
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
FDA Orange Book

0
Europe

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
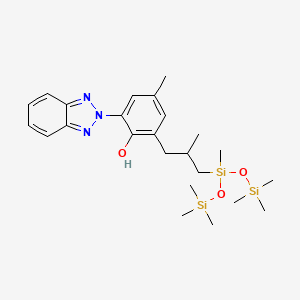

1. Mexoryl Xl
2. Silatrizole
1. 155633-54-8
2. Silatrizole
3. Mexoryl Xl
4. Drometrizole Trisiloxane [inci]
5. Drometrizole Trisiloxane [who-dd]
6. 2-(benzotriazol-2-yl)-4-methyl-6-[2-methyl-3-[methyl-bis(trimethylsilyloxy)silyl]propyl]phenol
7. Or 10154
8. Or-10154
9. Hc22845i1x
10. Phenol, 2-(2h-benzotriazol-2-yl)-4-methyl-6-(2-methyl-3-(1,3,3,3-tetramethyl-1-((trimethylsilyl)oxy)-1-disiloxanyl)propyl)-
11. Phenol, 2-(2h-benzotriazol-2-yl)-4-methyl-6-(2-methyl-3-(1,3,3,3-tetramethyl-1-((trimethylsilyl)oxy)disiloxanyl)propyl)-
12. 2-(2h-benzo[d][1,2,3]triazol-2-yl)-6-(3-(1,1,1,3,5,5,5-heptamethyltrisiloxan-3-yl)-2-methylpropyl)-4-methylphenol
13. Phenol, 2-(2h-benzotriazol-2-yl)-4-methyl-6-[2-methyl-3-[1,3,3,3-tetramethyl-1-[(trimethylsilyl)oxy]-1-disiloxanyl]propyl]-
14. Unii-hc22845i1x
15. Mexoryl Xl;silatrizole
16. Schembl15380
17. Amy224
18. Dtxsid40431753
19. Act12435
20. Bcp28866
21. Db11585
22. Drometrizole Trisiloxane [usp-rs]
23. Ncgc00532508-01
24. Db-091411
25. Drometrizole Trisiloxane, Analytical Standard
26. Q5308268
27. Drometrizole Trisiloxane, United States Pharmacopeia (usp) Reference Standard
28. 2-(2h-benzotriazol-2-yl)-4-methyl-6-(2-methyl-3-(1,3,3,3-tetramethyl-1-(trimethylsilyloxy)-disiloxanyl)propyl)phenol
29. 320341-73-9
| Molecular Weight | 501.8 g/mol |
|---|---|
| Molecular Formula | C24H39N3O3Si3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 9 |
| Exact Mass | 501.22992172 g/mol |
| Monoisotopic Mass | 501.22992172 g/mol |
| Topological Polar Surface Area | 69.4 Ų |
| Heavy Atom Count | 33 |
| Formal Charge | 0 |
| Complexity | 613 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Drometrizole trisiloxane is used as an active ingredient in various sunscreens for the indication of protecting the skin by absorbing the damaging UV radiation of sunlight.
As an active ingredient in sunscreen products, drometrizole trisiloxane is applied directly onto human skin where it acts as a chemical sunscreen layer between skin and sunlight that also directly absorbs the UV sunlight radiation. Since drometrizole trisiloxane is also considered to have little to no absorption through the skin, little systemic exposure and pharmacokinetics are expected and users can freely wash off and re-apply the compound as necessary.
Absorption
Drometrizole trisiloxane is reported as having little to no absorption through the skin. At this time, however, studies demonstrate that the components of most commonly used sunscreens are likely absorbed into the skin at least to some extent - although penetration to deeper tissues and the cutaneous circulation remains limited. Despite the extensive use of sunscreen products around the world, there have been few reports of adverse effects related to their use.
Route of Elimination
Drometrizole trisiloxane is reported as having little to no absorption through the skin. The systemic presence of the compound is consequently expected to be minimal.
Volume of Distribution
Drometrizole trisiloxane is reported as having little to no absorption through the skin. The systemic presence of the compound is consequently expected to be minimal.
Clearance
Drometrizole trisiloxane is reported as having little to no absorption through the skin. The systemic presence of the compound is consequently expected to be minimal.
Drometrizole trisiloxane is reported as having little to no absorption through the skin. The systemic presence of the compound is consequently expected to be minimal.
Drometrizole trisiloxane is reported as having little to no absorption through the skin. The systemic presence of the compound is consequently expected to be minimal.
Ultraviolet radiation is the invisible energy component to sunlight and consists of three wavelength ranges: (a) UVA is long-range UV radiation between 320-400nm. Although not as energetic as UVB, UVA can penetrate deep into the dermis. UVA can cause immediate tanning, premature skin aging, and can also play a role in the formation of some skin cancers. Approximately 95% of UVA from the sun passes through Earth's ozone layer. (b) UVB is short-wavelength UV radiation between 280-320nm. It is capable of penetrating the outer protective layer of the skin and is responsible for delayed tanning, sunburns, and most skin cancers. A large amount of UVB is absorbed by the ozone layer, however, as only 5% reaches the Earth's surface. (c) UVC is comprised of wavelengths between 100-280nm and is very energetic. It is very dangerous to all forms of life, even when the exposure is short. However, UVC radiation is generally filtered out by the ozone layer and never reaches the Earth. Ultimately, the shorter the wavelength, the more harmful the UV radiation - although shorter wavelength UV radiation is less able to penetrate the skin. Subsequently, drometrizole trisiloxane is a broad spectrum lipophilic benzotriazole derivative chemical sunscreen that is capable of absorbing UVA and UVB radiation. It is also photostable, meaning that it will not degrade in the presence of sunlight, unlike other UV filters like the widely used UVA absorber avobenzone. When combined with the UV blocker ecamsule, it has been shown that the two UV blockers elicit a synergistic effect involving an enhanced protective action for the skin against UVA and UVB radiation. Additionally, drometrizole trisiloxane is usually combined with other active sunscreen agents like titanium dioxide, avobenzone, and others to ensure the combined product covers or protects against as broad a spectrum of UV radiation as possible, considering drometrizole trisiloxane does not absorb against the entire range of UV radiation. And finally, at the molecular level, it is believed that the general structure of various UV blockers like drometrizole trisiloxane as aromatic molecules conjugated with carbonyl groups is capable of absorbing high energy ultraviolet rays and then consequently releasing that energy as less harmful, lower energy rays.
Related Excipient Companies

