
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
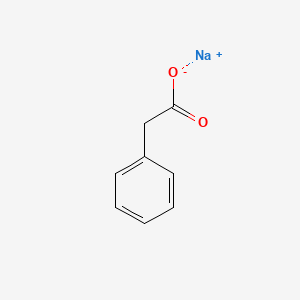

1. Phenylacetate
2. Phenylacetic Acid
3. Phenylacetic Acid, Ammonium Salt
4. Phenylacetic Acid, Calcium Salt
5. Phenylacetic Acid, Cesium Salt
6. Phenylacetic Acid, Lithium Salt
7. Phenylacetic Acid, Mercury Salt
8. Phenylacetic Acid, Potassium Salt
9. Phenylacetic Acid, Rubidium Salt
10. Phenylacetic Acid, Sodium Salt
11. Phenylacetic Acid, Sodium Salt , Carboxy-(11)c-labeled Cpd
1. 114-70-5
2. Sodium 2-phenylacetate
3. Phenylacetic Acid Sodium Salt
4. Sodium Benzeneacetate
5. Benzeneacetic Acid, Sodium Salt
6. Sodium Phenylacetate [usan]
7. Sodium Phenylacetic Acid
8. Nsc-3039
9. 48n6u1781g
10. Sodium 2-phenylethanoate
11. Sodium Phenylacetate;sodium 2-phenylacetate
12. Phenylacetate Sodium Salt
13. Hsdb 5581
14. Acetic Acid, Phenyl-, Sodium Salt
15. Nsc 3039
16. Einecs 204-052-7
17. Phenylessigsaure Natrium-salz [german]
18. Phenylessigsaure Natrium-salz
19. Unii-48n6u1781g
20. Sodium,2-phenylacetate
21. Sodium;2-phenylacetate
22. Schembl62440
23. Chembl1200358
24. Dtxsid8059427
25. Sodium Phenylacetate (jan/usan)
26. Hms2089o12
27. Hms2094m19
28. Sodium Phenylacetate [jan]
29. Sodium Phenylacetate [hsdb]
30. Sodium Phenylacetate [vandf]
31. Sodium Phenylacetate [mart.]
32. Sodium Phenylacetate [who-dd]
33. Akos003052998
34. Akos015890373
35. Sodium Phenylacetate [orange Book]
36. Db-041227
37. Ammonul Component Sodium Phenylacetate
38. Ucephan Component Sodium Phenylacetate
39. D05867
40. Sodium Phenylacetate Component Of Ammonul
41. Sodium Phenylacetate Component Of Ucephan
42. A800813
43. A824291
44. Q27259162
| Molecular Weight | 158.13 g/mol |
|---|---|
| Molecular Formula | C8H7NaO2 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 2 |
| Exact Mass | 158.03437374 g/mol |
| Monoisotopic Mass | 158.03437374 g/mol |
| Topological Polar Surface Area | 40.1 Ų |
| Heavy Atom Count | 11 |
| Formal Charge | 0 |
| Complexity | 119 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Antimetabolites, Antineoplastic
National Library of Medicine's Medical Subject Headings. Phenylacetic acid. Online file (MeSH, 2017). Available from, as of July 12, 2017: https://www.nlm.nih.gov/mesh/2017/mesh_browser/MBrowser.html
/CLINICAL TRIALS/ ClinicalTrials.gov is a registry and results database of publicly and privately supported clinical studies of human participants conducted around the world. The Web site is maintained by the National Library of Medicine (NLM) and the National Institutes of Health (NIH). Each ClinicalTrials.gov record presents summary information about a study protocol and includes the following: Disease or condition; Intervention (for example, the medical product, behavior, or procedure being studied); Title, description, and design of the study; Requirements for participation (eligibility criteria); Locations where the study is being conducted; Contact information for the study locations; and Links to relevant information on other health Web sites, such as NLM's MedlinePlus for patient health information and PubMed for citations and abstracts for scholarly articles in the field of medicine. Chloramphenicol is included in the database.
NIH/NLM; ClinicalTrials.Gov. Available from, as of July 12, 2017: https://clinicaltrials.gov/
Sodium phenylacetate and sodium benzoate injection is indicated as adjunctive therapy in pediatric and adult patients for the treatment of acute hyperammonemia and associated encephalopathy in patients with deficiencies in enzymes of the urea cycle. During acute hyperammonemic episodes, arginine supplementation, caloric supplementation, dietary protein restriction, hemodialysis, and other ammonia lowering therapies should be considered.
