
Synopsis
Synopsis
0
JDMF
0
VMF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
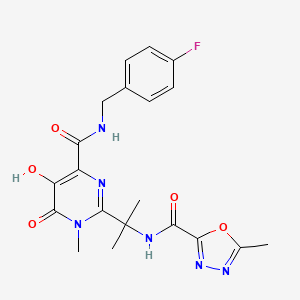

1. 0518, Mk
2. Isentress
3. Mk 0518
4. Mk-0518
5. Mk0518
6. Potassium, Raltegravir
7. Raltegravir Potassium
1. 518048-05-0
2. Mk-0518
3. Isentress
4. Raltegravir (mk-0518)
5. Mk0518
6. Raltegravir (inn)
7. Mk 0518
8. N-(2-(4-((4-fluorobenzyl)carbamoyl)-5-hydroxy-1-methyl-6-oxo-1,6-dihydropyrimidin-2-yl)propan-2-yl)-5-methyl-1,3,4-oxadiazole-2-carboxamide
9. 22vkv8053u
10. 518048-05-0 (free)
11. 4-pyrimidinecarboxamide, N-((4-fluorophenyl)methyl)-1,6-dihydro-5-hydroxy-1-methyl-2-(1-methyl-1-(((5-methyl-1,3,4-oxadiazol-2-yl)carbonyl)amino)ethyl)-6-oxo-
12. N-(2-(4-(4-fluorobenzylcarbamoyl)-5-hydroxy-1-methyl-6-oxo-1,6-dihydropyrimidin-2-yl)propan-2-yl)-5-methyl-1,3,4-oxadiazole-2-carboxamide
13. Ncgc00184997-01
14. Raltegravir [inn]
15. Dsstox_cid_28586
16. Dsstox_rid_82857
17. Dsstox_gsid_48660
18. N-[(4-fluorophenyl)methyl]-5-hydroxy-1-methyl-2-{2-[(5-methyl-1,3,4-oxadiazol-2-yl)formamido]propan-2-yl}-6-oxo-1,6-dihydropyrimidine-4-carboxamide
19. Isentress(tm)
20. N-[2-[4-[(4-fluorophenyl)methylcarbamoyl]-5-hydroxy-1-methyl-6-oxopyrimidin-2-yl]propan-2-yl]-5-methyl-1,3,4-oxadiazole-2-carboxamide
21. Cas-518048-05-0
22. Raltegravir [usan:inn]
23. Raltegravirum
24. Unii-22vkv8053u
25. Hsdb 8124
26. N-(4-fluorobenzyl)-5-hydroxy-1-methyl-2-(1-methyl-1-(((5-methyl-1,3,4-oxadiazol-2-yl)carbonyl)amino)ethyl)-6-oxo-1,6-dihydropyrimidine-4-carboxamide
27. N-(4-fluorobenzyl)-5-hydroxy-1-methyl-2-(1-methyl-1-{[(5-methyl-1,3,4-oxadiazol-2-yl)carbonyl]amino}ethyl)-6-oxo-1,6-di Hydropyrimidine-4-carboxamide
28. N-(4-fluorobenzyl)-5-hydroxy-1-methyl-2-(1-methyl-1-{[(5-methyl-1,3,4-oxadiazol-2-yl)carbonyl]amino}ethyl)-6-oxo-1,6-dihydropyrimidine-4-carboxamide
29. Rlt
30. Raltegravir- Bio-x
31. Raltegravir [mi]
32. N-[1-[4-[(4-fluorophenyl)methylcarbamoyl]-5-hydroxy-1-methyl-6-oxo-pyrimidin-2-yl]-1-methyl-ethyl]-5-methyl-1,3,4-oxadiazole-2-carboxamide