Excipients by Applications
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
100
PharmaCompass offers a list of Silatrizole API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Silatrizole manufacturer or Silatrizole supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Silatrizole manufacturer or Silatrizole supplier.
PharmaCompass also assists you with knowing the Silatrizole API Price utilized in the formulation of products. Silatrizole API Price is not always fixed or binding as the Silatrizole Price is obtained through a variety of data sources. The Silatrizole Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Silatrizole manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Silatrizole, including repackagers and relabelers. The FDA regulates Silatrizole manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Silatrizole API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Silatrizole supplier is an individual or a company that provides Silatrizole active pharmaceutical ingredient (API) or Silatrizole finished formulations upon request. The Silatrizole suppliers may include Silatrizole API manufacturers, exporters, distributors and traders.
Silatrizole Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Silatrizole GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Silatrizole GMP manufacturer or Silatrizole GMP API supplier for your needs.
A Silatrizole CoA (Certificate of Analysis) is a formal document that attests to Silatrizole's compliance with Silatrizole specifications and serves as a tool for batch-level quality control.
Silatrizole CoA mostly includes findings from lab analyses of a specific batch. For each Silatrizole CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Silatrizole may be tested according to a variety of international standards, such as European Pharmacopoeia (Silatrizole EP), Silatrizole JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Silatrizole USP).
