

Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
FDF
0
FDA Orange Book

0
Canada

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
EDQM
0
USP
0
JP
0
Others
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
US Medicaid
NA
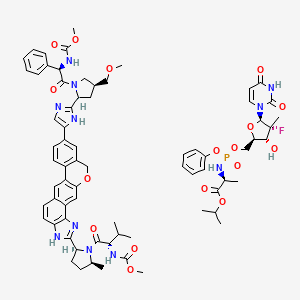

1. Sofosbuvir Velpatasvir Drug Combination
2. Sofosbuvir, Velpatasvir Drug Combination
3. Sofosbuvir-velpatasvir Drug Combination
1. Sofosbuvir & Velpatasvir
2. Sofosbuvir + Velpatasvir
3. Epclusa Tablet
4. Sofosbuvir / Velpatasvir
5. Velpatasvir / Sofosbuvir
6. Sofosbuvir And Velpatasvir
7. Sof / Vlp
8. Sofosbuvir Mixture With Velpatasvir
9. S900007160
| Molecular Weight | 1412.5 g/mol |
|---|---|
| Molecular Formula | C71H83FN11O17P |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 21 |
| Rotatable Bond Count | 24 |
| Exact Mass | 1411.56900538 g/mol |
| Monoisotopic Mass | 1411.56900538 g/mol |
| Topological Polar Surface Area | 346 Ų |
| Heavy Atom Count | 101 |
| Formal Charge | 0 |
| Complexity | 2600 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 12 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Epclusa is indicated for the treatment of chronic hepatitis C virus (HCV) infection in patients 3 years of age and older (see sections 4. 2, 4. 4 and 5. 1).
Antiviral Agents
Agents used in the prophylaxis or therapy of VIRUS DISEASES. Some of the ways they may act include preventing viral replication by inhibiting viral DNA polymerase; binding to specific cell-surface receptors and inhibiting viral penetration or uncoating; inhibiting viral protein synthesis; or blocking late stages of virus assembly. (See all compounds classified as Antiviral Agents.)
J05A
J - Antiinfectives for systemic use
J05 - Antivirals for systemic use
J05A - Direct acting antivirals
J05AP - Antivirals for treatment of hcv infections
J05AP55 - Sofosbuvir and velpatasvir
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