33. Raltegravir; Mk-0518
34. K-0518
35. Raltegravir [vandf]
36. Raltegravir - Mk-0518
37. Raltegravir [mart.]
38. Hydropyrimidine-4-carboxamide
39. Schembl51817
40. Raltegravir [who-dd]
41. Mls006011985
42. Schembl996804
43. Raltegravir [ema Epar]
44. Chembl254316
45. Schembl2112870
46. Dtxsid2048660
47. Bdbm25351
48. Chebi:82960
49. Gtpl11571
50. Bcpp000093
51. Hms3655b09
52. Bcp01394
53. Ex-a2147
54. Tox21_113019
55. Mfcd10698872
56. Nsc762522
57. S2005
58. Zinc13831130
59. Akos015902444
60. Akos025149884
61. Akos032960305
62. Tox21_113019_1
63. Zinc114994525
64. Ac-5261
65. Ccg-269170
66. Db06817
67. Nsc-762522
68. Pb13312
69. Sb20935
70. Ncgc00274066-01
71. Ncgc00274066-05
72. As-16992
73. Br164312
74. Hy-10353
75. Smr003601806
76. Ft-0649660
77. Sw220138-1
78. A25486
79. D06676
80. Mk-0518;mk 0518;mk0518
81. Yl}-6-oxo-1,6-dihydropyrimidine-4-carboxamide
82. Ab01566833_01
83. 038r721
84. 048r050
85. A828788
86. Q421552
87. Brd-k05658747-237-01-1
88. [(5-methyl-1,3,4-oxadiazol-2-yl)formamido]propan-2-
89. N-[(4-fluorophenyl)methyl]-5-hydroxy-1-methyl-2-{2-
90. Z1551429745
91. (z)-n-(2-(4-(((4-fluorophenyl)(methyl)amino)(hydroxy)methylene)-1-methyl-5,6-dioxo-1,4,5,6-tetrahydropyrimidin-2-yl)propan-2-yl)-5-methyl-1,3,4-oxadiazole-2-carboxamide
92. Mk-0518;n-[(4-fluorophenyl)methyl]-1,6-dihydro-5-hydroxy-1-methyl-2-[1-methyl-1-[[(5-methyl-1,3,4-oxadiazol-2-yl)carbonyl]amino]ethyl]-6-oxo-4-pyrimidinecarboxamide Potassium Salt
93. N-(2-(4-(4-fluorobenzylcarbamoyl)-5-hydroxy-1-methyl-6-oxo-1, 6-dihydropyrimidin-2-yl)propan-2-yl) -5-methyl-1,3,4-oxadiazole-2- Carboxamide
94. N-(2-(4-(4-fluorobenzylcarbamoyl)-5-hydroxy-1-methyl-6-oxo-1,6- Dihydropyrimidin-2-yl)propan-2-yl)-5-methyl-1,3,4-oxadiazole-2-carboxamide
95. N-(2-(4-(4-fluorobenzylcarbamoyl)-5-oh-1-me-6-oxo-pyrimidin-2-yl)propan-2-yl)-5-me-1,3,4-oxadiazole-2-carboxamide
96. N-(4-fluorobenzyl)-5-hydroxy-1-methyl-2-(1-methyl-1-{[(5-methyl-1,3,4-oxadiazol-2-yl)carbonyl]amino}ethyl)-6-oxo-1,6-di
97. N-(4-fluorobenzyl)-5-hydroxy-1-methyl-2-(2-{[(5-methyl-1,3,4-oxadiazol-2-yl)carbonyl]amino}propan-2-yl)-6-oxo-1,6-dihydropyrimidine-4-carboxamide
98. N-[2-(4-{[(4-fluorophenyl)methyl]carbamoyl}-5-hydroxy-1-methyl-6-oxo-1,6-dihydropyrimidin-2-yl)propan-2-yl]-5-methyl-1,3,4-oxadiazole-2-carboxamide
| Molecular Weight | 444.4 g/mol |
|---|---|
| Molecular Formula | C20H21FN6O5 |
| XLogP3 | 1.1 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 6 |
| Exact Mass | 444.15574595 g/mol |
| Monoisotopic Mass | 444.15574595 g/mol |
| Topological Polar Surface Area | 150 Ų |
| Heavy Atom Count | 32 |
| Formal Charge | 0 |
| Complexity | 836 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 4 | |
|---|---|
| Drug Name | Isentress |
| PubMed Health | Raltegravir (By mouth) |
| Drug Classes | Antiretroviral Agent |
| Drug Label | ISENTRESS contains raltegravir potassium, a human immunodeficiency virus integrase strand transfer inhibitor. The chemical name for raltegravir potassium is N-[(4-Fluorophenyl)methyl]-1,6-dihydro-5-hydroxy-1-methyl-2-[1-methyl-1-[[(5-methyl-1,3,4-oxa... |
| Active Ingredient | Raltegravir potassium |
| Dosage Form | Tablet; Powder; Tablet, chewable |
| Route | Oral |
| Strength | eq 100mg base; eq 400mg base; eq 100mg base/packet; eq 25mg base |
