

Synopsis
0
VMF
0
Australia

0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
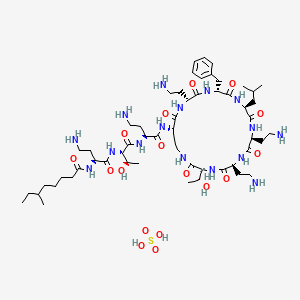

1. Polymyxin B, Sulfate (salt)
2. 1405-20-5
3. Polymixin B Sulphate
4. Ks-1428
5. Pmb
6. Akos025392172
7. Polymyxin B Sulfate, Bacillus Polymyxa
8. M02428
9. Polymyxin B Sulfate (1404-26-8 Free Base)
10. 405p205
| Molecular Weight | 1301.6 g/mol |
|---|---|
| Molecular Formula | C56H100N16O17S |
| Hydrogen Bond Donor Count | 20 |
| Hydrogen Bond Acceptor Count | 22 |
| Rotatable Bond Count | 29 |
| Exact Mass | 1300.71730696 g/mol |
| Monoisotopic Mass | 1300.71730696 g/mol |
| Topological Polar Surface Area | 574 Ų |
| Heavy Atom Count | 90 |
| Formal Charge | 0 |
| Complexity | 2240 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 12 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Polymyxin b sulfate |
| Drug Label | Polymyxin B for Injection, USP is one of a group of basic polypeptide antibiotics derived from B polymyxa (B aerosporous). Polymyxin B sulfate is the sulfate salt of Polymyxins B1 and B2, which are produced by the growth of Bacillus polymyxa (Prazmow... |
| Active Ingredient | Polymyxin b sulfate |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | eq 500,000 u base/vial |
| Market Status | Prescription |
| Company | X Gen Pharms; Xellia Pharms Aps; Fresenius Kabi Usa; Eurohlth Intl |
| 2 of 2 | |
|---|---|
| Drug Name | Polymyxin b sulfate |
| Drug Label | Polymyxin B for Injection, USP is one of a group of basic polypeptide antibiotics derived from B polymyxa (B aerosporous). Polymyxin B sulfate is the sulfate salt of Polymyxins B1 and B2, which are produced by the growth of Bacillus polymyxa (Prazmow... |
| Active Ingredient | Polymyxin b sulfate |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | eq 500,000 u base/vial |
| Market Status | Prescription |
| Company | X Gen Pharms; Xellia Pharms Aps; Fresenius Kabi Usa; Eurohlth Intl |
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 23997
Submission : 2010-07-02
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-09-29
Pay. Date : 2013-12-11
DMF Number : 23876
Submission : 2010-05-31
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 13199
Submission : 1998-04-30
Status : Inactive
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2013-11-20
Pay. Date : 2013-11-08
DMF Number : 22030
Submission : 2008-10-01
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 35892
Submission : 2021-07-08
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 31587
Submission : 2017-03-15
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 21244
Submission : 2008-01-09
Status : Inactive
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2012-11-08
Pay. Date : 2012-10-31
DMF Number : 13536
Submission : 1998-09-01
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2012-11-08
Pay. Date : 2012-10-31
DMF Number : 14187
Submission : 1999-06-10
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 28616
Submission : 2014-09-08
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Polymyxin B sulfate, Micronised
Certificate Number : R1-CEP 2011-166 - Rev 00
Status : Valid
Issue Date : 2017-12-20
Type : Chemical
Substance Number : 203

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R1-CEP 2009-159 - Rev 02
Status : Valid
Issue Date : 2021-01-08
Type : Chemical
Substance Number : 203

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : CEP 2021-287 - Rev 01
Status : Valid
Issue Date : 2023-10-04
Type : Chemical and TSE
Substance Number : 203

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : CEP 2023-251 - Rev 00
Status : Valid
Issue Date : 2024-03-19
Type : Chemical
Substance Number : 203

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R1-CEP 1998-062 - Rev 06
Status : Valid
Issue Date : 2019-04-05
Type : Chemical
Substance Number : 203

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R1-CEP 1998-061 - Rev 05
Status : Valid
Issue Date : 2018-11-21
Type : Chemical
Substance Number : 203

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Date of Issue : 2021-01-22
Valid Till : 2023-08-01
Written Confirmation Number : WC-0275
Address of the Firm : N-211/2/10, MIDC, Boisar, Dist-Thane-401506, Mahrashtra

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Dexamethasonum; Neomycinum; Polymyxini B sulfas
Brand Name : Maxitrol
Dosage Form : Gt Opht
Dosage Strength :
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Dexamethasonum; Neomycinum; Polymyxini B sulfas
Brand Name : Maxitrol
Dosage Form : Eye Gel
Dosage Strength :
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Polymyxini B sulfas; Neomycinum
Brand Name : Spersapolymyxin
Dosage Form : Gtt Opht
Dosage Strength :
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : Turkey
Dexamethasone; Framycetin; Phenylephrine; Polymyxin B
Brand Name :
Dosage Form : NASAL SPRAY
Dosage Strength : 0.25MG; 10MG; 2.5MG; 10.000IU
Packaging : 15 Ml/Bottle
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : Turkey

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : Turkey
Dexamethasone; Neomycin Sulfate; Polymyxin B
Brand Name :
Dosage Form : EYE AND EAR DROPS
Dosage Strength : 1MG; 3.5MG; 6.000IU
Packaging : 5 Ml/Bottle
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : Turkey

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Neomycinum; Polymyxini B sulfas; Fludrocortisoni acetas; Lidocaini hydrochloridum monohydricum
Brand Name : Panotile
Dosage Form : Tropfen
Dosage Strength :
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

Regulatory Info :
Registration Country : Italy
Hydrocortisone; Polymyxin B; Neomycin Sulfate; Lidocaine
Brand Name : Mixotone
Dosage Form :
Dosage Strength : Gtt Oto 1Vial+ 1Vial Solv 10 Ml
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
