
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
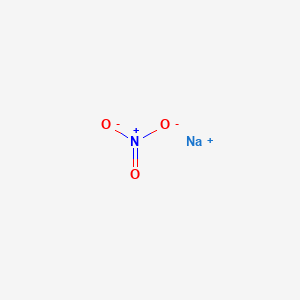

1. 7631-99-4
2. Chile Saltpeter
3. Cubic Niter
4. Soda Niter
5. Nitrate Of Soda
6. Sodium Saltpeter
7. Nitrate, Sodium
8. Nitrate De Sodium
9. Nitric Acid, Sodium Salt
10. Nitric Acid Sodium Salt (1:1)
11. Nitric Acid Monosodium Salt
12. Sodium(i) Nitrate (1:1)
13. Sodium;nitrate
14. Natrum Nitricum
15. Mfcd00011119
16. 8m4l3h2zvz
17. Chili Saltpeter
18. Ins No.251
19. 15621-57-5
20. Chembl1644698
21. Chebi:63005
22. Ins-251
23. Nsc-77390
24. E-251
25. Na (n O3)
26. Sodium Nitrate(dot)
27. Caswell No. 781
28. Sodiumnitrate
29. Chile Salpeter
30. Saltpeter (chile) (van)
31. Nitrate De Sodium [french]
32. Ccris 558
33. Hsdb 726
34. Nano3
35. Einecs 231-554-3
36. Nsc 77390
37. Un1498
38. Unii-8m4l3h2zvz
39. Epa Pesticide Chemical Code 076104
40. Cubicniter
41. Natriumnitrat
42. Dsstox_cid_937
43. Sodium Nitrate (nano3)
44. Ec 231-554-3
45. Sodium Trioxidonitrate(1-)
46. Dsstox_rid_75877
47. Sodium Nitrate [ii]
48. Sodium Nitrate [mi]
49. Dsstox_gsid_20937
50. Sodium Nitrate, Acs Reagent
51. Sodium Nitrate [fcc]
52. Sodium Nitrate [hsdb]
53. Sodium Nitrate [inci]
54. Natrum Nitricum [hpus]
55. Sodium Nitrate [vandf]
56. Sodium Nitrate [mart.]
57. Sodium Nitrate, Lr, >=98%
58. Dtxsid6020937
59. Sodium Nitrate [who-dd]
60. Sodium Nitrate, Acs Reagent Grade
61. Sodium Nitrate, Analytical Standard
62. Sodium Nitrate, Ar, >=99.5%
63. Tox21_202595
64. Akos015902552
65. Akos025243988
66. Sodium Nitrate, Bioxtra, >=99.0%
67. Sodium Nitrate, Beads, 1-2 Mm, 98%
68. Sodium Nitrate, Containing In The Dry State More Than 16,3 Per Cent By Weight Of Nitrogen
69. Ncgc00260143-01
70. Sodium Nitrate [un1498] [oxidizer]
71. E251
72. Sodium Nitrate, Acs Reagent, >=99.0%
73. Cas-7631-99-4
74. Sodium Nitrate, 99.999% (metals Basis)
75. Sodium Nitrate, Bioultra, >=99.0% (t)
76. Sodium Nitrate, Reagentplus(r), >=99.0%
77. Sodium Nitrate, 99.995% Trace Metals Basis
78. Sodium Nitrate, Saj First Grade, >=99.0%
79. Sodium Nitrate, Jis Special Grade, >=99.0%
80. Q184373
81. Sodium Nitrate, >=99.0%, Plant Cell Culture Tested
82. Sodium Nitrate, Anhydrous, 99.995% Trace Metals Basis
83. Nitrogen And Oxygen Isotopes In Nitrate, Nist(r) Rm 8569
84. Sodium Nitrate, Puriss. P.a., Acs Reagent, Reag. Iso, >=99.5%
85. Sodium Nitrate, Meets Analytical Specification Of Fcc, E251, 99.0-100.5%
86. Sodium Nitrate, P.a., Acs Reagent, Reag. Iso, Reag. Ph. Eur., 99%
87. Sodium Nitrate, Anhydrous, Free-flowing, Redi-dri(tm), Reagentplus(r), >=99%
| Molecular Weight | 84.995 g/mol |
|---|---|
| Molecular Formula | NNaO3 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Exact Mass | 84.97758714 g/mol |
| Monoisotopic Mass | 84.97758714 g/mol |
| Topological Polar Surface Area | 62.9 Ų |
| Heavy Atom Count | 5 |
| Formal Charge | 0 |
| Complexity | 18.8 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
3= MODERATELY TOXIC: Probable oral lethal dose (human) 0.5-5 g/kg, Between 1 ounce and 1 pint (or 1 lb) for 79 kg person (150 lb).
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. II-124
Carcinogens
Substances that increase the risk of NEOPLASMS in humans or animals. Both genotoxic chemicals, which affect DNA directly, and nongenotoxic chemicals, which induce neoplasms by other mechanism, are included. (See all compounds classified as Carcinogens.)