| Market Status | Prescription |
| Company | Merck Sharp Dohme |
| 2 of 4 | |
|---|---|
| Drug Name | Raltegravir |
| PubMed Health | Raltegravir (By mouth) |
| Drug Classes | Antiretroviral Agent |
| Drug Label | ISENTRESS contains raltegravir potassium, a human immunodeficiency virus integrase strand transfer inhibitor. The chemical name for raltegravir potassium is N-[(4-Fluorophenyl)methyl]-1,6-dihydro-5-hydroxy-1-methyl-2-[1-methyl-1-[[(5-methyl-1,3,4-oxa... |
| Active Ingredient | Raltegravir |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 400mg |
| Market Status | Tentative Approval |
| Company | Hetero Labs Ltd Iii |
| 3 of 4 | |
|---|---|
| Drug Name | Isentress |
| PubMed Health | Raltegravir (By mouth) |
| Drug Classes | Antiretroviral Agent |
| Drug Label | ISENTRESS contains raltegravir potassium, a human immunodeficiency virus integrase strand transfer inhibitor. The chemical name for raltegravir potassium is N-[(4-Fluorophenyl)methyl]-1,6-dihydro-5-hydroxy-1-methyl-2-[1-methyl-1-[[(5-methyl-1,3,4-oxa... |
| Active Ingredient | Raltegravir potassium |
| Dosage Form | Tablet; Powder; Tablet, chewable |
| Route | Oral |
| Strength | eq 100mg base; eq 400mg base; eq 100mg base/packet; eq 25mg base |
| Market Status | Prescription |
| Company | Merck Sharp Dohme |
| 4 of 4 | |
|---|---|
| Drug Name | Raltegravir |
| PubMed Health | Raltegravir (By mouth) |
| Drug Classes | Antiretroviral Agent |
| Drug Label | ISENTRESS contains raltegravir potassium, a human immunodeficiency virus integrase strand transfer inhibitor. The chemical name for raltegravir potassium is N-[(4-Fluorophenyl)methyl]-1,6-dihydro-5-hydroxy-1-methyl-2-[1-methyl-1-[[(5-methyl-1,3,4-oxa... |
| Active Ingredient | Raltegravir |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 400mg |
| Market Status | Tentative Approval |
| Company | Hetero Labs Ltd Iii |
Pyrrolidinones
National Library of Medicine's Medical Subject Headings online file (MeSH, 2009)
ISENTRESS (raltegravir) is indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus (HIV-1) infection. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information for ISENTRESS (raltegravir) tablet, film coated; tablet, chewable (Janurary 2013). Available from, as of March 14, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=89a5ec53-d956-4329-8004-0f40f51c88a3
ISENTRESS (raltegravir) is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in children and adolescents 2 years of age and older and weighing at least 10 kg. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information for ISENTRESS (raltegravir) tablet, film coated; tablet, chewable (Janurary 2013). Available from, as of March 14, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=89a5ec53-d956-4329-8004-0f40f51c88a3
/EXPERIMENTAL THER/ /Investigators/ describe five patients with HIV-2 infection (four antiretroviral-experienced and one antiretroviral-naive) treated with a regimen containing raltegravir. All responded to treatment as demonstrated by viral load and CD4(+) T-cell count monitoring. /This/ series confirms the clinical effectiveness of raltegravir in HIV-2-infected patients when given with other antiretrovirals to which the virus is susceptible.
