
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
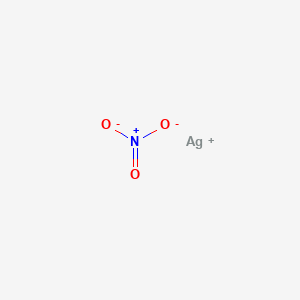

1. Alcoholic Silver Nitrate
2. Silver Nitrate, Alcoholic
3. Silver Nitrate, Silver (2+) Salt (2:1)
1. 7761-88-8
2. Silver(i) Nitrate
3. Silvernitrate
4. Argenti Nitras
5. Nitric Acid Silver(i) Salt
6. Nitrate D'argent
7. Silver(1+) Nitrate
8. Silver Mononitrate
9. Nitric Acid Silver(1+) Salt
10. Agno3
11. Nitrato De Plata
12. Argentum Nitricum
13. 95it3w8jze
14. Silbernitrat
15. Chebi:32130
16. Nsc-135800
17. Silver Nitrate Solution
18. Argerol
19. Nitric Acid Silver (1+) Salt
20. Silver Nitrate(dot)
21. Caswell No. 737
22. Silver Nitrate [jan]
23. Silver Nitrate Solution, 0.25n
24. Silver, Reference Standard Solution
25. Mfcd00003414
26. Nitrate D'argent [french]
27. Nitrato De Plata [spanish]
28. Hsdb 685
29. Uts Silverator Water Treatment Unit
30. Einecs 231-853-9
31. Un1493
32. Unii-95it3w8jze
33. Epa Pesticide Chemical Code 072503
34. Nitric Acid Silver(1+) Salt (1:1)
35. Nsc 135800
36. Silver Nitrate, Acs
37. Silver Nitrate [usp:jan]
38. Silver;nitrate
39. Silver Nitrate-
40. Nitrato De Prata
41. Silver(i)nitrate
42. Nitric Acid, Silver (1+) Salt
43. Nitrato D'argento
44. Silver Nitrate Bp
45. Silver Nitrate Ip
46. Silver Nitrate Usp
47. Silver (i) Nitrate
48. Silver (1) Nitrate
49. Silver (1+) Salt
50. Silver Nitrate (tn)
51. Silver (1+) Nitrate
52. Silver Nitrate Acs Grade
53. Silver Nitrate Concentrate
54. Silver Nitrate, Premion?
55. Ec 231-853-9
56. Silver Nitrate [mi]
57. Silver Nitrate, Acs Reagent
58. Silver Nitrate (jp17/usp)
59. Silver Nitrate [hsdb]
60. Silver Nitrate [inci]
61. Silver Nitrate, Ag 63.3%
62. Ag (n O3)
63. Silver Nitrate [vandf]
64. Chembl177367
65. Silver Nitrate [mart.]
66. Argentum Nitricum [hpus]
67. Dtxsid3032042
68. Silver Nitrate [who-dd]
69. Silver Nitrate [who-ip]
70. Silver Nitrate Solution, 0.1 M
71. Silver Nitrate Solution, 0.01 M
72. Silver Nitrate Solution, 0.02 M
73. Silver Nitrate Solution, 0.05 M
74. Silver Nitrate, Ar, >=99.9%
75. Silver Nitrate, Lr, >=99.5%
76. Silver Nitrate [ep Monograph]
77. Silver Nitrate [usp Impurity]
78. Silver Nitrate Solution, 0.0282 M
79. Akos015833341
80. Argenti Nitras [who-ip Latin]
81. At27248
82. Db11080
83. Silver(1+) Nitrate [who-ip]
84. Silver Nitrate Solution, 20 % (w/v)
85. Silver Nitrate [un1493] [oxidizer]
86. Bp-30056
87. Silver Nitrate, Acs Reagent, >=99.0%
88. Silver Nitrate, 0.1n Standardized Solution
89. Silver Nitrate, 1.0n Standardized Solution
90. Silver Nitrate, Bioxtra, >99% (titration)
91. Silver Nitrate, Tested According To Ph.eur.
92. Silver Nitrate, Saj First Grade, >=99.8%
93. Silver Nitrate, Trace Metals Grade,99.995%
94. D01730
95. Silver Nitrate, 99.9999% Trace Metals Basis
96. Silver Nitrate, Jis Special Grade, >=99.8%
97. Silver Nitrate, Meets Usp Testing Specifications
98. Toughened Silver Nitrate [usp Monograph]
99. Q207442
100. Silver Nitrate, Puriss. P.a., >=99.5% (at)
101. Sr-01000944542
102. Sr-01000944542-1
103. Silver Nitrate Solution, 2.5 % (w/v) Agno3 In H2o
104. Silver Nitrate, Anhydrous, 99.999% Trace Metals Basis
105. Silver Nitrate, P.a., Acs Reagent, Reag. Iso, 99.8%
106. Silver Nitrate, Reagentplus(r), >=99.0% (titration)
107. Assay-isotopic Standard For Silver, Nist(r) Srm(r) 978a
