

Synopsis
Synopsis
0
VMF
0
Australia

0
South Africa

0
EDQM
0
USP
0
JP
0
Others
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Gtpl7055
2. Q420394
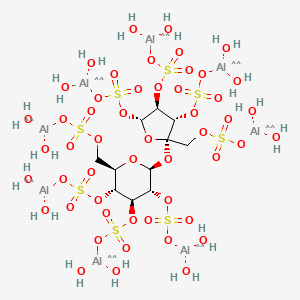
3. [({[(2s,3r,4s,5r,6r)-4,5-bis({[(dihydroxyalumanyl)oxy]sulfonyl}oxy)-6-[({[(dihydroxyalumanyl)oxy]sulfonyl}oxy)methyl]-2-{[(2r,3s,4s,5r)-3,4,5-tris({[(dihydroxyalumanyl)oxy]sulfonyl}oxy)-2-[({[(dihydroxyalumanyl)oxy]sulfonyl}oxy)methyl]oxolan-2-yl]oxy}oxan-3-yl]oxy}sulfonyl)oxy]alumanediol
| Molecular Weight | 1464.8 g/mol |
|---|---|
| Molecular Formula | C11H44Al8O51S8 |
| Hydrogen Bond Donor Count | 16 |
| Hydrogen Bond Acceptor Count | 51 |
| Rotatable Bond Count | 36 |
| Exact Mass | 1463.713824 g/mol |
| Monoisotopic Mass | 1463.713824 g/mol |
| Topological Polar Surface Area | 532 Ų |
| Heavy Atom Count | 78 |
| Formal Charge | 0 |
| Complexity | 2400 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 9 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 17 |
| 1 of 2 | |
|---|---|
| Drug Name | Carafate |
| PubMed Health | Sucralfate (By mouth) |
| Drug Classes | Antiulcer, Protectant |
| Drug Label | CARAFATE Suspension contains sucralfate and sucralfate is an -D-glucopyranoside, -D-fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex.CARAFATE Suspension for oral administration contains 1 g of sucralfate per 10 mL.CARAFATE Sus... |
| Active Ingredient | Sucralfate |
| Dosage Form | Tablet; Suspension |
| Route | Oral |
| Strength | 1gm/10ml; 1gm |
| Market Status | Prescription |
| Company | Forest Labs |
| 2 of 2 | |
|---|---|
| Drug Name | Carafate |
| PubMed Health | Sucralfate (By mouth) |
| Drug Classes | Antiulcer, Protectant |
| Drug Label | CARAFATE Suspension contains sucralfate and sucralfate is an -D-glucopyranoside, -D-fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex.CARAFATE Suspension for oral administration contains 1 g of sucralfate per 10 mL.CARAFATE Sus... |
| Active Ingredient | Sucralfate |
| Dosage Form | Tablet; Suspension |
| Route | Oral |
| Strength | 1gm/10ml; 1gm |
| Market Status | Prescription |
| Company | Forest Labs |
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
 Rochem, your partner in developing, sourcing, and supplying pharmaceutical & animal health ingredients of Chinese origin.
Rochem, your partner in developing, sourcing, and supplying pharmaceutical & animal health ingredients of Chinese origin.

GDUFA
DMF Review : Reviewed
Rev. Date : 2019-04-08
Pay. Date : 2019-01-24
DMF Number : 33479
Submission : 2019-01-25
Status : Active
Type : II
Date of Issue : 2021-01-08
Valid Till : 2023-11-23
Written Confirmation Number : WC-0310
Address of the Firm :

NDC Package Code : 66039-946
Start Marketing Date : 2019-01-25
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : Reviewed
Rev. Date : 2022-10-18
Pay. Date : 2022-09-28
DMF Number : 37502
Submission : 2022-09-30
Status : Active
Type : II

NDC Package Code : 66039-946
Start Marketing Date : 2019-01-25
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : Reviewed
Rev. Date : 2014-04-18
Pay. Date : 2014-04-09
DMF Number : 16589
Submission : 2003-05-13
Status : Active
Type : II

NDC Package Code : 55512-0002
Start Marketing Date : 2009-07-30
End Marketing Date : 2025-12-31
Dosage Form (Strength) : POWDER (50kg/50kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : Reviewed
Rev. Date : 2016-07-05
Pay. Date : 2015-12-18
DMF Number : 30056
Submission : 2016-02-03
Status : Active
Type : II

Certificate Number : R0-CEP 2019-185 - Rev 01
Issue Date : 2023-08-18
Type : Chemical
Substance Number : 1796
Status : Valid

NDC Package Code : 69984-001
Start Marketing Date : 2016-01-19
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Kukjeon Pharmaceutical Co., Ltd.
Registration Date : 2019-04-22
Registration Number : 20190211-211-J-310(2)
Manufacturer Name : Zhejiang Haisen Pharmaceutical Co., Ltd
Manufacturer Address : Xiangtan Village, Liushi Street, Dongyang City, Zhejiang Province, China.


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 3542
Submission : 1979-06-12
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 11876
Submission : 1996-02-28
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 5825
Submission : 1985-05-14
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 7552
Submission : 1988-06-21
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 6108
Submission : 1985-10-11
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 3542
Submission : 1979-06-12
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 7282
Submission : 1988-01-13
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 6048
Submission : 1985-09-27
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 7160
Submission : 1987-09-25
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 7552
Submission : 1988-06-21
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 5825
Submission : 1985-05-14
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 6331
Submission : 1985-10-25
Status : Inactive
Type : II

USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 6057
Submission : 1985-09-05
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 7186
Submission : 1987-11-03
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
