

Synopsis
Synopsis
0
CEP/COS
0
VMF
DRUG PRODUCT COMPOSITIONS
0
Weekly News Recap #Phispers
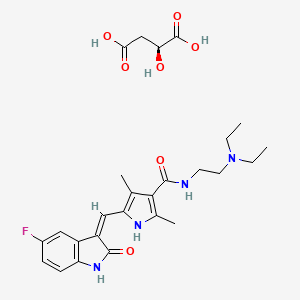

1. 5-(5-fluoro-2-oxo-1,2-dihydroindolylidenemethyl)-2,4-dimethyl-1h-pyrrole-3-carboxylic Acid (2-diethylaminoethyl)amide
2. Su 011248
3. Su 11248
4. Su-011248
5. Su-11248
6. Su011248
7. Su11248
8. Sunitinib
9. Sutent
1. 341031-54-7
2. Sutent
3. Su011248 L-malate Salt
4. Sunitinib Malate [usan]
5. Pha-290940ad
6. Su010398
7. Sunitinib L-malate
8. Su-011248 L-malate Salt
9. Lvx8n1ut73
10. Su-010398
11. (z)-n-(2-(diethylamino)ethyl)-5-((5-fluoro-2-oxoindolin-3-ylidene)methyl)-2,4-dimethyl-1h-pyrrole-3-carboxamide (s)-2-hydroxysuccinate
12. Su 11248
13. N-[2-(diethylamino)ethyl]-5-[(z)-(5-fluoro-2-oxo-1h-indol-3-ylidene)methyl]-2,4-dimethyl-1h-pyrrole-3-carboxamide;(2s)-2-hydroxybutanedioic Acid
14. Butanedioic Acid, Hydroxy-, (2s)-, Compd. With N-(2-(diethylamino)ethyl)-5-((z)-(5-fluoro-1,2-dihydro-2-oxo-3h-indol-3-ylidene)methyl)-2,4-dimethyl-1h-pyrrole-3-carboxamide (1:1)
15. N-(2-(diethylamino)ethyl)-5-((z)-(5-fluoro-1,2-dihydro-2-oxo-3h-indol-3-ylidene)methyl)-2,4-dimethyl-1h-pyrrole-3-carboxamide (2s)-hydroxybutanedioate
16. (2s)-2-hydroxybutanedioic Acid; N-[2-(diethylamino)ethyl]-5-{[(3z)-5-fluoro-2-oxo-2,3-dihydro-1h-indol-3-ylidene]methyl}-2,4-dimethyl-1h-pyrrole-3-carboxamide
17. Butanedioic Acid, 2-hydroxy-, (2s)-, Compd. With N-[2-(diethylamino)ethyl]-5-[(z)-(5-fluoro-1,2-dihydro-2-oxo-3h-indol-3-ylidene)methyl]-2,4-dimethyl-1h-pyrrole-3-carboxamide (1:1)
18. Su 011248
19. Unii-lvx8n1ut73
20. Butanedioic Acid, 2-hydroxy-, (2s)-, Compd. With N-(2-(diethylamino)ethyl)-5-((z)-(5-fluoro-1,2-dihydro-2-oxo-3h-indol-3-ylidene)methyl)-2,4-dimethyl-1h-pyrrole-3-carboxamide (1:1)
21. Mfcd08282795
22. Pdgf Tk Antagonist
23. Sutent (tn)
24. Sunitinib Malate- Bio-x
25. Sutent (tn) (pfizer)
26. Sunitinib Malate (sutent)
27. Chembl1567
28. Su 11248 (malate)
29. Su-11248 L-malate Salt
30. Sunitinib Malate [mi]
31. Sunitinib Malate (jan/usan)
32. Sunitinib Malate [jan]
33. Schembl1258610
34. Schembl1772213
35. Dtxsid4046492
36. Sunitinib Malate [vandf]
37. Sunitinib Malate [mart.]
38. Pnu-290940ad
39. Sunitinib Malate [who-dd]
40. Hms3261h09
41. Sunitinib Malate, >=98% (hplc)
42. Tox21_500514
43. Nsc736511
44. S1042
45. Su-11248j
46. Akos005145765
47. Akos025149097
48. Sunitinib Malate (sunitinib L-malate)
49. Sunitinib Malate [orange Book]
50. Ccg-221818
51. Cs-0177
52. Ks-5023
53. Ncgc00261199-01
54. Bs164428
55. Hy-10255
56. Am20090634
57. Sw219407-1
58. D06402
59. 031s547
60. A822079
61. J-019449
62. Q27283213
63. Sunitinib Malate
64. 341031-54-7
65. Sutent
66. Su-11248
67. Sunitinib
68. (2s)-2-hydroxybutanedioic Acid Compd. With N-[2-(diethylamino)ethyl]-5-[(z)-(5-fluoro-1,2-dihydro-2-oxo-3h-indol-3-ylidene)methyl]-2,4-dimethyl-1h-pyrrole-3-carboxamide (1:1)
69. (z)-n-(2-(diethylamino)ethyl)-5-((5-fluoro-2-oxoindolin-3-ylidene)methyl)-2,4-dimethyl-1h-pyrrole-3-carboxamide(s)-2-hydroxysuccinate
70. 1h-pyrrole-3-carboxamide, N-(2-(diethylamino)ethyl)-5-((z)-(5-fluoro-1,2-dihydro-2-oxo-3h-indol-3-ylidene)methyl)-2,4-dimethyl-, (2s)-hydroxybutanedioate (1:1)
71. 1h-pyrrole-3-carboxamide,2-dihydro-2-oxo-3h-indol-3-ylidene)methyl]-2,4-dimethyl-, (2s)-hydroxybutanedioate (1:1)
72. 1h-pyrrole-3-carboxamide,2-dihydro-2-oxo-3h-indol-3-ylidene)methyl]-2,4-dimethyl-, Malate Salt
73. Butanedioic Acid, (2s)-, Compd. With N-[2-(diethylamino)ethyl]-5-[(z)-(5-fluoro-1,2-dihydro-2-oxo-3h-indol-3-ylidene)methyl]-2,4-dimethyl-1h-pyrrole-3-carboxamide (1:1)
74. Butanedioic Acid, (2s)-, Compd. With N-[2-(diethylamino)ethyl]-5-[(z)-(5-fluoro-1,2-dihydro-2-oxo-3h-indol-3-ylidene)methyl]-2,4-dimethyl-1h-pyrrole-3-carboxamide (1:1) (ca Index Name)
75. N-(2-diethylamino)ethyl)-5-((z)-(5-fluoro-2-oxo-1,2-dihydro-3h-indol-3-ylidene)methyl)-2,4-dimethyl-1h-pyrrole-3-carboxamide Hydrogen (2s)-2-hydroxybutanedioate
76. N-[2-(diethylamino)ethyl]-5-[(z)-(5-fluoro-1,2-dihydro-2-oxo-3h-indol-3-ylidine)methyl]-2,4-dimethyl-1h-pyrrole-3-carboxamide (2s)-2-hydroxybutanedioate Salt
