

Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
API
0
FDF
0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
EDQM
0
USP
0
JP
0
Others
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
US Medicaid
NA
Annual Reports
NA
Finished Drug Prices
NA
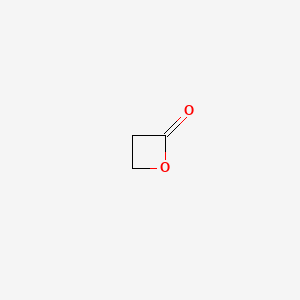

1. Beta Propiolactone
2. Propanolide
3. Propiolactone
1. 2-oxetanone
2. Propiolactone
3. 57-57-8
4. Oxetan-2-one
5. Betaprone
6. Propanolide
7. 3-propanolide
8. 1,3-propiolactone
9. 3-propiolactone
10. Beta-propionolactone
11. .beta.-propiolactone
12. Beta-propiolakton
13. Hydracrylic Acid Beta-lactone
14. 3-hydroxypropionic Acid Lactone
15. Propiolactona
16. Propiolactonum
17. Beta-propanoic Acid Lactone
18. Betapron
19. Nsc-21626
20. .beta.-proprolactone
21. Bpl
22. .beta.-propionolactone
23. Propiolactone .beta.-
24. 3-hydroxypropionic Acid Beta-lactone
25. 6rc3zt4hb0
26. Beta-lactone Hydracrylic Acid
27. .beta.-propanoic Acid Lactone
28. Hydracrylic Acid .beta.-lactone
29. 3-hydroxypropionic Acid, Lactone
30. Chebi:49073
31. Nsc21626
32. Ncgc00090802-01
33. Propiolattone
34. B-propiolactone
35. Beta-proprolactone
36. Caswell No. 709
37. Propiolattone [dcit]
38. Propionolactone
39. Oxetanone
40. Beta-propiolakton [czech]
41. Propiolactonum [inn-latin]
42. Nsc 21626
43. Propiolactona [inn-spanish]
44. Ccris 536
45. Hsdb 811
46. Propiolactone, Beta-
47. Propiolactone [usan:inn:ban]
48. Einecs 200-340-1
49. Unii-6rc3zt4hb0
50. Epa Pesticide Chemical Code 010901
51. Brn 0001360
52. Propanoic Acid, 3-hydroxy-, Beta-lactone
53. Propionic Acid, 3-hydroxy-, Beta-lactone
54. Ai3-24257
55. Oxetan-2one
56. 2-oxooxetane
57. 2-oxacyclobutanone
58. Betaprone (tn)
59. .beta.-propiolakton
60. Mfcd00005169
61. .beta.-propriolactone
62. Spectrum_000216
63. Spectrum2_001982
64. Spectrum4_001274
65. Wln: T4ovtj
66. Dsstox_cid_1197
67. Ec 200-340-1
68. Hydracrylic Acid Ss-lactone
69. Propiolactone [inn]
70. Propiolactone (usan/inn)
71. Dsstox_rid_76006
72. Propiolactone [hsdb]
73. Propiolactone [usan]
74. Dsstox_gsid_21197
75. Kbiogr_001888
76. Kbioss_000696
77. 5-17-09-00003 (beilstein Handbook Reference)
78. Divk1c_001003
79. Propiolactone [mart.]
80. Spectrum1503234
81. Spbio_002163
82. Hydracylic Acid, .beta.lactone
83. .beta.-lactone Hydracrylic Acid
84. Propanoic Acid, .beta.-lactone
85. Propionic Acid, .beta.-lactone
86. Chembl1200627
87. Dtxsid8021197
88. Hms503i07
89. Kbio1_001003
90. Kbio2_000696
91. Kbio2_003264
92. Kbio2_005832
93. Beta-propiolactone [iarc]
94. Hydracrylic Acid, .beta.-lactone
95. Beta-hydroxypropionic Acid Lactone
96. Ninds_001003
97. Hms1922k19
98. Hms2092p12
99. Pharmakon1600-01503234
100. .beta.-propiolactone [mi]
101. Propiolactone [orange Book]
102. Bcp07366
103. Zinc1482154
104. Tox21_111026
105. Ccg-39865
106. Nsc758422
107. Akos015906936
108. (2s)-2-amino-3-hydroxy-propanoicacid
109. 3-hydroxypropionic Acid .beta.-lactone
110. At19319
111. Db09348
112. Nsc-758422
113. Beta-propiolactone, Grade Ii, >=90%
114. Cas-57-57-8
115. Idi1_001003
116. Ncgc00090802-02
117. Ncgc00090802-03
118. Bp-31191
119. Sbi-0051797.p002
120. Db-003748
121. Am20020040
122. Ft-0622937
123. H0168
124. Propanoic Acid, 3-hydroxy-, .beta.-lactone
125. Propionic Acid, 3-hydroxy-, .beta.-lactone
126. C19297
127. D05630
128. Ab00052333_02
129. A936942
130. Q420715
131. Sr-05000001970
132. Sr-05000001970-1
133. 25037-58-5
| Molecular Weight | 72.06 g/mol |
|---|---|
| Molecular Formula | C3H4O2 |
| XLogP3 | -0.2 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | 72.021129366 g/mol |
| Monoisotopic Mass | 72.021129366 g/mol |
| Topological Polar Surface Area | 26.3 Ų |
| Heavy Atom Count | 5 |
| Formal Charge | 0 |
| Complexity | 57.9 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Because production of skin cancer is felt to be the overriding consideration in the toxic potential of BPL, all contact with liquid BPL should be avoided.
