

Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
0
Canada

DRUG PRODUCT COMPOSITIONS
0
EDQM
0
USP
0
JP
0
Others
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
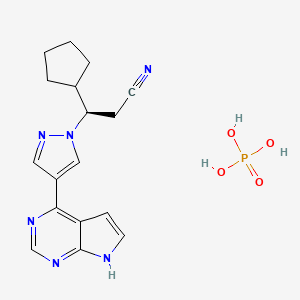

1. 3r)-3-cyclopentyl-3-(4-(7h-pyrrolo(2,3-d)pyrimidin-4-yl)pyrazol-1-yl)propanenitrile
2. Inc-424
3. Inc424
4. Inca24
5. Incb-018424
6. Incb-018424 Phosphate
7. Incb-018424 Salt
8. Incb-18424
9. Incb-18424 Phosphate
10. Incb018424
11. Incb018424 Phosphate
12. Jakafi
13. Jakavi
14. Opzelura
15. Ruxolitinib
16. Ruxolitinib (as Phosphate)
17. Ruxolitinib Monophosphate
1. 1092939-17-7
2. Jakafi
3. (r)-3-(4-(7h-pyrrolo[2,3-d]pyrimidin-4-yl)-1h-pyrazol-1-yl)-3-cyclopentylpropanenitrile Phosphate
4. Jakavi
5. Ruxolitinib (phosphate)
6. Ruxolitinib Monophosphate
7. Incb018424 Phosphate
8. Ruxolitinib (as Phosphate)
9. Incb-018424 Phosphate
10. Incb-018424 Salt
11. Ruxolitinib Phosphate [usan]
12. Incb-18424 Phosphate
13. Incb018424 Salt
14. Chebi:66917
15. 436lru32h5
16. 1092939-17-7 (phosphate)
17. (betar)-beta-cyclopentyl-4-(7h-pyrrolo[2,3-d]pyrimidin-4-yl)-1h-pyrazole-1-propanenitrile Phosphate
18. Incb-18424
19. Ruxolitinib Phosphate Salt
20. (3r)-3-cyclopentyl-3-[4-(7h-pyrrolo[2,3-d]pyrimidin-4-yl)-1h-pyrazol-1-yl]propanenitrile Phosphate
21. (3r)-3-cyclopentyl-3-(4-(7h-pyrrolo(2,3-d)pyrimidin-4-yl)-1h-pyrazol-1-yl)propanenitrile Phosphate (1:1)
22. Phosphenoperoxoic Acid Compound With (r)-3-(4-(7h-pyrrolo[2,3-d]pyrimidin-4-yl)- 1h-pyrazol-1-yl)-3-cyclopentylpropanenitrile And Dihydrogen (1:1:1)
23. Opzelura
24. Unii-436lru32h5
25. (3r)-3-cyclopentyl-3-[4-(7h-pyrrolo[2,3-d]pyrimidin-4-yl)-1h-pyrazol-1-yl]propanenitrilephosphoricacid
26. Jakafi (tn)
27. Jakavi (tn)
28. Ruxolitinib Phosphate(incb018424)
29. Inc 424 Phosphate
30. Incb 018424 Phosphate
31. Incb-424
32. Schembl1369365
33. Chembl1795071
34. Amy5620
35. Dtxsid00911086
36. Ruxolitinib Phosphate (jan/usan)
37. Ruxolitinib Phosphate [mi]
38. Ruxolitinib Phosphate [jan]
39. Ex-a2660
40. Cs1956
41. Mfcd18452860
42. S5243
43. Akos024464417
44. Ruxolitinib (incb-18424) Phosphate
45. Ruxolitinib Phosphate [who-dd]
46. Bcp9000783
47. Ccg-268687
48. Cs-0326
49. 1h-pyrazole-1-propanenitrile, Beta-cyclopentyl-4-(7h-pyrrolo(2,3-d)pyrimidin-4-yl)-,(betar)-, Phosphate (1:1)
50. Ac-30901
51. As-74723
52. Hy-50858
53. Ruxolitinib Phosphate [orange Book]
54. Ruxolitinib (as Phosphate) [ema Epar]
55. D09960
56. J-501793
57. Q27135517
58. (3r)-3-cyclopentyl-3-(4-{7h-pyrrolo[2,3-d]pyrimidin-4-yl}-1h-pyrazol-1-yl)propanenitrile; Phosphoric Acid
59. (3r)-3-cyclopentyl-3-[4-(7h-pyrrolo[2,3-d]pyrimidin-4-yl)pyrazol-1-yl]propanenitrile;phosphoric Acid
60. (r)-3-(4-(7h-pyrrolo[2,3-d]pyrimidin-4-yl)-1h-pyrazol-1-yl)-3-cyclopentylpropanenitrilephosphate
61. 1h-pyrazole-1-propanenitrile,.beta.-cyclopentyl-4-(7h-pyrrolo(2,3-d)pyrimidin-4-yl)-,(.beta.r)-,phosphate (1:1)
62. Phosphoric Acid--3-cyclopentyl-3-[4-(1h-pyrrolo[2,3-d]pyrimidin-4-yl)-1h-pyrazol-1-yl]propanenitrile (1/1)
| Molecular Weight | 404.4 g/mol |
|---|---|
| Molecular Formula | C17H21N6O4P |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 4 |
| Exact Mass | 404.13619017 g/mol |
| Monoisotopic Mass | 404.13619017 g/mol |
| Topological Polar Surface Area | 161 Ų |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 503 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Jakafi |
| PubMed Health | Ruxolitinib (By mouth) |
| Drug Classes | Antineoplastic Agent |
| Active Ingredient | Ruxolitinib phosphate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 5mg base; eq 20mg base; eq 15mg base; eq 10mg base; eq 25mg base |
| Market Status | Prescription |
| Company | Incyte |
| 2 of 2 | |
|---|---|
| Drug Name | Jakafi |
| PubMed Health | Ruxolitinib (By mouth) |
| Drug Classes | Antineoplastic Agent |
| Active Ingredient | Ruxolitinib phosphate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 5mg base; eq 20mg base; eq 15mg base; eq 10mg base; eq 25mg base |
| Market Status | Prescription |
| Company | Incyte |
* Myelofibrosis (MF):
Jakavi is indicated for the treatment of disease related splenomegaly or symptoms in adult patients with primary myelofibrosis (also known as chronic idiopathic myelofibrosis), post polycythaemia vera myelofibrosis or post essential thrombocythaemia myelofibrosis.
* Polycythaemia vera (PV):
Jakavi is indicated for the treatment of adult patients with polycythaemia vera who are resistant to or intolerant of hydroxyurea.
* Graft versus host disease (GvHD):
Jakavi is indicated for the treatment of patients aged 12 years and older with acute graft versus host disease or chronic graft versus host disease who have inadequate response to corticosteroids or other systemic therapies (see section 5. 1).
Treatment of chronic Graft versus Host Disease (cGvHD)
Treatment of acute graft-versus-host disease (aGvHD)
Treatment of vitiligo
L01EJ01
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39763
Submission : 2024-03-27
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39494
Submission : 2024-03-29
Status : Inactive
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2015-11-16
Pay. Date : 2015-07-31
DMF Number : 29480
Submission : 2015-05-29
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38506
Submission : 2023-07-13
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2021-12-16
Pay. Date : 2021-09-29
DMF Number : 32221
Submission : 2017-11-30
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39789
Submission : 2024-03-28
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39500
Submission : 2024-03-28
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38709
Submission : 2023-09-30
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Date of Issue : 2022-09-02
Valid Till : 2025-05-05
Written Confirmation Number : WC-0349
Address of the Firm : MIs. MSN Laboratories Private Limited, Unit-II, sv. No, 50, Kardanur (Village), ...