| Molecular Weight | 532.6 g/mol |
|---|---|
| Molecular Formula | C26H33FN4O7 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 10 |
| Exact Mass | 532.23332757 g/mol |
| Monoisotopic Mass | 532.23332757 g/mol |
| Topological Polar Surface Area | 172 Ų |
| Heavy Atom Count | 38 |
| Formal Charge | 0 |
| Complexity | 765 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Sunitinib malate |
| Drug Label | SUTENT, an oral multi-kinase inhibitor, is the malate salt of sunitinib. Sunitinib malate is described chemically as Butanedioic acid, hydroxy-, (2S)-, compound with N-[2-(diethylamino)ethyl]-5-[(Z)-(5-fluoro-1,2-dihydro-2-oxo-3H-indol-3-ylidine)meth... |
| Active Ingredient | Sunitinib malate |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | eq 50mg base; eq 12.5mg base; eq 25mg base; eq 37.5mg base |
| Market Status | Prescription |
| Company | Mylan Pharms |
| 2 of 2 | |
|---|---|
| Drug Name | Sunitinib malate |
| Drug Label | SUTENT, an oral multi-kinase inhibitor, is the malate salt of sunitinib. Sunitinib malate is described chemically as Butanedioic acid, hydroxy-, (2S)-, compound with N-[2-(diethylamino)ethyl]-5-[(Z)-(5-fluoro-1,2-dihydro-2-oxo-3H-indol-3-ylidine)meth... |
| Active Ingredient | Sunitinib malate |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | eq 50mg base; eq 12.5mg base; eq 25mg base; eq 37.5mg base |
| Market Status | Prescription |
| Company | Mylan Pharms |
* Gastrointestinal stromal tumour (GIST):
Sutent is indicated for the treatment of unresectable and/or metastatic malignant gastrointestinal stromal tumour (GIST) in adults after failure of imatinib mesilate treatment due to resistance or intolerance.
* Metastatic renal cell carcinoma (MRCC):
Sutent is indicated for the treatment of advanced/metastatic renal cell carcinoma (MRCC) in adults.
* Pancreatic neuroendocrine tumours (pNET):
Sutent is indicated for the treatment of unresectable or metastatic, well-differentiated pancreatic neuroendocrine tumours with disease progression in adults.
Experience with Sutent as first-line treatment is limited (see section 5. 1).
Angiogenesis Inhibitors
Agents and endogenous substances that antagonize or inhibit the development of new blood vessels. (See all compounds classified as Angiogenesis Inhibitors.)
Antineoplastic Agents
Substances that inhibit or prevent the proliferation of NEOPLASMS. (See all compounds classified as Antineoplastic Agents.)
Protein Kinase Inhibitors
Agents that inhibit PROTEIN KINASES. (See all compounds classified as Protein Kinase Inhibitors.)
L01EX01
 Medichem is a vertically integrated pharmaceutical company specializing in the development & manufacturing of APIs & FDFs.
Medichem is a vertically integrated pharmaceutical company specializing in the development & manufacturing of APIs & FDFs.
NDC Package Code : 16436-0091
Start Marketing Date : 2010-06-04
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (75kg/75kg)
Marketing Category : BULK INGREDIENT
 Medichem is a vertically integrated pharmaceutical company specializing in the development & manufacturing of APIs & FDFs.
Medichem is a vertically integrated pharmaceutical company specializing in the development & manufacturing of APIs & FDFs.
NDC Package Code : 53296-0091
Start Marketing Date : 2010-06-02
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (75kg/75kg)
Marketing Category : BULK INGREDIENT
NDC Package Code : 68554-0078
Start Marketing Date : 2006-01-26
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 54893-0081
Start Marketing Date : 2018-11-28
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 68724-1234
Start Marketing Date : 2019-07-01
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 82920-037
Start Marketing Date : 2023-09-27
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 53183-4012
Start Marketing Date : 2018-03-26
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (100kg/100kg)
Marketing Category : BULK INGREDIENT

