
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
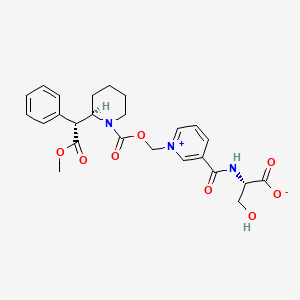

1. Kp-415 Inner Salt
2. 0h8kz470dw
3. Pyridinium, 3-((((1s)-1-carboxy-2-hydroxyethyl)amino)carbonyl)-1-(((((2r)-2-((1r)-2-methoxy-2-oxo-1-phenylethyl)-1-piperidinyl)carbonyl)oxy)methyl)-
4. Unii-0h8kz470dw
5. Gtpl11450
6. Kp484
7. Serdexmethylphenidate [inn]
8. Kp-484
9. Serdexmethylphenidate [who-dd]
10. Azstarys (serdexmethylphenidate + Dexmethylphenidate)
11. (2s)-3-hydroxy-2-[[1-[[(2r)-2-[(1r)-2-methoxy-2-oxo-1-phenylethyl]piperidine-1-carbonyl]oxymethyl]pyridin-1-ium-3-carbonyl]amino]propanoate
12. 1996626-29-9
13. 2014450-84-9
14. N-(1-((((2r)-2-((1r)-2-methoxy-2-oxo-1-phenylethyl)piperidine-1-carbonyl)oxy)methyl)pyridin-1-ium-3-carbonyl)-l-serinate
| Molecular Weight | 499.5 g/mol |
|---|---|
| Molecular Formula | C25H29N3O8 |
| XLogP3 | 2.2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 10 |
| Exact Mass | 499.19546489 g/mol |
| Monoisotopic Mass | 499.19546489 g/mol |
| Topological Polar Surface Area | 149 Ų |
| Heavy Atom Count | 36 |
| Formal Charge | 0 |
| Complexity | 767 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 3 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Serdexmethylphenidate is a prodrug of [dexmethylphenidate] that is indicated in combination with [dexmethylphenidate] for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in patients aged six years and older.
Serdexmethylphenidate is a prodrug of the CNS stimulant [dexmethylphenidate], which increases extracellular levels of dopamine and norepinephrine in the CNS, leading to altered neurotransmission. As a CNS stimulant, serdexmethylphenidate carries a risk of abuse, misuse, and dependence, which should be monitored. Also, CNS stimulants are associated with increased blood pressure, heart rate, and risk of serious cardiovascular reactions, including stroke, myocardial infarction, and sudden death; patients should be assessed before starting therapy and monitored for cardiovascular abnormalities. Similarly, CNS stimulants may also result in peripheral vasculopathy, including Raynaud's phenomenon. Due to its ability to alter neurological function, serdexmethylphenidate may exacerbate pre-existing psychoses, induce manic episodes in patients with bipolar disorder, or result in newly diagnosable manic or psychotic symptoms. Frequent, sustained, and painful erections, which may require medical attention, have been observed in patients who have been treated for some time with serdexmethylphenidate, often associated with a dose increase. Finally, like other CNS stimulants, serdexmethylphenidate has been associated with weight loss and growth retardation, which may require treatment interruption in serious cases.
Absorption
Following a single dose of serdexmethylphenidate/dexmethylphenidate (52.3/10.4 mg) compared to extended-release dexmethylphenidate (40 mg) capsules in healthy volunteers under fasted conditions, the Cmax and AUC of dexmethylphenidate were 14.0 ng/mL and 186 ng\*h/mL and 28.2 ng/mL and 248 ng\*h/mL, respectively. The kinetics are approximately linear over a range of concentrations, with steady-state being reached after the third once-daily dose. Serdexmethylphenidate has a low oral bioavailability of 3%. The Tmax for both serdexmethylphenidate and dexmethylphenidate is approximately two hours under fasted conditions when coadministered. When serdexmethylphenidate is administered as a single entity, the dexmethylphenidate Tmax is approximately eight hours. Different ratios of serdexmethylphenidate to dexmethylphenidate, 64/8, 56/12, and 48/16 mg, each equivalent to 40 mg of dexmethylphenidate, were tested in healthy adult volunteers under fasted conditions. In each case, dexmethylphenidate reached peak plasma concentrations in roughly two hours (mean between 1.6-1.8 hours), which gradually decreased over 24 hours. The Cmax varied from 15.5 3.7 to 23.8 5.7 ng/mL while the AUC0-24h varied from 187.0 41.0 to 207 54.4 ng\*h/mL. Another study investigated the pharmacokinetics of serdexmethylphenidate/dexmethylphenidate (28/6 or 56/12 mg) in patients aged between six and 17 years of age. In general, the Cmax and AUC varied between cohorts and dose but were roughly equivalent when normalized for both dose and body weight.
Route of Elimination
Following oral serdexmethylphenidate dosing in humans, roughly 62% and 37% of the initial dose was recovered in the urine and feces, of which about 0.4% and 11% of the initial dose was recovered unchanged, respectively. Ritalinic acid accounted for approximately 63% of the recovered dose. When methylphenidate was administered orally as a racemate, about 90% of the dose was recovered in urine, of which racemic acid accounted for approximately 80% of the dose.
