
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
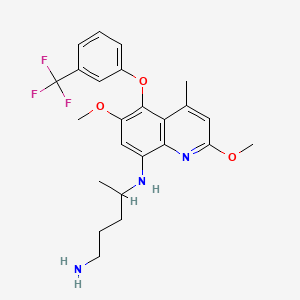

1. (r)-tafenoquine Succinate
2. Arakoda
3. Butanedioic Acid, Compd. With N4-(2,6-dimethoxy-4-methyl-5-(3-(trifluoromethyl)phenoxy)-8-quinolinyl)-1,4-pentanediamine (1:1)
4. Butanedioic Acid, Compd. With N4-(2,6-dimethoxy-4-methyl-5-(3-(trifluoromethyl)phenoxy)-8-quinolinyl)-1,4-pentanediamine (1:1), (r)-
5. Krintafel
6. N(4)-(2,6-dimethoxy-4-methyl-5-((3-trifluoromethyl)phenoxy)-8-quinolinyl)-1,4-pentanediamine
7. Sb-252263-ax
8. Sb-252263ax
9. Sb252263-ax
10. Sb252263ax
11. Tafenoquine Maleate
12. Tafenoquine Succinate
13. Tafenoquine Succinate, (r)-
14. Wr 238605
15. Wr-238605
16. Wr238605
1. 106635-80-7
2. Etaquine
3. Arakoda
4. Krintafel
5. Wr 238605
6. Sb-252263-aab
7. Tafenoquine Free Base
8. Wr238605
9. 262p8gs9l9
10. 106635-80-7 (free Base)
11. 4-n-[2,6-dimethoxy-4-methyl-5-[3-(trifluoromethyl)phenoxy]quinolin-8-yl]pentane-1,4-diamine
12. Tafenoquine (usan)
13. Tafenoquine [usan]
14. N4-(2,6-dimethoxy-4-methyl-5-(3-(trifluoromethyl)phenoxy)quinolin-8-yl)pentane-1,4-diamine
15. 1,4-pentanediamine, N4-(2,6-dimethoxy-4-methyl-5-(3-(trifluoromethyl)phenoxy)-8-quinolinyl)-
16. Sb-252263
17. Unii-262p8gs9l9
18. Tafenoquine [usan:inn:ban]
19. 1,4-pentanediamine, N4-[2,6-dimethoxy-4-methyl-5-[3-(trifluoromethyl)phenoxy]-8-quinolinyl]-
20. N(4)-(2,6-dimethoxy-4-methyl-5-((3-trifluoromethyl)phenoxy)-8-quinolinyl)-1,4-pentanediamine
21. Tafenoquine [mi]
22. Tafenoquine [inn]
23. (r)-n3-(2,6-dimethoxy-4-methyl-5-(3-trifluoromethyl)phenoxy)quinolin-8-yl)pentane-1,4-diamine
24. Tafenoquine [mart.]
25. Tafenoquine [who-dd]
26. Schembl347388
27. Chembl298470
28. Gtpl9722
29. Dtxsid90869466
30. Chebi:141487
31. Ex-a3146
32. Db06608
33. Sb16555
34. (rs)-n(sup 3)-(2,6-dimethoxy-4-methyl-5-(3-trifluoro-methylphenoxy)quinolin-8-yl)pentane-1,4-diamine
35. Hy-111529
36. Cs-0042381
37. Ft-0775258
38. D10490
39. D87160
40. Q2387553
41. N4-[2,6-dimethoxy-4-methyl-5-[3-(trifluoromethyl)phenoxy]-8-quinolyl]pentane-1,4-diamine
42. (+/-)-8-[(4-amino-1-methylbutyl)amino]-2,6-dimethoxy-4-methyl-5-(3-trifluoromethylphenoxy) Quinoline
43. (4-amino-1-methylbutyl){2,6-dimethoxy-4-methyl-5-[3-(trifluoromethyl)phenoxy](8-quinolyl)}amine
44. N(4)-{2,6-dimethoxy-4-methyl-5-[3-(trifluoromethyl)phenoxy]quinolin-8-yl}pentane-1,4-diamine
| Molecular Weight | 463.5 g/mol |
|---|---|
| Molecular Formula | C24H28F3N3O3 |
| XLogP3 | 5.4 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 9 |
| Exact Mass | 463.20827625 g/mol |
| Monoisotopic Mass | 463.20827625 g/mol |
| Topological Polar Surface Area | 78.6 Ų |
| Heavy Atom Count | 33 |
| Formal Charge | 0 |
| Complexity | 597 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Tafenoquine is used for the treatment and prevention of relapse of Vivax malaria in patients 16 years and older. Tafenoquine is not indicated to treat acute vivax malaria. Malaria is a disease that remains to occur in many tropical countries. Vivax malaria, caused by _Plasmodium vivax_, is known to be less virulent and seldom causes death. However, it causes a substantive illness-related burden in endemic areas and it is known to present dormant forms in the hepatocytes named hypnozoites which can remain dormant for weeks or even months. This dormant form produces ongoing relapses.