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
About the Company : LGM Pharma is a global leader in sourcing hard-to-find APIs and intermediates for the pharmaceutical and biotech industries. LGM is also a full service CDMO providing formulation, ...
 Gonane has API manufacturing expertise in new-age Corticosteroids, Hormones and other pharma raw materials.
Gonane has API manufacturing expertise in new-age Corticosteroids, Hormones and other pharma raw materials.
About the Company : Gonane Pharma, is a contract pharmaceutical company located in Gujarat, India, specializing in the manufacturing and marketing of Corticosteroids, Hormones, Antivirals, and Oncolog...
 Faran Shimi: Leading producer of high-quality APIs & alkaloid opiates, serving major pharmaceutical companies across the Middle East.
Faran Shimi: Leading producer of high-quality APIs & alkaloid opiates, serving major pharmaceutical companies across the Middle East.
About the Company : Faran Shimi Pharmaceutical Company, established in 2001 and affiliated with Golrang Pharmaceutical Investment Co, manufactures high-quality Active Pharmaceutical Ingredients (APIs)...
About the Company : HRV Global is a leading global manufacturer, seller & exporter of a wide range of APIs, advanced intermediates, pellets, food grade chemicals, food additives & food ingredients. It...
 Medichem is a vertically integrated pharmaceutical company specializing in the development & manufacturing of APIs & FDFs.
Medichem is a vertically integrated pharmaceutical company specializing in the development & manufacturing of APIs & FDFs.
About the Company : Medichem is an indepently-owned company based in Barcelona. Founded in 1972 and with more than 45 years, Medichem has been engaged in the process development and manufacture of Act...
About the Company : Established in May 2012, Shandong Loncom Pharmaceutical operates as a fully owned subsidiary of Shandong Bestcomm Pharmaceutical Co., Ltd. Situated in the Qihe Economic Development...
 Tofigh Daru develops & synthesizes a diverse range of APIs in Anticancer, Narcotics, Cardiovascular to Immunomodulatory Segments.
Tofigh Daru develops & synthesizes a diverse range of APIs in Anticancer, Narcotics, Cardiovascular to Immunomodulatory Segments.
About the Company : Tofigh Daru (TODACO) belongs to the Tamin Pharmaceutical Investment Company, the biggest pharmaceutical holding in Iran. TODACO develops novel APIs in therapeutic categories like A...
About the Company : Established in 2011 and situated in Hangzhou, Zhejiang, China, Hangzhou Longshine Bio-Tech CO., Ltd is dedicated to providing services for pharmaceutical and chemical products, cat...

