

Synopsis
Synopsis
0
VMF
0
Australia

0
South Africa

0
EDQM
0
USP
0
JP
0
Others
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Gtpl7055
2. Q420394
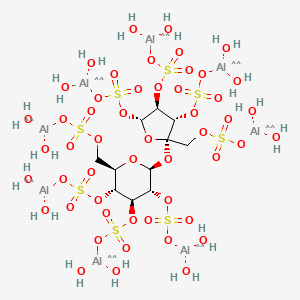
3. [({[(2s,3r,4s,5r,6r)-4,5-bis({[(dihydroxyalumanyl)oxy]sulfonyl}oxy)-6-[({[(dihydroxyalumanyl)oxy]sulfonyl}oxy)methyl]-2-{[(2r,3s,4s,5r)-3,4,5-tris({[(dihydroxyalumanyl)oxy]sulfonyl}oxy)-2-[({[(dihydroxyalumanyl)oxy]sulfonyl}oxy)methyl]oxolan-2-yl]oxy}oxan-3-yl]oxy}sulfonyl)oxy]alumanediol
| Molecular Weight | 1464.8 g/mol |
|---|---|
| Molecular Formula | C11H44Al8O51S8 |
| Hydrogen Bond Donor Count | 16 |
| Hydrogen Bond Acceptor Count | 51 |
| Rotatable Bond Count | 36 |
| Exact Mass | 1463.713824 g/mol |
| Monoisotopic Mass | 1463.713824 g/mol |
| Topological Polar Surface Area | 532 Ų |
| Heavy Atom Count | 78 |
| Formal Charge | 0 |
| Complexity | 2400 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 9 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 17 |
| 1 of 2 | |
|---|---|
| Drug Name | Carafate |
| PubMed Health | Sucralfate (By mouth) |
| Drug Classes | Antiulcer, Protectant |
| Drug Label | CARAFATE Suspension contains sucralfate and sucralfate is an -D-glucopyranoside, -D-fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex.CARAFATE Suspension for oral administration contains 1 g of sucralfate per 10 mL.CARAFATE Sus... |
| Active Ingredient | Sucralfate |
| Dosage Form | Tablet; Suspension |
| Route | Oral |
| Strength | 1gm/10ml; 1gm |
| Market Status | Prescription |
| Company | Forest Labs |
| 2 of 2 | |
|---|---|
| Drug Name | Carafate |
| PubMed Health | Sucralfate (By mouth) |
| Drug Classes | Antiulcer, Protectant |
| Drug Label | CARAFATE Suspension contains sucralfate and sucralfate is an -D-glucopyranoside, -D-fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex.CARAFATE Suspension for oral administration contains 1 g of sucralfate per 10 mL.CARAFATE Sus... |
| Active Ingredient | Sucralfate |
| Dosage Form | Tablet; Suspension |
| Route | Oral |
| Strength | 1gm/10ml; 1gm |
| Market Status | Prescription |
| Company | Forest Labs |
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
 Rochem, your partner in developing, sourcing, and supplying pharmaceutical & animal health ingredients of Chinese origin.
Rochem, your partner in developing, sourcing, and supplying pharmaceutical & animal health ingredients of Chinese origin.

GDUFA
DMF Review : Reviewed
Rev. Date : 2019-04-08
Pay. Date : 2019-01-24
DMF Number : 33479
Submission : 2019-01-25
Status : Active
Type : II
Date of Issue : 2021-01-08
Valid Till : 2023-11-23
Written Confirmation Number : WC-0310
Address of the Firm :

NDC Package Code : 66039-946
Start Marketing Date : 2019-01-25
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : Reviewed
Rev. Date : 2022-10-18
Pay. Date : 2022-09-28
DMF Number : 37502
Submission : 2022-09-30
Status : Active
Type : II

NDC Package Code : 66039-946
Start Marketing Date : 2019-01-25
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : Reviewed
Rev. Date : 2014-04-18
Pay. Date : 2014-04-09
DMF Number : 16589
Submission : 2003-05-13
Status : Active
Type : II

NDC Package Code : 55512-0002
Start Marketing Date : 2009-07-30
End Marketing Date : 2025-12-31
Dosage Form (Strength) : POWDER (50kg/50kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : Reviewed
Rev. Date : 2016-07-05
Pay. Date : 2015-12-18
DMF Number : 30056
Submission : 2016-02-03
Status : Active
Type : II

Certificate Number : R0-CEP 2019-185 - Rev 01
Issue Date : 2023-08-18
Type : Chemical
Substance Number : 1796
Status : Valid

NDC Package Code : 69984-001
Start Marketing Date : 2016-01-19
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Kukjeon Pharmaceutical Co., Ltd.
Registration Date : 2019-04-22
Registration Number : 20190211-211-J-310(2)
Manufacturer Name : Zhejiang Haisen Pharmaceutical Co., Ltd
Manufacturer Address : Xiangtan Village, Liushi Street, Dongyang City, Zhejiang Province, China.


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 3542
Submission : 1979-06-12
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 11876
Submission : 1996-02-28
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 5825
Submission : 1985-05-14
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 7552
Submission : 1988-06-21
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 6108
Submission : 1985-10-11
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 3542
Submission : 1979-06-12
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 7282
Submission : 1988-01-13
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 6048
Submission : 1985-09-27
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 7160
Submission : 1987-09-25
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 7552
Submission : 1988-06-21
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 5825
Submission : 1985-05-14
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 6331
Submission : 1985-10-25
Status : Inactive
Type : II

USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 6057
Submission : 1985-09-05
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 7186
Submission : 1987-11-03
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

ABOUT THIS PAGE
We have 14 companies offering Sucralfate
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
