
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
FDA Orange Book

0
Canada

0
Australia

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. A 45975
2. Adecur
3. Apo-terazosin
4. Deflox
5. Dysalfa
6. Flotrin
7. Heitrin
8. Hytrin
9. Hytrin Bph
10. Hytrine
11. Magnurol
12. Novo-terazosin
13. Nu-terazosin
14. Pms-terazosin
15. Ratio-terazosin
16. Sutif
17. Tazusin
18. Terazoflo
19. Terazosin Azu
20. Terazosin Hexal
21. Terazosin Hydrochloride
22. Terazosin Hydrochloride Anhydrous
23. Terazosin, Monohydrochloride, Dihydrate
24. Terazosina Alter
25. Terazosina Kern
26. Terazosina Qualix
27. Zayasel
1. 63590-64-7
2. Terazosine
3. Hytrin
4. Terazosina
5. Terazosinum
6. Flumarc
7. Vasomet
8. Terazosine [inn-french]
9. Terazosinum [inn-latin]
10. Terazosina [inn-spanish]
11. Terazosabb
12. Terazosin Hcl
13. Terazosin (inn)
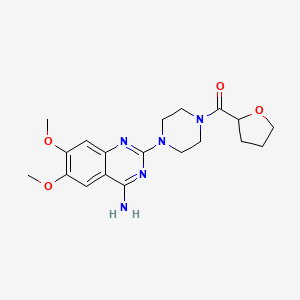
14. 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-((tetrahydro-2-furanyl)carbonyl)piperazine
15. Chembl611
16. [4-(4-amino-6,7-dimethoxyquinazolin-2-yl)piperazin-1-yl]-(oxolan-2-yl)methanone
17. Mls000069703
18. Chebi:9445
19. 141269-44-5
20. Terazosin (hydrochloride)
21. 8l5014xet7
22. 141269-45-6
23. Smr000058309
24. Terazosin [inn]
25. Terazosin [inn:ban]
26. 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-(tetrahydro-2-furoyl)piperazine
27. 6,7-dimethoxy-2-[4-(oxolane-2-carbonyl)piperazin-1-yl]quinazolin-4-amine
28. (4-(4-amino-6,7-dimethoxyquinazolin-2-yl)piperazin-1-yl)(tetrahydrofuran-2-yl)methanone
29. [4-(4-amino-6,7-dimethoxyquinazolin-2-yl)piperazin-1-yl](tetrahydrofuran-2-yl)methanone
30. 6,7-dimethoxy-2-[4-(tetrahydrofuran-2-ylcarbonyl)piperazin-1-yl]quinazolin-4-amine
31. Abbott 45975
32. 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-[(tetrahydro-2-furanyl)carbonyl]piperazine
33. 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-{(tetnaphydro-2-furanyl)carbonyl}piperazine
34. 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)4-[(tetrahydro-2-furanyl)carbonyl]piperazine
35. Terazosabb (tn)
36. 6,7-dimethoxy-2-[4-(tetrahydrofuran-2-ylcarbonyl)piperazin-1-yl]quinazolin-4-amine Hydrochloride
37. Terazosin A
38. Unii-8l5014xet7
39. Terazosinhydrochloride
40. Terazosin [mi]
41. 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)4-[(tetrahydro-2-furanyl)carbonyl]piperazine Hydrochloride
42. Opera_id_1910
43. Prestwick0_000751
44. Prestwick1_000751
45. Prestwick2_000751
46. Prestwick3_000751
47. Terazosin [vandf]
48. Schembl6528
49. Terazosin [who-dd]
50. Lopac0_001138
51. Regid_for_cid_5401
52. Bspbio_000762
53. Mls001201836
54. Mls006011889
55. Spbio_002701
56. Bpbio1_000840
57. Gtpl7302
58. Dtxsid3023639
59. Hms2090p21
60. Hms2232n21
61. Hms3259f04
62. Hms3369p14
63. Hms3371e20
64. Hms3742i09
65. Hy-b0371
66. Bbl010743
67. Bdbm50033111
68. Stk567029
69. Akos005266642
70. Ccg-205212
71. Db01162
72. Nc00689
73. Sdccgsbi-0051105.p002
74. 6,7-bis(methyloxy)-2-[4-(tetrahydrofuran-2-ylcarbonyl)piperazin-1-yl]quinazolin-4-amine
75. Piperazine, 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-((tetrahydro-2-furanyl)carbonyl)-
76. Ncgc00016026-04
77. Ncgc00016026-05
78. Ncgc00016026-08
79. Ncgc00016026-11
80. Ncgc00025191-03
81. Ac-11120
82. Ls-14728
83. Ft-0630739
84. C07127
85. D08569
86. 590t647
87. A834201
88. L000692
89. Q280786
90. Brd-a22256192-003-03-7
91. Brd-a22256192-003-14-4
92. Z1172269406
93. 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-(2-tetrahydrofuroyl)piperazine
94. 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-(2-tetrahydrofuroyl)-piperazine
95. 6,7-dimethoxy-2-[4-(oxolane-2-carbonyl)piperazin-1-yl]-3,4-dihydroquinazolin-4-imine
96. 6,7-dimethoxy-2-[4-(tetrahydro-2-furanylcarbonyl)-1-piperazinyl]-4-quinazolinamine #
97. 6,7-dimethoxy-2-{4-[(oxolan-2-yl)carbonyl]piperazin-1-yl}quinazolin-4-amine
98. 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-[(tetrahydro-2-furanyl)carbonyl]-piperazinehydrochloride
99. 1-(4-amino-6,7-dimethoxyquinazolin-2-yl)-4-[[(2rs)-2,3,4,5-tetrahydrofuran-2-yl]carbonyl]piperazine
| Molecular Weight | 387.4 g/mol |
|---|---|
| Molecular Formula | C19H25N5O4 |
| XLogP3 | 1.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 4 |
| Exact Mass | 387.19065430 g/mol |
| Monoisotopic Mass | 387.19065430 g/mol |
| Topological Polar Surface Area | 103 Ų |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 544 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Terazosin is indicated for use in treating symptomatic benign prostatic hyperplasia and hypertension.
FDA Label
Terazosin is a quinazoline derivative alpha-1-selective adrenergic blocker.