About the Company : Specialità Fine Chemicals was established in 2013, through an initiative of Prati-Donaduzzi Group, aiming at the development of the manufacturing process of Active Pharmaceutical ...

About the Company : Synthland is an integrated pharmaceutical company that develops, produce and supplies Active Pharmaceutical Ingredients (APIs) and Intermediates. We also represent some well-known...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
CGT9486 (bezuclastinib), is a KIT mutant inhibitor, being investigated in combination with sunitinib for patients living with gastrointestinal stromal tumors.
Lead Product(s): Bezuclastinib,Sunitinib
Therapeutic Area: Oncology Brand Name: CGT9486
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable September 03, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Bezuclastinib,Sunitinib
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Cogent Completes Enrollment in Phase 3 PEAK Trial for Gastrointestinal Stromal Tumors
Details : CGT9486 (bezuclastinib), is a KIT mutant inhibitor, being investigated in combination with sunitinib for patients living with gastrointestinal stromal tumors.
Brand Name : CGT9486
Molecule Type : Small molecule
Upfront Cash : Not Applicable
September 03, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
The net proceeds will be used for the clinical development of CGT9486 (bezuclastinib), which is a KIT mutant inhibitor for patients living with systemic mastocytosis and gastrointestinal stromal tumors.
Lead Product(s): Bezuclastinib,Sunitinib
Therapeutic Area: Oncology Brand Name: CGT9486
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: J.P. Morgan
Deal Size: $225.0 million Upfront Cash: Undisclosed
Deal Type: Private Placement February 14, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Bezuclastinib,Sunitinib
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : J.P. Morgan
Deal Size : $225.0 million
Deal Type : Private Placement
Cogent Biosciences Announces Oversubscribed $225 Million Private Placement
Details : The net proceeds will be used for the clinical development of CGT9486 (bezuclastinib), which is a KIT mutant inhibitor for patients living with systemic mastocytosis and gastrointestinal stromal tumors.
Brand Name : CGT9486
Molecule Type : Small molecule
Upfront Cash : Undisclosed
February 14, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Cogent intends to fund the development, regulatory and commercial preparation activities relating to CGT9486 (bezuclastinib), a selective tyrosine kinase inhibitor that is designed to potently inhibit the KIT D816V mutation as well as other mutations in KIT exon 17.
Lead Product(s): Bezuclastinib,Sunitinib
Therapeutic Area: Oncology Brand Name: CGT9486
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: J.P. Morgan
Deal Size: $150.0 million Upfront Cash: Undisclosed
Deal Type: Public Offering June 06, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Bezuclastinib,Sunitinib
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : J.P. Morgan
Deal Size : $150.0 million
Deal Type : Public Offering
Cogent Biosciences Announces Pricing of Upsized Public Offering of Shares of Common Stock
Details : Cogent intends to fund the development, regulatory and commercial preparation activities relating to CGT9486 (bezuclastinib), a selective tyrosine kinase inhibitor that is designed to potently inhibit the KIT D816V mutation as well as other mutations in ...
Brand Name : CGT9486
Molecule Type : Small molecule
Upfront Cash : Undisclosed
June 06, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Cabometyx (cabozantinib) is a multiple receptor tyrosine kinases inhibitor. These receptors are involved in both normal cellular function and pathologic processes such as oncogenesis, metastasis, tumor angiogenesis, maintenance of the tumor microenvironment, etc.
Lead Product(s): Cabozantinib,Nivolumab,Sunitinib
Therapeutic Area: Oncology Brand Name: Cabometyx
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable February 13, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Cabozantinib,Nivolumab,Sunitinib
Therapeutic Area : Oncology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Cabometyx® In Combination with Nivolumab Shows Durable Survival Benefits at Over Three-Years’ F...
Details : Cabometyx (cabozantinib) is a multiple receptor tyrosine kinases inhibitor. These receptors are involved in both normal cellular function and pathologic processes such as oncogenesis, metastasis, tumor angiogenesis, maintenance of the tumor microenvironm...
Brand Name : Cabometyx
Molecule Type : Small molecule
Upfront Cash : Not Applicable
February 13, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