PMID:22892365 Peterson K et al; Antivir Ther.17 (6):1097-100 (2012)
/EXPERIMENTALTHER VET/ Feline leukemia virus (FeLV) is a gammaretrovirus that is a significant cause of neoplastic-related disorders affecting cats worldwide. Treatment options for FeLV are limited, associated with serious side effects, and can be cost-prohibitive. The development of drugs used to treat a related retrovirus, human immunodeficiency virus type 1 (HIV-1), has been rapid, leading to the approval of five drug classes. Although structural differences affect the susceptibility of gammaretroviruses to anti-HIV drugs, the similarities in mechanism of replication suggest that some anti-HIV-1 drugs may also inhibit FeLV. This study demonstrates the anti-FeLV activity of four drugs approved by the US FDA (Food and Drug Administration) at non-toxic concentrations. Of these, tenofovir and raltegravir are anti-HIV-1 drugs, while decitabine and gemcitabine are approved to treat myelodysplastic syndromes and pancreatic cancer, respectively, but also have anti-HIV-1 activity in cell culture. Our results indicate that these drugs may be useful for FeLV treatment and should be investigated for mechanism of action and suitability for veterinary use.
PMID:22258856 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3542715 Greggs WM 3rd et al; J Gen Virol. 93 (Pt 4): 900-5 (2012)
Severe, potentially life-threatening skin reactions have been reported, including some fatalities. Stevens-Johnson syndrome, toxic epidermal necrolysis, and hypersensitivity reactions characterized by rash, constitutional findings, and organ dysfunction (including hepatic failure) have occurred.
American Society of Health-System Pharmacists 2012; Drug Information 2012. Bethesda, MD. 2012
Raltegravir and any other suspect agents should be discontinued immediately if signs or symptoms of severe skin or hypersensitivity reactions occur, including (but not limited to) severe rash or rash accompanied by fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, facial edema, hepatitis, eosinophilia, or angioedema. Clinical status, including liver aminotransferases, should be monitored and appropriate therapy initiated. A life-threatening reaction could occur if there is a delay in discontinuing raltegravir or other suspect agents after the onset of severe rash.
American Society of Health-System Pharmacists 2012; Drug Information 2012. Bethesda, MD. 2012
During initial treatment, patients who respond to antiretroviral therapy may develop an inflammatory response to indolent or residual opportunistic infections (e.g., Mycobacterium avium, M. tuberculosis, cytomegalovirus (CMV), Pneumocystis jiroveci (formerly P. carinii), varicella-zoster virus (VZV)); this may necessitate further evaluation and treatment. Autoimmune disorders (e.g., Graves' disease, polymyositis, Guillain-Barre syndrome) have been reported to occur in the setting of immune reconstitution; the time to onset is more variable and can occur many months after initiation of antiretroviral therapy.
American Society of Health-System Pharmacists 2012; Drug Information 2012. Bethesda, MD. 2012, p. 691
Individuals with phenylketonuria (i.e., homozygous genetic deficiency of phenylalanine hydroxylase) and other individuals who must restrict their intake of phenylalanine should be advised that raltegravir chewable tablets contain aspartame (NutraSweet), which is metabolized in the GI tract to phenylalanine. Each 25-mg chewable tablet provides approximately 0.05 mg of phenylalanine and each 100-mg chewable tablet provides approximately 0.1 mg of phenylalanine.
American Society of Health-System Pharmacists 2012; Drug Information 2012. Bethesda, MD. 2012
For more Drug Warnings (Complete) data for Raltegravir (12 total), please visit the HSDB record page.
For the treatment of HIV-1 infection in conjunction with other antiretrovirals.
