
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
FDF
0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
EDQM
0
USP
0
JP
0
Others
0
Weekly News Recap #Phispers
US Medicaid
NA
Finished Drug Prices
NA
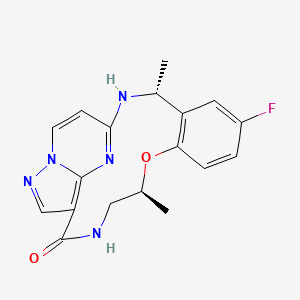

1. (1z)-6-fluoro-3,11-dimethyl-10-oxa-2,13,17,18,21-pentaazatetracyclo(13.5.2.04,9.018,22)docosa-1,4,6,8,15,19,21-heptaen-14-one
2. 1,15-etheno-1h-pyrazolo(4,3-f)(1,4,8,10)benzoxatriazacyclotridecin-4(5h)-one, 11-fluoro-2,6,7,13-tetrahydro-7,13-dimethyl-, (14z)-
1. Tpx-0005
2. 1802220-02-5
3. Ropotrectinib
4. 08o3fq4unp
5. Repotrectinib [usan]
6. Tpx0005
7. (3r,11s)-6-fluoro-3,11-dimethyl-10-oxa-2,13,17,18,21-pentazatetracyclo[13.5.2.04,9.018,22]docosa-1(21),4(9),5,7,15(22),16,19-heptaen-14-one
8. Trx-0005
9. Repotrectinib (usan)
10. 1,15-etheno-1h-pyrazolo(4,3-f)(1,4,8,10)benzoxatriazacyclotridecin-4(5h)-one, 11-fluoro-6,7,13,14-tetrahydro-7,13-dimethyl-, (7s,13r)-
11. Repotrectinib(tpx-005)
12. Unii-08o3fq4unp
13. Repotrectinib [inn]
14. Repotrectinib [who-dd]
15. Chembl4298138
16. Schembl16946804
17. Gtpl10316
18. Tpx 0005 [who-dd]
19. Bdbm374727
20. Ex-a2572
21. Tpx 0005
22. Us10246466, Example 93
23. Nsc800522
24. S8583
25. Akos037648789
26. Ccg-268096
27. Cs-7628
28. Nsc-800522
29. Ac-31283
30. Bs-15622
31. Example 93 [us20170334929a1]
32. Hy-103022
33. C73438
34. D11454
35. A910911
36. (13e,14e,3r,6s)-45-fluoro-3,6-dimethyl-5-oxa-2,8-diaza-1(5,3)-pyrazolo[1,5-a]pyrimidina-4(1,2)-benzenacyclononaphan-9-one
37. (13e,14e,3r,6s)-45-fluoro-3,6-dimethyl-5-oxa-2,8-diaza-1(5,3)pyrazolo[1,5-a]pyrimidina-4(1,2)-benzenacyclononaphan-9-one
38. (1z)-6-fluoro-3,11-dimethyl-10-oxa-2,13,17,18,21-pentaazatetracyclo(13.5.2.04,9.018,22)docosa-1,4,6,8,15,19,21-heptaen-14-one
39. (3r,11s)-6-fluoro-3,11-dimethyl-10-oxa-2,13,17,18,21-pentaazatetracyclo[13.5.2.04,9.018,22]docosa-1(21),4,6,8,15(22),16,19-heptaen-14-one
40. (3r,6s,)-45-fluoro-3,6-dimethyl-5-oxa-2,8-diaza-1(5,3)-pyrazolo(1,5-a)pyrimidina-4(1,2)-benzenanonaphan-9-one
41. (7s,13r)-11-fluoro-6,7,13,14-tetrahydro-7,13-dimethyl-1,15-etheno-1h-pyrazolo[4,3-f][1,4,8,10]benzoxatriazacyclotridecin-4(5h)-one
42. (7s,13r)-11-fluoro-7,13-dimethyl-6,7,13,14- Tetrahydro-1,15-ethenopyrazolo[4,3- F][1,4,8,10]benzoxatriazacyclotridecin-4(5h)- One
43. 1,15-etheno-1h-pyrazolo(4,3-f)(1,4,8,10)benzoxatriazacyclotridecin-4(5h)-one, 11-fluoro-2,6,7,13-tetrahydro-7,13-dimethyl-, (14z)-
44. 1,15-etheno-1h-pyrazolo[4,3-f][1,4,8,10]benzoxatriazacyclotridecin-4(5h)-one,11-fluoro-6,7,13,14-tetrahydro-7,13-dimethyl-, (7s,13r)-
| Molecular Weight | 355.4 g/mol |
|---|---|
| Molecular Formula | C18H18FN5O2 |
| XLogP3 | 2.1 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 0 |
| Exact Mass | 355.14445300 g/mol |
| Monoisotopic Mass | 355.14445300 g/mol |
| Topological Polar Surface Area | 80.6 Ų |
| Heavy Atom Count | 26 |
| Formal Charge | 0 |
| Complexity | 524 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Global Sales Information

Market Place

Reply
22 Jan 2024

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Patents & EXCLUSIVITIES
ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?