The net proceeds from the offering will be used for development, regulatory and commercial preparation activities relating to CGT9486 (bezuclastinib) and other product candidates, as well as for working capital and general corporate purposes.
Lead Product(s): Bezuclastinib,Sunitinib
Therapeutic Area: Oncology Brand Name: CGT9486
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Jefferies
Deal Size: $172.6 million Upfront Cash: Undisclosed
Deal Type: Public Offering June 16, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Bezuclastinib,Sunitinib
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Jefferies
Deal Size : $172.6 million
Deal Type : Public Offering
Details : The net proceeds from the offering will be used for development, regulatory and commercial preparation activities relating to CGT9486 (bezuclastinib) and other product candidates, as well as for working capital and general corporate purposes.
Brand Name : CGT9486
Molecule Type : Small molecule
Upfront Cash : Undisclosed
June 16, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Cogent intends to use the net proceeds from the offering for development, regulatory and commercial preparation activities relating to CGT9486 (bezuclastinib) and other product candidates, as well as for working capital and general corporate purposes.
Lead Product(s): Bezuclastinib,Sunitinib
Therapeutic Area: Oncology Brand Name: CGT9486
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Jefferies
Deal Size: $150.0 million Upfront Cash: Undisclosed
Deal Type: Public Offering June 14, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Bezuclastinib,Sunitinib
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Jefferies
Deal Size : $150.0 million
Deal Type : Public Offering
Details : Cogent intends to use the net proceeds from the offering for development, regulatory and commercial preparation activities relating to CGT9486 (bezuclastinib) and other product candidates, as well as for working capital and general corporate purposes.
Brand Name : CGT9486
Molecule Type : Small molecule
Upfront Cash : Undisclosed
June 14, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Updated outcomes from trial evaluating combination of Cabometyx (Cabozantinib) plus atezolizumab in an expanded patient population in NSCLC demonstrate encouraging clinical activity with manageable toxicity in people previously treated with an immune checkpoint inhibitor.
Lead Product(s): Cabozantinib,Nivolumab,Sunitinib
Therapeutic Area: Oncology Brand Name: Cabometyx
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable May 26, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Cabozantinib,Nivolumab,Sunitinib
Therapeutic Area : Oncology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Ipsen: ASCO 2022: New Cabometyx® Data Show Encouraging Results in Monotherapy and in Combination ...
Details : Updated outcomes from trial evaluating combination of Cabometyx (Cabozantinib) plus atezolizumab in an expanded patient population in NSCLC demonstrate encouraging clinical activity with manageable toxicity in people previously treated with an immune che...
Brand Name : Cabometyx
Molecule Type : Small molecule
Upfront Cash : Not Applicable
May 26, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
GB-102 is a microparticle formulation of a pan-VEGF inhibitor, sunitinib, for the treatment of wet age-related macular degeneration designed for a twice-per-year intravitreal injection. GB-102 has the potential to also benefit patients with diabetic retinopathy.
Lead Product(s): Sunitinib
Therapeutic Area: Ophthalmology Brand Name: GB-102
Study Phase: Phase IIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable May 19, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Sunitinib
Therapeutic Area : Ophthalmology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Details : GB-102 is a microparticle formulation of a pan-VEGF inhibitor, sunitinib, for the treatment of wet age-related macular degeneration designed for a twice-per-year intravitreal injection. GB-102 has the potential to also benefit patients with diabetic reti...
Brand Name : GB-102
Molecule Type : Small molecule
Upfront Cash : Not Applicable
May 19, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
The MERECA study showed that the combination of ilixadencel with the tyrosine kinase inhibitor sunitinib was safe and associated with a positive trend for improved tumor response rate and overall survival, compared with sunitinib monotherapy (control group).
Lead Product(s): Ilixadencel,Sunitinib
Therapeutic Area: Oncology Brand Name: Intuvax
Study Phase: Phase IIProduct Type: Cell and Gene therapy
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable April 28, 2022

