

Synopsis
Synopsis
0
KDMF
0
VMF
0
Australia

0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
Annual Reports
NA
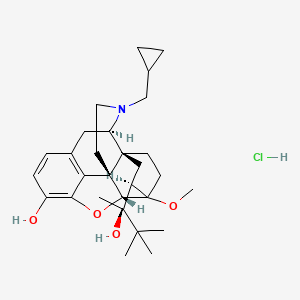

1. 6029 M
2. 6029-m
3. 6029m
4. Buprenex
5. Buprenorphine
6. Buprenorphine Hydrochloride
7. Buprex
8. Hydrochloride, Buprenorphine
9. Prefin
10. Rx 6029 M
11. Rx-6029-m
12. Rx6029m
13. Subutex
14. Temgsic
15. Temgesic
1. Buprenorphine Hydrochloride
2. Ncgc00247733-01
3. Dsstox_cid_28831
4. Dsstox_rid_83100
5. Dsstox_gsid_48905
6. Tox21_112899
7. Cas-53152-21-9
| Molecular Weight | 504.1 g/mol |
|---|---|
| Molecular Formula | C29H42ClNO4 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 5 |
| Exact Mass | 503.2802365 g/mol |
| Monoisotopic Mass | 503.2802365 g/mol |
| Topological Polar Surface Area | 62.2 Ų |
| Heavy Atom Count | 35 |
| Formal Charge | 0 |
| Complexity | 869 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 6 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Buprenorphine hydrochloride |
| Drug Label | Buprenorphine hydrochloride sublingual tablets contains buprenorphine HCl.Buprenorphine is a partial agonist at the mu-opioid receptor and an antagonist at the kappa-opioid receptor. Buprenorphine is a Schedule III narcotic under the Controlled Subst... |
| Active Ingredient | Buprenorphine hydrochloride |
| Dosage Form | Injectable; Tablet |
| Route | Sublingual; Injection |
| Strength | eq 2mg base; eq 0.3mg base/ml; eq 8mg base |
| Market Status | Prescription |
| Company | Hospira; Ethypharm; Luitpold; Eurohlth Intl; Roxane; Barr |
| 2 of 2 | |
|---|---|
| Drug Name | Buprenorphine hydrochloride |
| Drug Label | Buprenorphine hydrochloride sublingual tablets contains buprenorphine HCl.Buprenorphine is a partial agonist at the mu-opioid receptor and an antagonist at the kappa-opioid receptor. Buprenorphine is a Schedule III narcotic under the Controlled Subst... |
| Active Ingredient | Buprenorphine hydrochloride |
| Dosage Form | Injectable; Tablet |
| Route | Sublingual; Injection |
| Strength | eq 2mg base; eq 0.3mg base/ml; eq 8mg base |
| Market Status | Prescription |
| Company | Hospira; Ethypharm; Luitpold; Eurohlth Intl; Roxane; Barr |
Sixmo is indicated for substitution treatment for opioid dependence in clinically stable adult patients who require no more than 8 mg/day of sublingual buprenorphine, within a framework of medical, social and psychological treatment.
Treatment of opioid dependence
Analgesics, Opioid
Compounds with activity like OPIATE ALKALOIDS, acting at OPIOID RECEPTORS. Properties include induction of ANALGESIA or NARCOSIS. (See all compounds classified as Analgesics, Opioid.)
Narcotic Antagonists
Agents inhibiting the effect of narcotics on the central nervous system. (See all compounds classified as Narcotic Antagonists.)
Narcotics
Agents that induce NARCOSIS. Narcotics include agents that cause somnolence or induced sleep (STUPOR); natural or synthetic derivatives of OPIUM or MORPHINE or any substance that has such effects. They are potent inducers of ANALGESIA and OPIOID-RELATED DISORDERS. (See all compounds classified as Narcotics.)
N07BC01
Date of Issue : 2018-01-09
Valid Till : 2019-01-09
Written Confirmation Number : 665/131411
Address of the Firm :
 Faran Shimi: Leading producer of high-quality APIs & alkaloid opiates, serving major pharmaceutical companies across the Middle East.
Faran Shimi: Leading producer of high-quality APIs & alkaloid opiates, serving major pharmaceutical companies across the Middle East.
 Noramco is a partner to the pharmaceutical industry for controlled substance development & manufacturing.
Noramco is a partner to the pharmaceutical industry for controlled substance development & manufacturing.

GDUFA
DMF Review : Reviewed
Rev. Date : 2012-11-23
Pay. Date : 2012-11-23
DMF Number : 23683
Submission : 2010-04-05
Status : Active
Type : II

Certificate Number : R1-CEP 2010-142 - Rev 05
Issue Date : 2023-08-23
Type : Chemical
Substance Number : 1181
Status : Valid

NDC Package Code : 51634-0608
Start Marketing Date : 2017-07-26
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
 SCI Pharmtech offers high-quality, cost-effective APIs, advanced intermediates, & custom products with global expertise and precision.
SCI Pharmtech offers high-quality, cost-effective APIs, advanced intermediates, & custom products with global expertise and precision.
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Certificate Number : R1-CEP 2010-291 - Rev 00
Issue Date : 2016-04-11
Type : Chemical
Substance Number : 1181
Status : Withdrawn by Holder

GDUFA
DMF Review : Reviewed
Rev. Date : 2012-12-26
Pay. Date : 2012-11-30
DMF Number : 19109
Submission : 2006-01-12
Status : Active
Type : II

Certificate Number : R1-CEP 2002-168 - Rev 07
Issue Date : 2019-09-25
Type : Chemical
Substance Number : 1181
Status : Valid

Registration Number : 218MF11027
Registrant's Address : 10 Wheatfield Road, Edinburgh EH11 2QA, United Kingdom
Initial Date of Registration : 2006-12-28
Latest Date of Registration : --

NDC Package Code : 12707-035
Start Marketing Date : 2017-12-14
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT

| Available Reg Filing : ROW |

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 7099
Submission : 1987-08-06
Status : Active
Type : II

Certificate Number : R1-CEP 2017-228 - Rev 00
Issue Date : 2023-01-23
Type : Chemical
Substance Number : 1181
Status : Valid
Date of Issue : 2021-08-27
Valid Till : 2024-06-10
Written Confirmation Number : WC-0278
Address of the Firm :
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