NIH; DailyMed. Current Medication Information for Sodium phenylacetate and sodium benzoate injection, solution, concentrate (Updated: February 2016). Available from, as of July 12, 2017: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=3529be10-0004-4628-9ca1-67a8acd86b51
/Sodium phenylacetate in combination with sodium benzoate/ is indicated as adjunctive therapy for the treatment of acute hyperammonemia and associated encephalopathy in patients with deficiencies in enzymes of the urea cycle. In acute neonatal hyperammonemic coma, in moderate to severe episodes of hyperammonemic encephalopathy, and in episodes of hyperammonemia which fail to respond to an initial course of /Sodium phenylacetate/sodium benzoate/ therapy, hemodialysis is the most rapid and effective technique for removing ammonia. In such cases, the concomitant administration of /Sodium phenylacetate/sodium benzoate/ can help prevent the re-accumulation of ammonia by increasing waste nitrogen excretion. /Included in US product label/
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 678
For more Therapeutic Uses (Complete) data for Sodium phenylacetate (9 total), please visit the HSDB record page.
Known hypersensitivity to sodium phenylacetate or sodium benzoate.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017
Plasma ammonia concentrations, neurologic status, laboratory tests, and clinical response should be monitored closely during drug treatment. Because urinary loss of potassium is enhanced by excretion of the non-absorbable anions phenylacetylglutamine and hippurate (conjugation products of phenylacetate and benzoate), plasma potassium concentrations should be carefully monitored and replacement therapy provided when necessary. In addition, serum electrolyte concentrations should be monitored and maintained within the normal range.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017
The Urea Cycle Disorders Conference Group and some experts recommend monitoring plasma concentrations of ammonia scavenging drugs (e.g., sodium phenylacetate and sodium benzoate) to avoid toxicity. In addition, these experts state that written orders for the drugs should be double-checked to avoid overdosage. In the absence of facilities for drug concentration monitoring, the risk of overdosage should be weighed against potential benefits of repeating a loading dose.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017
Each g of sodium phenylacetate provides 6.3 mEq (145 mg) of sodium, and each g of sodium benzoate provides 7 mEq (160 mg of sodium); each mL of injection concentrate labeled as containing 100 mg each of sodium phenylacetate and sodium benzoate provides 1.33 mEq (30.5 mg) of sodium. Sodium phenylacetate and sodium benzoate injection should be used with caution, if at all, in patients with congestive heart failure (CHF), severe renal impairment, or sodium retention with edema. If adverse effects associated with increased sodium concentrations occur, the drug should be discontinued, the patient promptly evaluated, and appropriate measures taken.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017
For more Drug Warnings (Complete) data for Sodium phenylacetate (14 total), please visit the HSDB record page.
Antimetabolites, Antineoplastic
Antimetabolites that are useful in cancer chemotherapy. (See all compounds classified as Antimetabolites, Antineoplastic.)
The pharmacokinetics of intravenously administered sodium phenylacetate and sodium benzoate were characterized in healthy adult volunteers. Both benzoate and phenylacetate exhibited nonlinear kinetics. Following 90 minute intravenous infusion mean AUC(last) for benzoate was 20.3, 114.9, 564.6, 562.8, and 1599.1 ug/mL following doses of 1, 2, 3.75, 4, and 5.5 g/sq m, respectively. The total clearance decreased from 5.19 to 3.62 L/h/sq m at the 3.75 and 5.5 g/sq m doses, respectively.
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 678
... Phenylacetate exhibited nonlinear kinetics following the priming dose regimens. AUC(last) was 175.6, 713.8, 2040.6, 2181.6, and 3829.2 ug/hr/mL following doses of 1, 2, 3.75, 4, and 5.5 g/sq m, respectively. The total clearance decreased from 1.82 to 0.89 ug/hr/mL with increasing dose (3.75 and 4 g/sq m, respectively). During the sequence of 90 minute priming infusion followed by a 24 hour maintenance infusion, phenylacetate was detected in the plasma at the end of infusion (Tmax of 2 hr at 3.75 g/sq m) whereas, benzoate concentrations declined rapidly (Tmax of 1.5 hr at 3.75 g/sq m) and were undetectable at 14 and 26 hours following the 3.75 and 4 g/sq m dose, respectively.
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 679
... Phenylacetylglutamine is excreted by the kidneys via glomerular filtration and tubular secretion. ...