Details : The MERECA study showed that the combination of ilixadencel with the tyrosine kinase inhibitor sunitinib was safe and associated with a positive trend for improved tumor response rate and overall survival, compared with sunitinib monotherapy (control gro...
Brand Name : Intuvax
Molecule Type : Cell and Gene therapy
Upfront Cash : Not Applicable
April 28, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Phase 2 study investigating CGT9486 for the treatment of patients with Advanced Systemic Mastocytosis (AdvSM), including patients with Aggressive SM (ASM), SM with Associated Hematologic Neoplasm (SM-AHN), and Mast Cell Leukemia (MCL).
Lead Product(s): Bezuclastinib,Sunitinib
Therapeutic Area: Oncology Brand Name: CGT9486
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable March 08, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Bezuclastinib,Sunitinib
Therapeutic Area : Oncology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Cogent Biosciences Announces Upcoming Presentations at AACR Annual Meeting 2022
Details : Phase 2 study investigating CGT9486 for the treatment of patients with Advanced Systemic Mastocytosis (AdvSM), including patients with Aggressive SM (ASM), SM with Associated Hematologic Neoplasm (SM-AHN), and Mast Cell Leukemia (MCL).
Brand Name : CGT9486
Molecule Type : Small molecule
Upfront Cash : Not Applicable
March 08, 2022

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Sunitinib Zentiva
Dosage Form : Kaps
Dosage Strength : 12.5mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Sunitinib Zentiva
Dosage Form : Kaps
Dosage Strength : 25mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Sunitinib Zentiva
Dosage Form : Caps
Dosage Strength : 50mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info : RX
Registration Country : USA
Brand Name : SUNITINIB MALATE
Dosage Form : CAPSULE;ORAL
Dosage Strength : EQ 12.5MG BASE
Packaging :
Approval Date : 2022-04-11
Application Number : 215843
Regulatory Info : RX
Registration Country : USA
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info : RX
Registration Country : USA
Brand Name : SUNITINIB MALATE
Dosage Form : CAPSULE;ORAL
Dosage Strength : EQ 25MG BASE
Packaging :
Approval Date : 2022-04-11
Application Number : 215843
Regulatory Info : RX
Registration Country : USA
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info : RX
Registration Country : USA
Brand Name : SUNITINIB MALATE
Dosage Form : CAPSULE;ORAL
Dosage Strength : EQ 37.5MG BASE
Packaging :
Approval Date : 2022-04-11
Application Number : 215843
Regulatory Info : RX
Registration Country : USA
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info : Lead Market Dossiers- Filed
Registration Country : India
Brand Name :
Dosage Form : Oral Solid Dosage Form
Dosage Strength : 12.5MG
Packaging :
Approval Date :
Application Number :
Regulatory Info : Lead Market Dossiers- Filed
Registration Country : India
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info : Lead Market Dossiers- Filed
Registration Country : India
Brand Name :
Dosage Form : Oral Solid Dosage Form
Dosage Strength : 25MG
Packaging :
Approval Date :
Application Number :
Regulatory Info : Lead Market Dossiers- Filed
Registration Country : India
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info : Lead Market Dossiers- Filed
Registration Country : India
Brand Name :
Dosage Form : Oral Solid Dosage Form
Dosage Strength : 37.5MG
Packaging :
Approval Date :
Application Number :
Regulatory Info : Lead Market Dossiers- Filed
Registration Country : India
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info : Lead Market Dossiers- Filed
Registration Country : India
Brand Name :
Dosage Form : Oral Solid Dosage Form
Dosage Strength : 50MG
Packaging :
Approval Date :
Application Number :
Regulatory Info : Lead Market Dossiers- Filed
Registration Country : India
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Excipients by Applications
Global Sales Information