Adrenergic alpha-1 Receptor Antagonists
Drugs that bind to and block the activation of ADRENERGIC ALPHA-1 RECEPTORS. (See all compounds classified as Adrenergic alpha-1 Receptor Antagonists.)
Urological Agents
Drugs used in the treatment of urological conditions and diseases such as URINARY INCONTINENCE and URINARY TRACT INFECTIONS. (See all compounds classified as Urological Agents.)
G04CA03
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
G - Genito urinary system and sex hormones
G04 - Urologicals
G04C - Drugs used in benign prostatic hypertrophy
G04CA - Alpha-adrenoreceptor antagonists
G04CA03 - Terazosin
Absorption
Approximately 90%.
Route of Elimination
Approximately 10% of the oral dose is excreted unchanged in the urine and approximately 20% is excreted in the feces. 40% of the total dose is eliminated in urine and 60% of the total dose is eliminated in the feces.
Volume of Distribution
25L to 30L.
Clearance
Plasma clearance is 80mL/min and renal clearance is 10mL/min.
The majority of terazosin is hepatically metabolized. The metabolites recovered include 6-O-demethyl terazosin, 7-O-methyl terazosin, a piperozine derivative, and a diamine derivative.
Terazosin has a mean half life 12 hours though this can be as high as 14 hours in patients over 70 years and as low as 11.4 hours in patients 20 to 39 years old.
Terazosin is selective for alpha-1-adrenoceptors but not their individual subtypes. Inhibition of these alpha-1-adrenoceptors results in relaxation of smooth muscle in blood vessels and the prostate, lowering blood pressure and improving urinary flow. Smooth muscle cells accounts for roughly 40% of the volume of the prostate and so their relaxation reduces pressure on the urethra. It has also been shown that catecholamines induce factors responsible for mitogenesis and alpha-1-adrenergic receptor blockers inhibit this effect. A final long term mechanism of terazosin and other alpha-1-adrenergic receptor blockers is the induction of apoptosis of prostate cells. Treatment with terazosin enhances the expression of transforming growth factor beta-1 (TGF-beta1), which upregulates p27kip1, and the caspase cascade.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
58
PharmaCompass offers a list of Terazosin API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Terazosin manufacturer or Terazosin supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Terazosin manufacturer or Terazosin supplier.
PharmaCompass also assists you with knowing the Terazosin API Price utilized in the formulation of products. Terazosin API Price is not always fixed or binding as the Terazosin Price is obtained through a variety of data sources. The Terazosin Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Terazosin manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Terazosin, including repackagers and relabelers. The FDA regulates Terazosin manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Terazosin API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Terazosin manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Terazosin supplier is an individual or a company that provides Terazosin active pharmaceutical ingredient (API) or Terazosin finished formulations upon request. The Terazosin suppliers may include Terazosin API manufacturers, exporters, distributors and traders.
click here to find a list of Terazosin suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Terazosin as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Terazosin API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Terazosin as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Terazosin and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Terazosin NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Terazosin suppliers with NDC on PharmaCompass.
Terazosin Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Terazosin GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Terazosin GMP manufacturer or Terazosin GMP API supplier for your needs.
A Terazosin CoA (Certificate of Analysis) is a formal document that attests to Terazosin's compliance with Terazosin specifications and serves as a tool for batch-level quality control.
Terazosin CoA mostly includes findings from lab analyses of a specific batch. For each Terazosin CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Terazosin may be tested according to a variety of international standards, such as European Pharmacopoeia (Terazosin EP), Terazosin JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Terazosin USP).