Date of Issue : 2019-08-05
Valid Till : 2022-05-05
Written Confirmation Number : WC-0349A3
Address of the Firm : Unit-||, Sy. No. 50, Kardanur Village, Patti/Post Patancheru Mandal,Medak Distri...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Regulatory Info :
Registration Country : Australia
Brand Name : Jakavi
Dosage Form :
Dosage Strength :
Packaging : 56
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Australia

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Australia
Brand Name : Jakavi
Dosage Form :
Dosage Strength :
Packaging : 56
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Australia

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Australia
Brand Name : Jakavi
Dosage Form :
Dosage Strength :
Packaging : 56
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Australia

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Australia
Brand Name : Jakavi
Dosage Form :
Dosage Strength :
Packaging : 56
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Australia

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Australia
Brand Name : Jakavi
Dosage Form :
Dosage Strength :
Packaging : 56
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Australia

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Australia
Brand Name : Jakavi
Dosage Form :
Dosage Strength :
Packaging : 56
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Australia

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Australia
Brand Name : Jakavi
Dosage Form :
Dosage Strength :
Packaging : 56
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Australia

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Australia
Brand Name : Jakavi
Dosage Form :
Dosage Strength :
Packaging : 56
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Australia

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Australia
Brand Name : Jakavi
Dosage Form :
Dosage Strength :
Packaging : 56
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Australia

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Australia
Brand Name : Jakavi
Dosage Form :
Dosage Strength :
Packaging : 56
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Australia

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Excipients by Applications
Global Sales Information

Main Therapeutic Indication : Oncology
Currency : USD
2020 Revenue in Millions : 1,339
2019 Revenue in Millions : 1,114
Growth (%) : 20

Main Therapeutic Indication : Oncology
Currency : USD
2021 Revenue in Millions : 1,595
2020 Revenue in Millions : 1,339
Growth (%) : 19

Main Therapeutic Indication : Hematology
Currency : USD
2022 Revenue in Millions : 1,561
2021 Revenue in Millions : 1,595
Growth (%) : -2

Main Therapeutic Indication : Oncology
Currency : USD
2023 Revenue in Millions : 1,720
2022 Revenue in Millions : 1,561
Growth (%) : 10

Main Therapeutic Indication : Oncology
Currency : USD
2014 Revenue in Millions : 71.20%
2013 Revenue in Millions :
Growth (%) :