FDA Label
In vitro studies have shown that tafenoquine presents an average 50% inhibitory concentration of 0.436 mcg against blood stages of seven strains of _P. falciparum_. In chloroquine-resistant _P. falciparum_ strains the IC50 of tafenoquine was greater when compared with primaquine and it ranged from 0.5 to 33.1 mcg. In studies evaluating the transmission-blocking activity of tafenoquine against the sporogonic stage of _P. vivax_, it was showed a reduced transmission at doses higher than 25 mg/kg. In clinical trials, it was reported a tafenoquine-induced relapse prevention of 91.9% in cases of vivax malaria when pretreated with chloroquine. In prophylactic studies, tafenoquine showed an efficacy range from 84 to 87% against falciparum malaria and 99.1% against vivax malaria.
Antimalarials
Agents used in the treatment of malaria. They are usually classified on the basis of their action against plasmodia at different stages in their life cycle in the human. (From AMA, Drug Evaluations Annual, 1992, p1585) (See all compounds classified as Antimalarials.)
P - Antiparasitic products, insecticides and repellents
P01 - Antiprotozoals
P01B - Antimalarials
P01BA - Aminoquinolines
P01BA07 - Tafenoquine
Absorption
The first-in-human pharmacokinetic study showed a tmax of 13.8 hours and this study suggested that the prolonged absorption from the gut can be due to absorption in the distal gastrointestinal tract combined with a slow clearance. The AUC and Cmax demonstrated an intersubject variability. The bioavailability of tafenoquine is increased in the presence of a high-fat meal by modifying the amount of drug absorbed rather than the rate of absorption. Once absorbed, the concentration of tafenoquine in the whole body is two-fold higher than the corresponding concentration in plasma and it seems to be highly distributed in the liver showing an AUC of approximately 80 times more than what is found in the plasma.
Route of Elimination
After degradation by different metabolic pathways, tafenoquine is slowly excreted from the body primarily in the feces and renal elimination of the unchanged form is very low.
Volume of Distribution
Tafenoquine presents a high volume of distribution of approximately 2 560 L.
Clearance
Tafenoquine presents a low clearance of approximately 6 L/h.
The activation of tafenoquine needs the activity of CYP 2D6 liver microsomal enzyme. This activation step produces the metabolite 5,6 ortho quinone tafenoquine. This metabolite is internalized by the parasite and reduced to radicals by ferredoxin-NADP+ reductase and diflavin reductase enzymes. In the human, tafenoquine is metabolized by several metabolic pathways including O-demethylation, N-dealkylation, N-oxidation and oxidative deamination as well as C-hydroxylation of the 8-aminoalkylamino side chain.
Tafenoquine presents a long half-life of approximately 14 days.
The mechanism of action of tafenoquine is not well established but studies have reported a longer and more effective action when compared to primaquine. The active moiety of tafenoquine, 5,6 ortho quinone tafenoquine, seems to be redox cycled by _P. falciparum_ which are upregulated in gametocytes and liver stages. Once inside, the oxidated metabolite produces hydrogen peroxide and hydroxyl radicals. It is thought that these radicals produce leads to the parasite death. On the other hand, tafenoquine inhibits heme polymerase in blood stage of parasites which explains the activity against blood stages of parasites.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Market Place

ABOUT THIS PAGE
11
PharmaCompass offers a list of Tafenoquine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Tafenoquine manufacturer or Tafenoquine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Tafenoquine manufacturer or Tafenoquine supplier.
PharmaCompass also assists you with knowing the Tafenoquine API Price utilized in the formulation of products. Tafenoquine API Price is not always fixed or binding as the Tafenoquine Price is obtained through a variety of data sources. The Tafenoquine Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Tafenoquine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Tafenoquine, including repackagers and relabelers. The FDA regulates Tafenoquine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Tafenoquine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Tafenoquine supplier is an individual or a company that provides Tafenoquine active pharmaceutical ingredient (API) or Tafenoquine finished formulations upon request. The Tafenoquine suppliers may include Tafenoquine API manufacturers, exporters, distributors and traders.
click here to find a list of Tafenoquine suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Tafenoquine as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Tafenoquine API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Tafenoquine as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Tafenoquine and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Tafenoquine NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Tafenoquine suppliers with NDC on PharmaCompass.
Tafenoquine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Tafenoquine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Tafenoquine GMP manufacturer or Tafenoquine GMP API supplier for your needs.
A Tafenoquine CoA (Certificate of Analysis) is a formal document that attests to Tafenoquine's compliance with Tafenoquine specifications and serves as a tool for batch-level quality control.
Tafenoquine CoA mostly includes findings from lab analyses of a specific batch. For each Tafenoquine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Tafenoquine may be tested according to a variety of international standards, such as European Pharmacopoeia (Tafenoquine EP), Tafenoquine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Tafenoquine USP).

