
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. 1,2 Ethanedicarboxylic Acid
2. 1,2-ethanedicarboxylic Acid
3. 1,4 Butanedioic Acid
4. 1,4-butanedioic Acid
5. Ammonium Succinate
6. Butanedioic Acid
7. Succinate
8. Succinate, Ammonium
1. Butanedioic Acid
2. 110-15-6
3. Amber Acid
4. Wormwood Acid
5. Asuccin
6. Dihydrofumaric Acid
7. Katasuccin
8. 1,2-ethanedicarboxylic Acid
9. Bernsteinsaure
10. Ethylenesuccinic Acid
11. 1,4-butanedioic Acid
12. Wormwood
13. Succinicum Acidum
14. Butandisaeure
15. Acidum Succinicum
16. Butanedionic Acid
17. Kyselina Jantarova
18. Butane Diacid
19. Ethylene Dicarboxylic Acid
20. Spirit Of Amber
21. Bernsteinsaure [german]
22. Kyselina Jantarova [czech]
23. Ammonium Succinate
24. Hsdb 791
25. Succinic-acid
26. Mfcd00002789
27. Succ
28. Nsc 106449
29. Ai3-06297
30. Ab6mnq6j6l
31. Nsc-106449
32. Butanedioic Acid, Homopolymer
33. E363
34. Fema No. 4719
35. Chebi:15741
36. C4-beta-polymorph
37. Nsc25949
38. Succinicacid(industrialgrade&foodgrade)
39. Ncgc00159372-02
40. Ncgc00159372-04
41. Succinellite
42. Acide Succinique
43. Sal Succini
44. Acid Of Amber
45. Dsstox_cid_3602
46. Wln: Qv2vq
47. Dsstox_rid_77102
48. Dsstox_gsid_23602
49. 26776-24-9
50. Sin
51. Ethylene Succinic Acid
52. Ethanedicarboxylic Acid
53. Bernsteinsaeure
54. Sodium Succinate (anhydrous)
55. Succinate, 9
56. Acide Butanedioique
57. Cas-110-15-6
58. Succinic Acid [nf]
59. Succinic Acid (8ci)
60. Butanedioic Acid (9ci)
61. Einecs 203-740-4
62. Unii-ab6mnq6j6l
63. Brn 1754069
64. Dihydrofumarate
65. Succinicate
66. Butanedioic Acid Diammonium Salt
67. Salt Of Amber
68. 1cze
69. Nat.succinic Acid
70. 1,4-butanedioate
71. Succinic Acid, 6
72. Succinic Acid, Fcc
73. Succinic Acide,(s)
74. Succinic Acid (sa)
75. 1,4-butandioic Acid
76. Succinic Acid, 99%
77. Succinic Acid, Natural
78. 4lh2
79. 1,2-ethanedicarboxylate
80. Substrate Analogue, 11
81. Suc
82. Succinic Acid, Acs Grade
83. Bmse000183
84. Bmse000968
85. Chembl576
86. Ec 203-740-4
87. Succinic Acid [ii]
88. Succinic Acid [mi]
89. Hooc-ch2-ch2-cooh
90. Succinic Acid [fcc]
91. A 12084
92. Succinic Acid [hsdb]
93. Succinic Acid [inci]
94. 4-02-00-01908 (beilstein Handbook Reference)
95. Succinic Acid [vandf]
96. Succinic Acid [mart.]
97. Gtpl3637
98. Succinic Acid [usp-rs]
99. Succinic Acid [who-dd]
100. Dtxsid6023602
101. Succinicum Acidum [hpus]
102. Bdbm26121
103. Succinic Acid (butanedioic Acid)
104. Hms3885o04
105. Zinc895030
106. Hy-n0420
107. Str02803
108. Tox21_111612
109. Tox21_201918
110. Tox21_303247
111. Bbl002473
112. Lmfa01170043
113. Nsc-25949
114. Nsc106449
115. S3791
116. Stk387105
117. Succinic Acid [usp Impurity]
118. Succinic Acid, >=99%, Fcc, Fg
119. Succinic Acid, Bioxtra, >=99.0%
120. Akos000118899
121. Tox21_111612_1
122. Ccg-266069
123. Db00139
124. Ncgc00159372-03
125. Ncgc00159372-05
126. Ncgc00159372-06
127. Ncgc00257092-01
128. Ncgc00259467-01
129. Succinic Acid, Acs Reagent, >=99.0%
130. Bp-21128
131. Succinic Acid, Reagentplus(r), >=99.0%
132. Adipic Acid Impurity B [ep Impurity]
133. Cs-0008946
134. Ft-0652509
135. Ft-0773657
136. N1941
137. S0100
138. Succinic Acid 100 Microg/ml In Acetonitrile
139. Succinic Acid, P.a., Acs Reagent, 99.0%
140. Succinic Acid, Saj First Grade, >=99.0%
141. Succinic Acid High Purity Grade 2.5kg
142. Succinic Acid, Purum P.a., >=99.0% (t)
143. Succinic Acid, Saj Special Grade, >=99.5%
144. 1,4-butanedioic Acid (succinic Acid)
145. A14596
146. C00042
147. D85169
148. Succinic Acid, Vetec(tm) Reagent Grade, 98%
149. Ab01332192-02
150. Q213050
151. Sr-01000944556
152. J-002386
153. Sr-01000944556-2
154. Z57127453
155. F2191-0239
156. 37e8fffb-70da-4399-b724-476bd8715ef0
157. Succinic Acid, Certified Reference Material, Tracecert(r)
158. Succinic Acid, Puriss. P.a., Acs Reagent, >=99.5% (t)
159. Succinic Acid, United States Pharmacopeia (usp) Reference Standard
160. Succinic Acid, Matrix Substance For Maldi-ms, >=99.5% (t), Ultra Pure
161. Succinic Acid, Anhydrous, Free-flowing, Redi-dri(tm), Acs Reagent, >=99.0%
162. Succinic Acid, Bioreagent, Suitable For Cell Culture, Suitable For Insect Cell Culture
163. Succinic Acid, Pharmaceutical Secondary Standard; Certified Reference Material
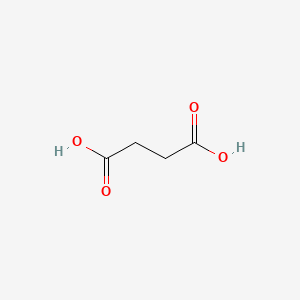
| Molecular Weight | 118.09 g/mol |
|---|---|
| Molecular Formula | C4H6O4 |
| XLogP3 | -0.6 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Exact Mass | 118.02660867 g/mol |
| Monoisotopic Mass | 118.02660867 g/mol |
| Topological Polar Surface Area | 74.6 Ų |