Company :
Sunitinib Malate
Drug Cost (USD) : 55,637,386
Year : 2022
Prescribers : 1179
Prescriptions : 6128

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company :
Sunitinib Malate
Drug Cost (USD) : 17,597,961
Year : 2022
Prescribers : 339
Prescriptions : 1442

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company :
Sunitinib Malate
Drug Cost (USD) : 15,553,046
Year : 2021
Prescribers : 644
Prescriptions : 1600

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company :
Sunitinib Malate
Drug Cost (USD) : 96,954,410
Year : 2021
Prescribers : 1515
Prescriptions : 7602

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company :
Sunitinib Malate
Drug Cost (USD) : 120,222,574
Year : 2020
Prescribers : 1887
Prescriptions : 9953

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company :
Sunitinib Malate
Drug Cost (USD) : 160,489,804
Year : 2019
Prescribers : 2595
Prescriptions : 13790

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company :
Sunitinib Malate
Drug Cost (USD) : 193,354,311
Year : 2018
Prescribers : 3371
Prescriptions : 17143

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company :
Sunitinib Malate
Drug Cost (USD) : 174,929,778
Year : 2017
Prescribers : 3428
Prescriptions : 16340

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company :
Sunitinib Malate
Drug Cost (USD) : 183,064,145
Year : 2016
Prescribers : 3883
Prescriptions : 18549

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company :
Sunitinib Malate
Drug Cost (USD) : 160,910,812
Year : 2015
Prescribers : 3982
Prescriptions : 16992

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
RX/OTC/DISCN : Class A
Dosage Form : Kaps
Dosage Strength : 12.5mg
Price Per Pack (Euro) : 800.81
Published in :
Country : Switzerland
RX/OTC/DISCN : Class A
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
RX/OTC/DISCN : Class A
Dosage Form : Caps
Dosage Strength : 50mg
Price Per Pack (Euro) : 3046.34
Published in :
Country : Switzerland
RX/OTC/DISCN : Class A
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
RX/OTC/DISCN : Class A
Dosage Form : Kaps
Dosage Strength : 25mg
Price Per Pack (Euro) : 1601.5
Published in :
Country : Switzerland
RX/OTC/DISCN : Class A
Dosage Form : Caps
Dosage Strength : 12.5mg
Price Per Pack (Euro) : 889.78
Published in :
Country : Switzerland
RX/OTC/DISCN : Class A

Dosage Form : Caps
Dosage Strength : 25mg
Price Per Pack (Euro) : 1779.44
Published in :
Country : Switzerland
RX/OTC/DISCN : Class A

Dosage Form : Caps
Dosage Strength : 50mg
Price Per Pack (Euro) : 3384.8
Published in :
Country : Switzerland
RX/OTC/DISCN : Class A

Dosage Form : Caps
Dosage Strength : 12.5mg
Price Per Pack (Euro) : 800.81
Published in :
Country : Switzerland
RX/OTC/DISCN : Class A

Dosage Form : Capsule, hard
Dosage Strength : 12.5 mg
Price Per Pack (Euro) : 1,513.02
Published in :
Country : Norway
RX/OTC/DISCN :

Dosage Form : Capsule, hard
Dosage Strength : 25 mg
Price Per Pack (Euro) : 3,021.22
Published in :
Country : Norway
RX/OTC/DISCN :

Dosage Form : Capsule, hard
Dosage Strength : 50 mg
Price Per Pack (Euro) : 6,037.42
Published in :
Country : Norway
RX/OTC/DISCN :

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
We have 8 companies offering Sunitinib Malate
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