108. Silver Standard For Icp, For Icp, Ready-to-use, In Nitric Acid
109. Silver Standard For Aas, Ready-to-use, Traceable To Bam, In Nitric Acid
110. Silver Nitrate On Silica Gel, Extent Of Labeling: ~10 Wt. % Loading, +230 Mesh
111. Silver Nitrate, Acculute Standard Volumetric Solution, Final Concentration 0.1n
112. Silver Nitrate, Acculute Standard Volumetric Solution, Final Concentration 0.282n
113. Silver Nitrate, Bioreagent, Suitable For Plant Cell Culture, >99% (titration)
114. Silver Nitrate, Meets Analytical Specification Of Ph. Eur., Bp, Usp, 99.8-100.5%
115. Silver Nitrate, Puriss. P.a., Acs Reagent, Reag. Iso, Reag. Ph. Eur., >=99.8%
116. Silver Atomic Spectroscopy Standard Concentrate 1.00 G Ag, 1.00 G/l, For 1l Standard Solution
117. Silver Standard Solution, Suitable For Atomic Absorption Spectrometry, 1 Mg/ml Ag, 1000 Ppm Ag
| Molecular Weight | 169.873 g/mol |
|---|---|
| Molecular Formula | AgNO3 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Exact Mass | 168.89291 g/mol |
| Monoisotopic Mass | 168.89291 g/mol |
| Topological Polar Surface Area | 62.9 Ų |
| Heavy Atom Count | 5 |
| Formal Charge | 0 |
| Complexity | 18.8 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Anti-Infective Agents, Local
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
SILVER NITRATE AS 0.5% SOLN IS USED IN TREATMENT OF EXTENSIVE BURNS, USUALLY IN CONJUNCTION WITH GENTAMICIN, KANAMYCIN, & OR MAFENIDE.
Gilman, A.G., L.S.Goodman, and A. Gilman. (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 7th ed. New York: Macmillan Publishing Co., Inc., 1985., p. 966
MEDICATION (VET): 1% AQ SOLN HAS ... ASTRINGENT & ANTISEPTIC FUNCTIONS & IS USED IN SINUSES OF TURKEYS, DAILY ON ULCERATED OR INFLAMED CORNEAS & CONJUNCTIVAL TISSUE (0.5-1.0%) OF FISH, CATTLE OR DOGS WHERE IT ALSO DESTROYS THELAZIA NEMATODES ... & TOPICALLY ON BURNS (0.5-1.0%). STRONGER SOLN ... TOPICALLY ON STOMATITIS LESIONS OF CATTLES ... .
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 527
/SRP: FORMER/ MEDICATION (VET): 50 ML OF 0.5% SOLN INTRAUTERINE HAS BEEN RECOMMENDED IN ENDOMETRITIS OF COWS. GLYCERINATED SOLN ... CAN BE USED TO DESTROY PATENT URACHUS IN FOALS. WEAK SOLN (1%) CAN BE USED ORALLY TO CONTROL GASTRIC BLEEDING, & IT /HAS BEEN/ ... USED AS INTESTINAL ANTISEPTIC.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 527
For more Therapeutic Uses (Complete) data for SILVER NITRATE (23 total), please visit the HSDB record page.
VET: STRONG SOLN OR "STICKS" ARE NO LONGER CONSIDERED SAFE FOR CAUTERY OF CORNEAL ULCERS.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 527
THE COMPOUND HAS ... BEEN USED IN TREATMENT OF TRAUMATIC WOUNDS, BUT PRESENT EVIDENCE INDICATES THAT SILVER NITRATE MAY ACTUALLY INCREASE INFECTION RATE.
Osol, A. (ed.). Remington's Pharmaceutical Sciences. 16th ed. Easton, Pennsylvania: Mack Publishing Co., 1980., p. 1108
MOST IMPORTANT ADVERSE EFFECT OF BURN THERAPY USING SILVER NITRATE IS DEPLETION OF CHLORIDE CAUSED BY PRECIPITATION OF INSOL SILVER CHLORIDE; THIS IS ... LIKELY TO OCCUR IF ... APPLIED TO EXTENSIVE AREAS OVER PROLONGED PERIODS. ... SMALL AMT MAY BE ABSORBED THROUGH SKIN OVER PROLONGED PERIODS, RESULTING IN ARGYRIA ... .