Main Therapeutic Indication : Oncology
Currency : USD
2015 Revenue in Millions : 279
2014 Revenue in Millions : 410
Growth (%) : 47%

Main Therapeutic Indication : Oncology
Currency : USD
2016 Revenue in Millions : 581
2015 Revenue in Millions : 410
Growth (%) : 42

Main Therapeutic Indication : Oncology
Currency : USD
2017 Revenue in Millions : 777
2016 Revenue in Millions : 581
Growth (%) : 34

Main Therapeutic Indication : Oncology
Currency : USD
2018 Revenue in Millions : 977
2017 Revenue in Millions : 777
Growth (%) : 26%

Main Therapeutic Indication : Oncology
Currency : USD
2019 Revenue in Millions : 1,114
2018 Revenue in Millions : 977
Growth (%) : 14

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

Reply
27 Aug 2024
Reply
24 Aug 2024
Reply
05 Aug 2024
Reply
12 Jul 2024
Reply
22 Jun 2024
Reply
02 Oct 2023
Reply
12 Jul 2023
Reply
30 Jan 2023
Reply
15 Oct 2022
Reply
13 Aug 2022
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Reply
17 Aug 2023
Reply
31 Mar 2022
Reply
10 Dec 2018

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Patents & EXCLUSIVITIES
Patent Expiration Date : 2026-12-12
US Patent Number : 9079912
Drug Substance Claim :
Drug Product Claim :
Application Number : 202192
Patent Use Code : U-3230
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2026-12-12

Patent Expiration Date : 2028-06-12
US Patent Number : 8822481
Drug Substance Claim :
Drug Product Claim :
Application Number : 202192
Patent Use Code : U-1573
Delist Requested :
Patent Use Description : USE OF RUXOLITINIB (JA...
Patent Expiration Date : 2028-06-12

Patent Expiration Date : 2027-12-24
US Patent Number : 8415362
Drug Substance Claim : Y
Drug Product Claim : Y
Application Number : 215309
Patent Use Code :
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2027-12-24

Patent Expiration Date : 2028-06-12
US Patent Number : 8829013
Drug Substance Claim :
Drug Product Claim :
Application Number : 202192
Patent Use Code : U-3227
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2028-06-12

Patent Expiration Date : 2028-06-12
US Patent Number : 8822481
Drug Substance Claim :
Drug Product Claim :
Application Number : 215309
Patent Use Code : U-3229
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2028-06-12

Patent Expiration Date : 2026-12-12
US Patent Number : 9079912
Drug Substance Claim :
Drug Product Claim :
Application Number : 202192
Patent Use Code : U-3226
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2026-12-12

Patent Expiration Date : 2028-12-12
US Patent Number : 10016429*PED
Drug Substance Claim :
Drug Product Claim :
Application Number : 202192
Patent Use Code :
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2028-12-12

Patent Expiration Date : 2028-06-12
US Patent Number : 8822481
Drug Substance Claim :
Drug Product Claim :
Application Number : 202192
Patent Use Code : U-3228
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2028-06-12

Patent Expiration Date : 2026-12-12
US Patent Number : 9974790
Drug Substance Claim :
Drug Product Claim :
Application Number : 215309
Patent Use Code : U-3229
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2026-12-12

Patent Expiration Date : 2027-06-12
US Patent Number : 9079912*PED
Drug Substance Claim :
Drug Product Claim :
Application Number : 202192
Patent Use Code :
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2027-06-12

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Exclusivity Code : ODE-238
Exclusivity Expiration Date : 2026-05-24
Application Number : 202192
Product Number : 1
Exclusivity Details :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Exclusivity Code : I-872
Exclusivity Expiration Date : 2024-09-22
Application Number : 202192
Product Number : 1
Exclusivity Details :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Exclusivity Code : M-285
Exclusivity Expiration Date : 2025-12-19
Application Number : 202192
Product Number : 1
Exclusivity Details :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Exclusivity Code : ODE-373
Exclusivity Expiration Date : 2028-09-22
Application Number : 202192
Product Number : 1
Exclusivity Details :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Exclusivity Code : ODE-238
Exclusivity Expiration Date : 2026-05-24
Application Number : 202192
Product Number : 2
Exclusivity Details :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Exclusivity Code : I-872
Exclusivity Expiration Date : 2024-09-22
Application Number : 202192
Product Number : 2
Exclusivity Details :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Exclusivity Code : M-285
Exclusivity Expiration Date : 2025-12-19
Application Number : 202192
Product Number : 2
Exclusivity Details :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Exclusivity Code : ODE-373
Exclusivity Expiration Date : 2028-09-22
Application Number : 202192
Product Number : 2
Exclusivity Details :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Exclusivity Code : ODE-238
Exclusivity Expiration Date : 2026-05-24
Application Number : 202192
Product Number : 3
Exclusivity Details :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Exclusivity Code : M-285
Exclusivity Expiration Date : 2025-12-19
Application Number : 202192
Product Number : 3
Exclusivity Details :

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