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 679
/It is/ not known whether sodium phenylacetate or sodium benzoate or their conjugated metabolites are distributed into milk.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017
For more Absorption, Distribution and Excretion (Complete) data for Sodium phenylacetate (11 total), please visit the HSDB record page.
Phenylacetate conjugates with glutamine in the liver and kidneys to form phenylacetylglutamine, via acetylation. Phenylacetylglutamine is excreted by the kidneys via glomerular filtration and tubular secretion. The nitrogen content of phenylacetylglutamine per mole is identical to that of urea (both contain 2 moles of nitrogen). ...
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 679
A difference in the metabolic rates for phenylacetate and benzoate was noted. The formation of hippurate from benzoate occurred more rapidly than that of phenylacetylglutamine from phenylacetate, and the rate of elimination for hippurate appeared to be more rapid than that for phenylacetylglutamine.
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 679
Sodium phenylacetate and sodium benzoate are metabolized in the liver.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017
Sodium phenylacetate and sodium benzoate are metabolized in the kidney and phenylacetylglutamine (metabolite of sodium phenylacetate) and hippuric acid (metabolite of sodium benzoate) are mainly excreted in urine.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017
Sodium phenylacetate and sodium benzoate decrease ammonia concentrations by serving as alternatives to urea for the excretion of waste nitrogen. Phenylacetate is conjugated with glutamine in the liver and kidneys to form phenylacetylglutamine, and benzoate is conjugated with glycine to form hippuric acid; phenylacetylglutamine and hippuric acid subsequently are excreted in urine. Conjugation of 1 mole of phenylacetate with glutamine removes 2 moles of nitrogen, while conjugation of 1 mole of benzoate with glycine removes 1 mole of nitrogen. The nitrogen content of phenylacetylglutamine is identical to that of urea (i.e., both contain 2 moles of nitrogen). Glutamine and glycine used in these reactions are replaced by synthesis, thereby reducing the nitrogen pool and attenuating the risk of ammonia- and glutamine-induced neurotoxicity in patients with deficiencies of urea cycle enzymes.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

ABOUT THIS PAGE
93
PharmaCompass offers a list of Sodium Phenylacetate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Sodium Phenylacetate manufacturer or Sodium Phenylacetate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Sodium Phenylacetate manufacturer or Sodium Phenylacetate supplier.
PharmaCompass also assists you with knowing the Sodium Phenylacetate API Price utilized in the formulation of products. Sodium Phenylacetate API Price is not always fixed or binding as the Sodium Phenylacetate Price is obtained through a variety of data sources. The Sodium Phenylacetate Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Sodium Phenylacetate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Sodium Phenylacetate, including repackagers and relabelers. The FDA regulates Sodium Phenylacetate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Sodium Phenylacetate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Sodium Phenylacetate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Sodium Phenylacetate supplier is an individual or a company that provides Sodium Phenylacetate active pharmaceutical ingredient (API) or Sodium Phenylacetate finished formulations upon request. The Sodium Phenylacetate suppliers may include Sodium Phenylacetate API manufacturers, exporters, distributors and traders.
click here to find a list of Sodium Phenylacetate suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Sodium Phenylacetate DMF (Drug Master File) is a document detailing the whole manufacturing process of Sodium Phenylacetate active pharmaceutical ingredient (API) in detail. Different forms of Sodium Phenylacetate DMFs exist exist since differing nations have different regulations, such as Sodium Phenylacetate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Sodium Phenylacetate DMF submitted to regulatory agencies in the US is known as a USDMF. Sodium Phenylacetate USDMF includes data on Sodium Phenylacetate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Sodium Phenylacetate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Sodium Phenylacetate suppliers with USDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Sodium Phenylacetate as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Sodium Phenylacetate API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Sodium Phenylacetate as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Sodium Phenylacetate and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Sodium Phenylacetate NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Sodium Phenylacetate suppliers with NDC on PharmaCompass.
Sodium Phenylacetate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Sodium Phenylacetate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Sodium Phenylacetate GMP manufacturer or Sodium Phenylacetate GMP API supplier for your needs.
A Sodium Phenylacetate CoA (Certificate of Analysis) is a formal document that attests to Sodium Phenylacetate's compliance with Sodium Phenylacetate specifications and serves as a tool for batch-level quality control.
Sodium Phenylacetate CoA mostly includes findings from lab analyses of a specific batch. For each Sodium Phenylacetate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Sodium Phenylacetate may be tested according to a variety of international standards, such as European Pharmacopoeia (Sodium Phenylacetate EP), Sodium Phenylacetate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Sodium Phenylacetate USP).