FDA Label
Isentress is indicated in combination with other anti-retroviral medicinal products for the treatment of human immunodeficiency virus (HIV 1) infection.
Treatment of human immunodeficiency virus (HIV-1) infection
Anti-HIV Agents
Agents used to treat AIDS and/or stop the spread of the HIV infection. These do not include drugs used to treat symptoms or opportunistic infections associated with AIDS. (See all compounds classified as Anti-HIV Agents.)
HIV Integrase Inhibitors
Inhibitors of HIV INTEGRASE, an enzyme required for integration of viral DNA into cellular DNA. (See all compounds classified as HIV Integrase Inhibitors.)
J05AJ01
J05AX08
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
J - Antiinfectives for systemic use
J05 - Antivirals for systemic use
J05A - Direct acting antivirals
J05AJ - Integrase inhibitors
J05AJ01 - Raltegravir
Absorption
Absorbed from the gastrointestinal tract.
Route of Elimination
Feces and urine
Volume of Distribution
Approximately 83% bound to human plasma protein and is minimally distributed into red blood cells (blood-to-plasma partitioning ratio of 0.6).
Clearance
The major mechanism of clearance of raltegravir in humans is glucuronidation mediated by UGT1A1, the renal clearance of unchanged drug is a minor pathway of elimination of raltegravir (9% of total dose).
Raltegravir (film-coated tablet) is absorbed with a Tmax of approximately 3 hours postdose in the fasted state. Raltegravir AUC and Cmax increase dose proportionally over the dose range 100 mg to 1600 mg. Raltegravir C12hr increases dose proportionally over the dose range of 100 to 800 mg and increases slightly less than dose proportionally over the dose range 100 mg to 1600 mg.
US Natl Inst Health; DailyMed. Current Medication Information for ISENTRESS (raltegravir) tablet, film coated; tablet, chewable (Janurary 2013). Available from, as of March 14, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=89a5ec53-d956-4329-8004-0f40f51c88a3
With twice-daily dosing, pharmacokinetic steady state is achieved within approximately the first 2 days of dosing. There is little to no accumulation in AUC and Cmax. The average accumulation ratio for C12hr ranged from approximately 1.2 to 1.6.
US Natl Inst Health; DailyMed. Current Medication Information for ISENTRESS (raltegravir) tablet, film coated; tablet, chewable (Janurary 2013). Available from, as of March 14, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=89a5ec53-d956-4329-8004-0f40f51c88a3
The absolute bioavailability of raltegravir has not been established. Based on a formulation comparison study in healthy adult volunteers, the chewable tablet has higher oral bioavailability compared to the 400 mg film-coated tablet.
US Natl Inst Health; DailyMed. Current Medication Information for ISENTRESS (raltegravir) tablet, film coated; tablet, chewable (Janurary 2013). Available from, as of March 14, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=89a5ec53-d956-4329-8004-0f40f51c88a3
In subjects who received 400 mg twice daily alone, raltegravir drug exposures were characterized by a geometric mean AUC0-12hr of 14.3 uM.hr and C12hr of 142 nM.
US Natl Inst Health; DailyMed. Current Medication Information for ISENTRESS (raltegravir) tablet, film coated; tablet, chewable (Janurary 2013). Available from, as of March 14, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=89a5ec53-d956-4329-8004-0f40f51c88a3
For more Absorption, Distribution and Excretion (Complete) data for Raltegravir (13 total), please visit the HSDB record page.
Hepatic (UGT1A1)
In feces, only raltegravir was present, most of which is likely derived from hydrolysis of raltegravir-glucuronide secreted in bile as observed in preclinical species. Two components, namely raltegravir and raltegravir-glucuronide, were detected in urine and accounted for approximately 9 and 23% of the dose, respectively. The major circulating entity was raltegravir and represented approximately 70% of the total radioactivity; the remaining radioactivity in plasma was accounted for by raltegravir-glucuronide.
