003044
28. Einecs 238-991-9
29. Unii-b66e5rk36q
30. Mfcd00078571
31. Opera_id_1594
32. Ncgc00016709-01
33. Madopa (hoffmann-laroche)
34. Cas-14919-77-8
35. Mls001148252
36. Mls002222160
37. Schembl364720
38. Spectrum1500137
39. Chembl1255778
40. Dtxsid0045412
41. Hy-b0404a
42. Benserazide Hydrochloride (jp17)
43. Benserazide Hydrochloride - Bio-x
44. Hms1570l18
45. Hms1920g05
46. Pharmakon1600-01500137
47. Bcp14068
48. Cba17287
49. Tox21_110573
50. Tox21_500233
51. Ccg-38998
52. Nsc755907
53. S2453
54. Serine, 2-(2,3,4-trihydroxybenzyl)hydrazide, Monohydrochloride, Dl-
55. Benserazide Hydrochloride [mi]
56. Akos007930267
57. Dl-serine, 2-((2,3,4-trihydroxyphenyl)methyl)hydrazide, Monohydrochloride
58. Tox21_110573_1
59. Ac-6825
60. H22b350
61. Lp00233
62. Nsc 755907
63. Sb44383
64. Benserazide Middot Hcl (ro 4-4602)
65. Benserazide Hydrochloride [mart.]
66. Ncgc00015163-09
67. Ncgc00093698-01
68. Ncgc00093698-02
69. Ncgc00093698-03
70. Ncgc00093698-04
71. Ncgc00093698-05
72. Ncgc00260918-01
73. As-13683
74. Bb166155
75. Benserazide Hydrochloride [who-dd]
76. B4108
77. Eu-0100233
78. Ft-0659734
79. Ft-0760799
80. Sw196391-3
81. B 7283
82. Benserazide Hydrochloride [ep Monograph]
83. D01653
84. D70746
85. Benserazide Hydrochloride, >=98% (tlc), Solid
86. A808862
87. Sr-01000003044-2
88. Sr-01000003044-8
89. W-108090
90. Q27114249
91. 2-amino-3-hydroxy-n'-(2,3,4-trihydroxybenzyl)propanehydrazidehydrochloride
92. Benserazide Hydrochloride, European Pharmacopoeia (ep) Reference Standard
93. Dl-serine, 2-[(2,3,4-trihydroxyphenyl)methyl]hydrazide Hydrochloride
94. Serine, 2-[(2,3,4-trihydroxyphenyl)methyl]hydrazide, Hydrochloride (1:1)
95. 2-amino-3-hydroxy-n'-(2,3,4-trihydroxybenzyl)-propanehydrazide Hydrochloride
96. Benserazide For Peak Identification, European Pharmacopoeia (ep) Reference Standard
97. 2-amino-3-hydroxy-n'-(2,3,4-trihydroxybenzyl)-propanehydrazide Hydrochloride;benserazide Hcl
| Molecular Weight | 293.70 g/mol |
|---|---|
| Molecular Formula | C10H16ClN3O5 |
| Hydrogen Bond Donor Count | 8 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 5 |
| Exact Mass | 293.0778483 g/mol |
| Monoisotopic Mass | 293.0778483 g/mol |
| Topological Polar Surface Area | 148 Ų |
| Heavy Atom Count | 19 |
| Formal Charge | 0 |
| Complexity | 278 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |

Certificate Number : R0-CEP 2020-137 - Rev 00
Issue Date : 2022-08-29
Type : Chemical
Substance Number : 1173
Status : Valid

Registrant Name : Aging Life Science Co., Ltd.
Registration Date : 2021-03-17
Registration Number : 20210317-211-J-888
Manufacturer Name : Fermion Oy
Manufacturer Address : Laaketehtaantie 2, Oulu, FI-90660, Finland
 Zhejiang Hengkang Pharm Group is a dynamic pharmaceutical entity, spanning drug research, large-scale production, and global marketing.
Zhejiang Hengkang Pharm Group is a dynamic pharmaceutical entity, spanning drug research, large-scale production, and global marketing.

Certificate Number : CEP 2022-310 - Rev 01
Issue Date : 2023-12-11
Type : Chemical
Substance Number : 1173
Status : Valid
 Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
 SCI Pharmtech offers high-quality, cost-effective APIs, advanced intermediates, & custom products with global expertise and precision.
SCI Pharmtech offers high-quality, cost-effective APIs, advanced intermediates, & custom products with global expertise and precision.

Certificate Number : R0-CEP 2023-108 - Rev 00
Issue Date : 2023-07-17
Type : Chemical
Substance Number : 1173
Status : Valid

Registrant Name : Aging Life Science Co., Ltd.
Registration Date : 2021-03-17
Registration Number : 20210317-211-J-888
Manufacturer Name : Fermion Oy
Manufacturer Address : Laaketehtaantie 2, Oulu, FI-90660, Finland
 Zhejiang Hengkang Pharm Group is a dynamic pharmaceutical entity, spanning drug research, large-scale production, and global marketing.
Zhejiang Hengkang Pharm Group is a dynamic pharmaceutical entity, spanning drug research, large-scale production, and global marketing.

Certificate Number : R0-CEP 2018-183 - Rev 00
Issue Date : 2021-09-27
Type : Chemical
Substance Number : 1173
Status : Valid

Date of Issue : 2023-04-11
Valid Till : 2026-01-26
Written Confirmation Number : ZJ230055
Address of the Firm :

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 36473
Submission : 2022-06-03
Status : Active
Type : II

Certificate Number : R1-CEP 2010-267 - Rev 02
Issue Date : 2018-05-14
Type : Chemical
Substance Number : 1173
Status : Valid


Certificate Number : R0-CEP 2021-345 - Rev 00
Issue Date : 2023-08-25
Type : Chemical
Substance Number : 1173
Status : Valid

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Global Sales Information

Market Place

Reply
01 Aug 2024
Reply
19 Apr 2021
Reply
08 Apr 2021
Reply
07 Apr 2021
Reply
18 Aug 2020
Reply
07 Jul 2020
Reply
14 Nov 2019
Reply
02 Jul 2019
Reply
29 Apr 2017
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?