In humans, ingested nitrate is rapidly absorbed from the proximal small bowel and distributed throughout the body. Nitrate then enters the large bowel from the blood, where it is rapidly converted to highly reactive nitrite, in part by fecal microorganisms. The formed nitrite is reabsorbed into the blood, where it reacts with the ferrous (Fe2+) iron of deoxyhemoglobin, forming methemoglobin with iron in the ferric (Fe3+) valence state. Ferric iron is unable to transport oxygen. Nitrates are rapidly converted in the liver to denitrated metabolites and inorganic nitrites, which are then excreted in urine. Approximately 60% to 70% of an ingested nitrate dose is excreted in urine within the first 24 hours. About 25% is excreted in saliva through an active blood nitrate transport system and potentially is reabsorbed. Half-lives of parent nitrate compounds are usually <1 hour; half-lives of metabolites range from 1 to 8 hours. /Nitrates/
ATSDR; Case Studies in Environmental Medicine. NITRATE/NITRITE TOXICITY. p 9. Course: SS3054. Revision Date: January 2001 Original Date: October 1991 Expiration Date: January 2007.
... Nitrate is concentrated in saliva, where a part of it is reduced to nitrite by bacterial nitrate reductases. /The authors/ tested if ingestion of inorganic nitrate would affect the salivary and systemic levels of nitrite and S-nitrosothiols, both considered to be circulating storage pools for NO. Levels of nitrate, nitrite, and S-nitrosothiols were measured in plasma, saliva, and urine before and after ingestion of sodium nitrate (10 mg/kg). Nitrate levels increased greatly in saliva, plasma, and urine after the nitrate load. Salivary S-nitrosothiols also increased, but plasma levels remained unchanged. A 4-fold increase in plasma nitrite was observed after nitrate ingestion. If, however, the test persons avoided swallowing after the nitrate load, the increase in plasma nitrite was prevented, thereby illustrating its salivary origin. We show that nitrate is a substrate for systemic generation of nitrite. There are several pathways to further reduce this nitrite to NO. These results challenge the dogma that nitrate is biologically inert and instead suggest that a complete reverse pathway for generation of NO from nitrate exists.
PMID:15223073 Lundberg JO, Govoni M; Free Radic Biol Med 37 (3): 395-400 (2004)
Nitrite was quickly eliminated from blood plasma of sheep (T1/2 = 0.49 hr). Three hours after its administration, nitrite ion disappeared from blood plasma. Simultaneously the appearance of nitrate was observed. After sodium nitrite or nitrate intravenous administration, the kinetic parameters for nitrate were similar (Kel = 0.150/ h and 0.154/ hr, respectively). Urine excretion of nitrite amounted to only 0.29%+/- 0.22 of the administered dose. Urine excretion of nitrate was 13.80%+/-5.78 of the administered dose of nitrite and was slightly lower than that observed after the administration of nitrate (16.12%+/- 6.95). The quick elimination of nitrite appears to be mainly from its conversion into nitrate, because the nitrate elimination in urine. as well as the values of its kinetic parameters, were similar after the administration of nitrite or nitrate. Elimination of nitrate from blood plasma of sheep over 80% was different than its excretion in urine in the unchanged.
Lewicki J et al; Small Ruminant Res; 13 (2). 141-6 (1994)
... Sodium ... nitrate /is/ for the most part rapidly absorbed & excreted unchanged ... /following oral ingestion/.
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. III-316
For more Absorption, Distribution and Excretion (Complete) data for SODIUM NITRATE (10 total), please visit the HSDB record page.
Transformation of nitrate to nitrite usually occurs within alimentary tract. Nitrite is intermediary product in redn of nitrate to ammonia within rumen of sheep & cow: it appears that above certain concn of nitrate, rate of redn of nitrite to ammonia becomes limiting & ... nitrite accumulates. Danger of nitrate poisoning is considered slight if nitrate is reduced in rumen or if complete redn to ammonia occurs rapidly ... /Inorganic nitrate salts/
Clarke, M. L., D. G. Harvey and D. J. Humphreys. Veterinary Toxicology. 2nd ed. London: Bailliere Tindall, 1981., p. 66
... Bacterial redn of nitrate to nitrite in human stomach was reported ... & n-nitrosodiphenylamine was identified in stomach contents of 31 human subjects who had received sodium nitrate & diphenylamine intragastrically.