American Medical Association, AMA Department of Drugs, AMA Drug Evaluations. 3rd ed. Littleton, Massachusetts: PSG Publishing Co., Inc., 1977., p. 897
... SILVER NITRATE MAY CAUSE SEVERE LOCAL IRRITATION /WHEN EMPLOYED FOR PROPHYLAXIS OF OPHTHALMIA NEONATORUM/.
American Medical Association, AMA Department of Drugs, AMA Drug Evaluations. 3rd ed. Littleton, Massachusetts: PSG Publishing Co., Inc., 1977., p. 955
For more Drug Warnings (Complete) data for SILVER NITRATE (8 total), please visit the HSDB record page.
Anti-Infective Agents, Local
Substances used on humans and other animals that destroy harmful microorganisms or inhibit their activity. They are distinguished from DISINFECTANTS, which are used on inanimate objects. (See all compounds classified as Anti-Infective Agents, Local.)
D - Dermatologicals
D08 - Antiseptics and disinfectants
D08A - Antiseptics and disinfectants
D08AL - Silver compounds
D08AL01 - Silver nitrate
SILVER, AS AG NITRATE, IS ABSORBED FROM THE RESP & GI TRACTS ... ABSORPTION OF SILVER NITRATE THROUGH THE INTACT SKIN IS OF NO PHYSIOLOGICAL SIGNIFICANCE, HOWEVER, SOME ABSORPTION THROUGH MUCOUS MEMBRANES OF THE NOSE & THROAT PROBABLY OCCURS. ABSORBED SILVER IS TENACIOUSLY RETAINED ... IN ELASTIC TISSUES THROUGHOUT THE BODY.
Clayton, G. D. and F. E. Clayton (eds.). Patty's Industrial Hygiene and Toxicology: Volume 2A, 2B, 2C: Toxicology. 3rd ed. New York: John Wiley Sons, 1981-1982., p. 1889
... Silver in brain and spinal cord sections from rats treated with ... silver nitrate was visualized by physical appearance. The silver penetrated the blood brain barrier and accumulated in the neurons and glia. The distribution of silver in the central nervous system was heterogeneous. Even with low doses and short survival periods, silver accumulated in large motorneurons in the brain stem and spinal cord and in the neurons in the cerebellar nuclei. Silver was only found in the diecenphalic and telecenphalic structures after extensive exposure. Silver distribution following oral ... AgNO3 treatment ... resulted in a relatively high content of silver in the glia. ... Electron microscopic studies /revealed/ ... silver ... intracellularly in the lysosomes and extracellularly in basement membranes and elastic fibers of the vessels.
PMID:6880627 Rungby J, Danscher G; Acta Neuropathol 60 (1-2): 92-8 (1983)
All forms of silver are cumulative once they enter body tissues, and very little is excreted.
Sittig, M. Handbook of Toxic and Hazardous Chemicals and Carcinogens, 1985. 2nd ed. Park Ridge, NJ: Noyes Data Corporation, 1985., p. 789
In argyric rats given 0.5 percent silver nitrate in their drinking water for nine months, silver was especially found in lysosomes of the liver's Kupffer cells, at the basal membrane of the capillaries, and the connective tissue cells of the pancreas.
USEPA; Ambient Water Quality Criteria Doc: Silver p.C-49 (1980) EPA 440/5-80-071
For more Absorption, Distribution and Excretion (Complete) data for SILVER NITRATE (17 total), please visit the HSDB record page.
/The investigators/ reported that rats injected iv with radioactive silver nitrate excreted silver in the bile mainly bound to a low molecular-wt complex which appeared to be glutathione.
Friberg, L., Nordberg, G.F., Kessler, E. and Vouk, V.B. (eds). Handbook of the Toxicology of Metals. 2nd ed. Vols I, II.: Amsterdam: Elsevier Science Publishers B.V., 1986., p. V2 525
A marked variation in biliary excretion was observed in different species administered silver as silver nitrate in a single iv injection at 0.1 mg/kg of silver over a 2 hour period ... . Thirty minutes after treatment, male Sprague Dawley rats excreted silver into the bile at a rate of 0.25 ug/min/kg, New Zealand White male rabbits excreted 0.05 ug/min/kg, and mongrel male dogs excreted 0.005 ug/min/kg. The concentration of silver in the plasma was markedly lower in the dog than in the rat or rabbit, indicating a larger volume of distribution in the dog. This variation appears to be attributable to differences in the transfer of silver from liver to bile. The species with the lowest biliary excretion rate (dog) had the highest liver concentration of silver (rat 1.24, rabbit = 2.13 and dog = 2.9 ug silver/g liver). In all species, the concentration of silver in the bile was greater than that in plasma with no observable dose gradient, thereby indicating an active transport process and a saturable mechanism.