| Heavy Atom Count | 8 |
| Formal Charge | 0 |
| Complexity | 92.6 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
/EXPTL THER/ Succinic acid (100 mM) significantly inhibited systemic anaphylaxis induced by compound 48/80 /a potent mast cell degranulator/ in mice and dose-dependently inhibited local anaphylaxis activated by anti-dinitrophenyl IgE. Further 10 and 100 mM significantly inhibited histamine release from rat peritoneal mast cells activated by compound 48/80 or anti-dinitrophenyl IgE. In addition succinic acid (0.1 and 1 mM) had a significant inhibitory effect on anti-dinitrophenyl IgE-induced tumor necrosis factor-alpha secretion from rat peritoneal mast cells. The level of cyclic AMP in rat peritoneal mast cells, when succinic acid (100 mM) was added, transiently and significantly increased about 4 times compared with that of basal cells. These results suggest a possible use of succinic acid in managing mast cell-dependent anaphylaxis.
Kim HM et al;
For nutritional supplementation, also for treating dietary shortage or imbalance
Succinic acid occurs normally in human urine (1.9-8.8 mg/L).
Bingham, E.; Cohrssen, B.; Powell, C.H.; Patty's Toxicology Volumes 1-9 5th ed. John Wiley & Sons. New York, N.Y. (2001)., p. V5 758
Succinic acid is a normal intermediary metabolite and a constituent of the citric acid cycle. It is readily metabolized when administered to animals, but may be partly excreted unchanged in the urine if large doses are fed.
Bingham, E.; Cohrssen, B.; Powell, C.H.; Patty's Toxicology Volumes 1-9 5th ed. John Wiley & Sons. New York, N.Y. (2001)., p. V5 757
Succinate is an essential component of the Krebs or citric acid cycle and serves an electron donor in the production of fumaric acid and FADH2. It also has been shown to be a good "natural" antibiotic because of its relative acidic or caustic nature (high concentrations can even cause burns). Succinate supplements have been shown to help reduce the effects of hangovers by activating the degradation of acetaldehyde - a toxic byproduct of alcohol metabolism - into CO2 and H2O through aerobic metabolism. Succinic acid has been shown to stimulate neural system recovery and bolster the immune system. Claims have also been made that it boosts awareness, concentration and reflexes.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Excipients by Applications
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
45
PharmaCompass offers a list of Succinic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Succinic Acid manufacturer or Succinic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Succinic Acid manufacturer or Succinic Acid supplier.
PharmaCompass also assists you with knowing the Succinic Acid API Price utilized in the formulation of products. Succinic Acid API Price is not always fixed or binding as the Succinic Acid Price is obtained through a variety of data sources. The Succinic Acid Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Succinic Acid manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Succinic Acid, including repackagers and relabelers. The FDA regulates Succinic Acid manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Succinic Acid API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Succinic Acid manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Succinic Acid supplier is an individual or a company that provides Succinic Acid active pharmaceutical ingredient (API) or Succinic Acid finished formulations upon request. The Succinic Acid suppliers may include Succinic Acid API manufacturers, exporters, distributors and traders.
click here to find a list of Succinic Acid suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
Succinic Acid Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Succinic Acid GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Succinic Acid GMP manufacturer or Succinic Acid GMP API supplier for your needs.
A Succinic Acid CoA (Certificate of Analysis) is a formal document that attests to Succinic Acid's compliance with Succinic Acid specifications and serves as a tool for batch-level quality control.
Succinic Acid CoA mostly includes findings from lab analyses of a specific batch. For each Succinic Acid CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Succinic Acid may be tested according to a variety of international standards, such as European Pharmacopoeia (Succinic Acid EP), Succinic Acid JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Succinic Acid USP).