Searle, C. E. (ed.). Chemical Carcinogens. ACS Monograph 173. Washington, DC: American Chemical Society, 1976., p. 590
Where bacteria are present and the environment can be anaerobic, nitrate can be reduced to nitrite. The main site for this reaction is mouth and stomach, but nitrite formation in the lower intestine and in the bladder (urinary infection) may also be of some toxicological importance. Nitrite may be further reduced to nitrogen by bacteria under some conditions. In blood, nitrite transforms hemoglobin to methemoglobin and is simultaneously oxidized to nitrate. Normally methemoglobin gradually reverts to hemoglobin through enzymatic reactions. Nitrite has vasodilating properties, probably through transformation into nitric oxide (NO) or a NO-containing molecule acting as a signal factor for smooth muscle relaxation. Nitrite easily transforms into a nitrosating agent in an acidic environment and can react with a variety of compounds, eg ascorbic acid, amines, amides. Nitrosation can also be mediated by bacteria, eg in the stomach. Some reaction products are carcinogenic (eg most nitrosoamines and amides). /Nitrate and nitrite/
IPCS; Poisons Information Monograph G016: Nitrates and nitrites. (September 1996). Available from, as of October 24, 2006: https://www.inchem.org/documents/pims/chemical/pimg016.htm
Nitrate salts as such are no more toxic than other neutral salts, but if not promptly absorbed, they may be reduced to nitrites by bacteria in the bowel ... /Nitrate salts/
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. 124
For more Metabolism/Metabolites (Complete) data for SODIUM NITRATE (8 total), please visit the HSDB record page.
There is an active endogenous nitrogen cycle in humans that involves nitrate and nitrite, which are interconvertible in vivo. Nitrosating agents that arise from nitrite under acidic gastric conditions react readily with nitrosatable compounds, especially secondary amines and amides, to generate N-nitroso compounds. These nitrosating conditions are enhanced following ingestion of additional nitrate, nitrite or nitrosatable compounds. Some of the N-nitroso compounds that could be formed in humans under these conditions are known carcinogens.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V. 94: p. 323 (2010)
The principal mechanism of nitrite toxicity is the oxidation of the ferrous iron (Fe2+) in deoxyhemoglobin to the ferric (Fe3+) valence state, producing methemoglobin. /Nitrates and nitrites/
ATSDR; Case Studies in Environmental Medicine. NITRATE/NITRITE TOXICITY.p 9-11. Course: SS3054. Revision Date: January 2001 Original Date: October 1991 Expiration Date: January 2007.
... The major concern of possible long-term effects of exposure to nitrate and nitrite is associated with formation of nitroso compounds, many of which are carcinogenic. This formation may take place wherever nitrite and nitrosable compounds are present, but it is favored by acidic conditions or the presence of some bacteria. The gastrointestinal tract and especially the stomach is regarded as the main formation site, but nitrosation reactions can also take place in an infected urinary bladder ... /Nitrate and nitrite poisoning/
IPCS; Poisons Information Monograph G016: Nitrates and nitrites. (September 1996). Available from, as of October 24, 2006: https://www.inchem.org/documents/pims/chemical/pimg016.htm
Nitrates can be reduced to nitrites which can react with amines or amides and form N-nitroso cmpd (containing the group =N-N=O). N-nitroso cmpd are carcinogenic in a wide range of animal species, most are mutagenic in test systems and some have been teratogenic in animals. It is highly probable that N-nitroso cmpd also may be carcinogenic in man. Therefore exposure to N-nitroso cmpd and their precursors (nitrite, amines and amides) should be kept as low as practically achievable. Relationships have been sought between occurrence of stomach cancer and nitrate content of soil and water in Chile, Colombia and the United Kingdom, but none was established. /Nitrate/
European Chemicals Bureau; IUCLID Dataset, Potassium nitrate (7757-79-1) (2000 CD-ROM edition). Available from, as of October 26, 2006: https://esis.jrc.ec.europa.eu/
Related Excipient Companies

Excipients by Applications
Market Place

ABOUT THIS PAGE
35
PharmaCompass offers a list of Sodium Nitrate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Sodium Nitrate manufacturer or Sodium Nitrate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Sodium Nitrate manufacturer or Sodium Nitrate supplier.
PharmaCompass also assists you with knowing the Sodium Nitrate API Price utilized in the formulation of products. Sodium Nitrate API Price is not always fixed or binding as the Sodium Nitrate Price is obtained through a variety of data sources. The Sodium Nitrate Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Sodium Nitrate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Sodium Nitrate, including repackagers and relabelers. The FDA regulates Sodium Nitrate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Sodium Nitrate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Sodium Nitrate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Sodium Nitrate supplier is an individual or a company that provides Sodium Nitrate active pharmaceutical ingredient (API) or Sodium Nitrate finished formulations upon request. The Sodium Nitrate suppliers may include Sodium Nitrate API manufacturers, exporters, distributors and traders.
click here to find a list of Sodium Nitrate suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
Sodium Nitrate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Sodium Nitrate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Sodium Nitrate GMP manufacturer or Sodium Nitrate GMP API supplier for your needs.
A Sodium Nitrate CoA (Certificate of Analysis) is a formal document that attests to Sodium Nitrate's compliance with Sodium Nitrate specifications and serves as a tool for batch-level quality control.
Sodium Nitrate CoA mostly includes findings from lab analyses of a specific batch. For each Sodium Nitrate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Sodium Nitrate may be tested according to a variety of international standards, such as European Pharmacopoeia (Sodium Nitrate EP), Sodium Nitrate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Sodium Nitrate USP).