USEPA; Reregistration Eligibility Decision Document - Silver. Washington, DC: USEPA, Off Pest Prog. USEPA 738-R-94-021, p.12 Sept 1992. Available from, as of Feb 24, 2002: https://www.epa.gov/pesticides/reregistration/status.htm
Silver injected into rats as radioactive silver nitrate had a half-time of 2.2 days. ... For rats given a single ip injection of radioactive silver nitrate, a 50% reduction in radioactivity of whole blood, plasma, kidney, & liver after approx 40 hr. A 50% reduction in the radioactivity content of spleen occurred after about 70 hr while that of brain took about 84 hr. The biological half-time of silver in the lungs of a human exposed to silver has been estimated to be 1 day, while that in the liver was 52 days.
Friberg, L., Nordberg, G.F., Kessler, E. and Vouk, V.B. (eds). Handbook of the Toxicology of Metals. 2nd ed. Vols I, II.: Amsterdam: Elsevier Science Publishers B.V., 1986., p. V2 525
The biphasic elimination half-times after silver nitrate ingestion by monkeys were 0.3 & 3 days.
American Conference of Governmental Industrial Hygienists, Inc. Documentation of the Threshold Limit Values and Biological Exposure Indices. 6th ed. Volumes I, II, III. Cincinnati, OH: ACGIH, 1991., p. 1397
Whole-body retention studies in mice and monkeys following oral dosing with radiolabeled silver nitrate indicate that silver excretion in these species follows a biexponential profile with biological half-lives of 0.1 and 1.6 days in mice and 0.3 and 3 days in monkeys. In similarly exposed rats and dogs, silver excretion followed a triexponential profile with biological half-lives of 0.1, 0.7, and 5.9 days in rats and 0.1, 7.6, and 33.8 days in dogs .. .
DHHS/ATSDR; Toxicological Profile for Silver p. 31 TP-90-24 (1990)
To elucidate the possible mechanism of initiation of lipid peroxidation in silver nitrate treated erythrocytes, the effect of AgNO3 on Hb catalyzed peroxidation of phospholipid liposomes was studied. Ag+ significantly increases the rate of Hb-catalyzed peroxidation. The inhibition of this effect by superoxide dismutase and catalase suggests that superoxide radical and H2O2 are involved. It was demonstrated also that Ag+ potentiates the Hb autoxidation and increases the binding of Hb to the erythrocyte membrane.
PMID:3707552 Ribarov S et al; Biomed Biochim Acta 45 (3): 321-30 (1986)
Silver nitrate exhibits antiseptic, germicidal, astringent, and caustic or escharotic activity. These effects may result from silver ions readily combining with sulfhydryl, carboxyl, phosphate, amino, and other biologically important chemical groups. When silver ions interact with proteins, physical properties of the protein are usually altered; the protein may be denatured and precipitation usually occurs. Silver ions act on the surface of bacteria causing substantial changes in the cell wall and membrane. The germicidal activity of silver nitrate may be attributed to precipitation of bacterial proteins by liberated silver ions.
American Hospital Formulary Service-Drug Information 88. Bethesda, MD: American Society of Hospital Pharmacists, 1988 (Plus supplements)., p. 1529

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

ABOUT THIS PAGE
60
PharmaCompass offers a list of Silver Nitrate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Silver Nitrate manufacturer or Silver Nitrate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Silver Nitrate manufacturer or Silver Nitrate supplier.
PharmaCompass also assists you with knowing the Silver Nitrate API Price utilized in the formulation of products. Silver Nitrate API Price is not always fixed or binding as the Silver Nitrate Price is obtained through a variety of data sources. The Silver Nitrate Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Silver Nitrate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Silver Nitrate, including repackagers and relabelers. The FDA regulates Silver Nitrate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Silver Nitrate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Silver Nitrate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Silver Nitrate supplier is an individual or a company that provides Silver Nitrate active pharmaceutical ingredient (API) or Silver Nitrate finished formulations upon request. The Silver Nitrate suppliers may include Silver Nitrate API manufacturers, exporters, distributors and traders.
click here to find a list of Silver Nitrate suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Silver Nitrate as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Silver Nitrate API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Silver Nitrate as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Silver Nitrate and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Silver Nitrate NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Silver Nitrate suppliers with NDC on PharmaCompass.
Silver Nitrate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Silver Nitrate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Silver Nitrate GMP manufacturer or Silver Nitrate GMP API supplier for your needs.
A Silver Nitrate CoA (Certificate of Analysis) is a formal document that attests to Silver Nitrate's compliance with Silver Nitrate specifications and serves as a tool for batch-level quality control.
Silver Nitrate CoA mostly includes findings from lab analyses of a specific batch. For each Silver Nitrate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Silver Nitrate may be tested according to a variety of international standards, such as European Pharmacopoeia (Silver Nitrate EP), Silver Nitrate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Silver Nitrate USP).