Volume of Distribution
Serdexmethylphenidate has a mean apparent volume of distribution of 29.3 L/kg following serdexmethylphenidate/dexmethylphenidate administration. Serdexmethylphenidate/dexmethylphenidate (28/6 or 56/12 mg) administered orally in patients aged between six and 17 years of age produced an apparent volume of distribution of dexmethylphenidate of between 37.6 and 66 L/kg.
Clearance
Serdexmethylphenidate has a mean apparent clearance of about 3.6 L/h/kg following oral serdexmethylphenidate/dexmethylphenidate administration. In patients aged 6-17 years following oral administration of 28/6 or 56/12 mg serdexmethylphenidate/dexmethylphenidate, dexmethylphenidate has a mean apparent clearance of between 2.5 and 3.4 L/h/kg when normalized for dose.
Serdexmethylphenidate is converted to dexmethylphenidate in the lower gastrointestinal tract by as yet unknown enzymes. Following this, dexmethylphenidate is mainly converted to d--phenyl-piperidine acetic acid (d-ritalinic acid) in the liver by carboxylesterase 1A1. Other metabolites include the oxidation products 6-oxo-methylphenidate and p-hydroxy-methylphenidate, which are then de-esterified to oxo-ritalinic acid and p-hydroxy-ritalinic acid, respectively. Methylphenidate may also be trans-esterified to form ethylphenidate.
Following a single oral dose of 52.3 mg/10.4 mg serdexmethylphenidate/dexmethylphenidate, the mean plasma terminal elimination half-lives of each component were roughly 5.7 and 11.7 hours, respectively. At steady-state in healthy adults under fasted conditions, serdexmethylphenidate/dexmethylphenidate at 64/8, 56/12, and 48/16 mg resulted in a dexmethylphenidate half-life of between 8.5 2.3 and 9.2 3.5 hours.
Attention Deficit Hyperactivity Disorder (ADHD) is an early-onset neurodevelopmental disorder that often extends into adulthood and is characterized by developmentally inappropriate and impaired attention, impulsivity, and motor hyperactivity. Proper diagnosis is hindered by a lack of biological markers (based on symptoms alone), a spectrum of severity, and frequent comorbidities such as autism spectrum disorder, reading disabilities, developmental coordination disorders, and tic disorders. Although the underlying cause(s) is unclear, dopaminergic, noradrenergic, serotonergic, cholinergic, glutaminergic, and opioid neurotransmission likely plays a role. Serdexmethylphenidate is a prodrug of the CNS stimulant [dexmethylphenidate](MPH), a common first-line treatment for ADHD. The main effect of MPH is to increase the extracellular levels of dopamine and norepinephrine, which has numerous potential downstream effects. This occurs mainly due to MPH's ability to inhibit the corresponding dopamine and norepinephrine monoamine transporters. Other studies have suggested additional possible MPH functions, including serotonin 5-HT1A receptor agonism, redistribution of vesicular monoamine transporter-2 (VMAT-2), and either direct or indirect activation of 2-adrenergic receptors. Overall, imaging studies reveal that MPH acts to alter brain activity in relevant regions associated with executive function, emotional regulation, reward processing, and working memory.
ABOUT THIS PAGE
46
PharmaCompass offers a list of Serdexmethylphenidate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Serdexmethylphenidate manufacturer or Serdexmethylphenidate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Serdexmethylphenidate manufacturer or Serdexmethylphenidate supplier.
PharmaCompass also assists you with knowing the Serdexmethylphenidate API Price utilized in the formulation of products. Serdexmethylphenidate API Price is not always fixed or binding as the Serdexmethylphenidate Price is obtained through a variety of data sources. The Serdexmethylphenidate Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Serdexmethylphenidate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Serdexmethylphenidate, including repackagers and relabelers. The FDA regulates Serdexmethylphenidate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Serdexmethylphenidate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Serdexmethylphenidate supplier is an individual or a company that provides Serdexmethylphenidate active pharmaceutical ingredient (API) or Serdexmethylphenidate finished formulations upon request. The Serdexmethylphenidate suppliers may include Serdexmethylphenidate API manufacturers, exporters, distributors and traders.
click here to find a list of Serdexmethylphenidate suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Serdexmethylphenidate as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Serdexmethylphenidate API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Serdexmethylphenidate as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Serdexmethylphenidate and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Serdexmethylphenidate NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Serdexmethylphenidate suppliers with NDC on PharmaCompass.
Serdexmethylphenidate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Serdexmethylphenidate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Serdexmethylphenidate GMP manufacturer or Serdexmethylphenidate GMP API supplier for your needs.
A Serdexmethylphenidate CoA (Certificate of Analysis) is a formal document that attests to Serdexmethylphenidate's compliance with Serdexmethylphenidate specifications and serves as a tool for batch-level quality control.
Serdexmethylphenidate CoA mostly includes findings from lab analyses of a specific batch. For each Serdexmethylphenidate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Serdexmethylphenidate may be tested according to a variety of international standards, such as European Pharmacopoeia (Serdexmethylphenidate EP), Serdexmethylphenidate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Serdexmethylphenidate USP).