US Natl Inst Health; DailyMed. Current Medication Information for ISENTRESS (raltegravir) tablet, film coated; tablet, chewable (Janurary 2013). Available from, as of March 14, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=89a5ec53-d956-4329-8004-0f40f51c88a3
Studies using isoform-selective chemical inhibitors and cDNA-expressed UDP-glucuronosyltransferases (UGT) show that UGT1A1 is the main enzyme responsible for the formation of raltegravir-glucuronide. Thus, the data indicate that the major mechanism of clearance of raltegravir in humans is UGT1A1-mediated glucuronidation.
US Natl Inst Health; DailyMed. Current Medication Information for ISENTRESS (raltegravir) tablet, film coated; tablet, chewable (Janurary 2013). Available from, as of March 14, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=89a5ec53-d956-4329-8004-0f40f51c88a3
9 hours
The apparent terminal half-life of raltegravir is approximately 9 hours, with a shorter alpha-phase half-life (approximetly1 hour) accounting for much of the AUC.
US Natl Inst Health; DailyMed. Current Medication Information for ISENTRESS (raltegravir) tablet, film coated; tablet, chewable (Janurary 2013). Available from, as of March 14, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=89a5ec53-d956-4329-8004-0f40f51c88a3
Raltegravir inhibits HIV integrase to prevent the viral genome being incorporated into the human genome. Raltegravir is primarily metabolized by glucuronidation.
Raltegravir inhibits the catalytic activity of HIV-1 integrase, an HIV-1 encoded enzyme that is required for viral replication. Inhibition of integrase prevents the covalent insertion, or integration, of unintegrated linear HIV-1 DNA into the host cell genome preventing the formation of the HIV-1 provirus. The provirus is required to direct the production of progeny virus, so inhibiting integration prevents propagation of the viral infection. Raltegravir did not significantly inhibit human phosphoryltransferases including DNA polymerases alpha, beta, and gamma.
US Natl Inst Health; DailyMed. Current Medication Information for ISENTRESS (raltegravir) tablet, film coated; tablet, chewable (Janurary 2013). Available from, as of March 14, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=89a5ec53-d956-4329-8004-0f40f51c88a3
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
About the Company : LGM Pharma is a global leader in sourcing hard-to-find APIs and intermediates for the pharmaceutical and biotech industries. LGM is also a full service CDMO providing formulation, ...
About the Company : HRV Global is a leading global manufacturer, seller & exporter of a wide range of APIs, advanced intermediates, pellets, food grade chemicals, food additives & food ingredients. It...
About the Company : Since 2004, Arene Lifesciences Private Limited is committed to a cause to deliver the best and to aim for continuous improvement in order to transform new knowledge into innovative...

About the Company : Lee helps you focus on the business results that matter to your company. Over the years, we have leveraged our position in the global market with an enviable list of clients and a ...

About the Company : Maithri Drugs is one of India's fast-growing pharmaceutical companies. Maithri's strategic focus is on active pharma ingredients (APIs). The company is widely recognized for its ex...

About the Company : Prajna is an innovation driven pharmaceutical company. We dedicated to Research, Process development and Manufacturing APIs and Advanced Intermediates for Active Pharmaceutical Ing...

About the Company : SAINTSUN PHARMA, we are engaging mainly in the manufacturing and distributing APIs, Pharmaceutical Intermediates, Chemicals, Formulations, Medical supplies, etc... With a self-owne...

About the Company : Founded in 1995, Shenzhen Oriental Pharma focussed on the production of Active Pharma Ingredients (APIs), Now the main product is Phenylephrine Hcl (PE), Doxylamine succinate.We ha...

About the Company : SMS Pharmaceuticals Ltd. is a global player in API manufacturing having a strong research and manufacturing team supported by state of the art facilities. What started off as a sin...

About the Company : In November 2020, Viatris was formed through the combination of Mylan and Upjohn, with a mission of empowering people worldwide to live healthier at every stage of life. Viatris (N...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?