American Conference of Governmental Industrial Hygienists. Documentation of the Threshold Limit Values and Biological Exposure Indices. 5th ed. Cincinnati, OH: American Conference of Governmental Industrial Hygienists, 1986., p. 497
Propiolactone was used for vaccines, tissue grafts, surgical instruments, and enzymes, as a sterilant of blood plasma, water, milk and nutrient broth as a vapor-phase disinfectant in enclosed spaces. Its sporicidal action is used against vegetative bacteria, pathologic fungi, and viruses. It is no longer used in medical procedures or in food.
When employed under conditions of maximum effectiveness, propiolactone is approximately 25 more active as a vapor phase disinfectant than formaldehyde, 4000 times more active than ethylene oxide and 50000 times more active than methyl bromide. It has been shown to be mutagenic by inducing cell transformation, chromosomal aberrations and chromatoid exchange. Propiolactone has been shown to be mutagenic in both somatic and germ cells.
Disinfectants
Substances used on inanimate objects that destroy harmful microorganisms or inhibit their activity. Disinfectants are classed as complete, destroying SPORES as well as vegetative forms of microorganisms, or incomplete, destroying only vegetative forms of the organisms. They are distinguished from ANTISEPTICS, which are local anti-infective agents used on humans and other animals. (From Hawley's Condensed Chemical Dictionary, 11th ed) (See all compounds classified as Disinfectants.)
THE LD50 BY SKIN APPLICATION IS LESS THAN 5 ML/KG IN THE GUINEA PIG, INDICATING CONSIDERABLE ABSORPTION.
Patty, F. (ed.). Industrial Hygiene and Toxicology: Volume II: Toxicology. 2nd ed. New York: Interscience Publishers, 1963., p. 1834
BETA-PROPIOLACTONE BINDS IN VIVO TO DNA, RNA & PROTEINS OF MOUSE SKIN. DEGREE OF TUMOR-INITIATING ACTIVITY IS PROPORTIONAL TO EXTENT OF DNA BINDING BUT NOT TO EXTENT OF RNA OR PROTEIN-BINDING. MAJOR RNA & DNA BINDING PRODUCT IS 7-(2-CARBOXYETHYL)GUANINE. S-2-CARBOXYETHYLCYSTEINE ... FOUND IN ACID HYDROLYSATE OF PROTEIN ... .
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V4 265 (1974)
Propiolactone is completely hydrolyzed after 3 hours of being in an aqueous solution and this time can be even faster in the presence of cellular debris and cell culture media. When in water, the lactone ring opens at the alkyl and acyl bonds. The degradation products of propiolactone are not toxic.
BETA-PROPIOLACTONE CAN REACT WITH CHLORIDE ION TO FORM 3-CHLOROPROPIONIC ACID, ESPECIALLY IN BLOOD PLASMA.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V4 265 (1974)
BETA-HYDROXYPROPIONIC ACID, THE HYDROLYSIS PRODUCT OF BETA-PROPIOLACTONE, FAILED TO PRODUCE EITHER LOCAL SARCOMAS IN SC STUDY IN RATS ... OR SKIN TUMORS AFTER APPLICATIONS TO SKIN OF MICE.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V4 265 (1974)
The half-life of propiolactone in water is of 225 minutes.
No reports found; [TDR, p. 1048]
TDR - Ryan RP, Terry CE, Leffingwell SS (eds). Toxicology Desk Reference: The Toxic Exposure and Medical Monitoring Index, 5th Ed. Washington DC: Taylor & Francis, 1999., p. 1048
Propiolactone is an alkylating agent that acts through alkylation of carboxyl- and hydroxyl- groups. The lactone ring splits either at the first or third carbon. Propiolactone reacts with polynucleotides and DNA, mainly at N7 of guanine and N1 of adenine to form carboxyethyl derivatives. It also forms adducts with N3 of cytosine and thymine.
Chemical mutagens 6-N-hydroxylaminopurine and propiolactone induce Lys2 mutants with high frequency in diploid yeast Saccharomyces cerevisiae. 6-N-hydroxylaminopurine induces such mutants even in tetraploid strains. The genetic analysis of mutants was performed. It is known that propiolactone induces mutants by means of mutation-mitotic segregation mechanism, while 6-N-hydroxylaminopurine induces mutants through novel mechamism both allele mutation. Manifestation of such mechanism is the null fertility after meiosis of diploid mutants induced by 6-N-hydroxylaminopurine.
PMID:3069577 Pavlov IuI et al; Genetika 24 (10): 1752-60 (1988)
Reactivity of beta-propiolactone, beta-butyrolactone and gamma-butyrolactone with guanosine, RNA, DNA and 4-(p-nitrobenzyl)pyridine was studied. beta-Propiolactone was 50 to 100 times more reactive with all the nucleophiles than beta-butyrolactone whereas gamma-butyrolactone was completely inactive. The rate of alkylation by lactones was guanosine greater than RNA= denatured DNA greater than double-stranded DNA. The type of the adducts formed were characterized by fluorescence and ultraviolet spectroscopy. Similar alkylation products were formed by the two lactones. The main sites alkylated were N-1 at adenosine, N-3 at cytidine and N-7 at guanosine. The results suggest that the carcinogenic potency of the lactones correlates with their reactivity rather than with specificity of the adducts formed.
PMID:6161710 Hemminki K; Chem Biol Interact 34 (3): 323-31 (1981)
ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
