
Synopsis
0
JDMF
0
KDMF
0
VMF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
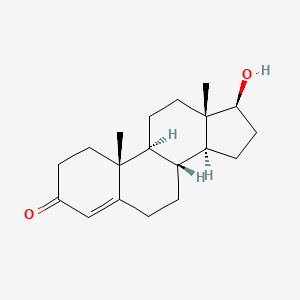

1. 17 Beta Hydroxy 4 Androsten 3 One
2. 17 Beta Hydroxy 8 Alpha 4 Androsten 3 One
3. 17-beta-hydroxy-4-androsten-3-one
4. 17-beta-hydroxy-8 Alpha-4-androsten-3-one
5. 8 Isotestosterone
6. 8-isotestosterone
7. Androderm
8. Androgel
9. Andropatch
10. Androtop
11. Histerone
12. Sterotate
13. Sustanon
14. Testim
15. Testoderm
16. Testolin
17. Testopel
18. Testosterone Sulfate
1. 58-22-0
2. Androderm
3. Testim
4. Testosteron
5. Virosterone
6. Mertestate
7. Sustanon
8. Testoderm
9. Androgel
10. Androlin
11. Homosterone
12. Oreton
13. Testiculosterone
14. Synandrol F
15. Andropatch
16. Homosteron
17. Orquisteron
18. Perandren
19. Primotest
20. Primoteston
21. Sustanone
22. Testandrone
23. Testobase
24. Testopropon
25. Testosteroid
26. Andronaq
27. Andrusol
28. Relibra
29. Testogel
30. Testrone
31. Testryl
32. Teslen
33. Cristerona T
34. Testoviron T
35. Malerone
36. Neotestis
37. Testoviron
38. Trans-testosterone
39. Testoviron Schering
40. Cristerone T
41. Geno-cristaux Gremy
42. Testostosterone
43. Testolin
44. Libigel
45. Sustason 250
46. Percutacrine Androgenique
47. 17beta-hydroxyandrost-4-en-3-one
48. Oreton-f
49. Testoject-50
50. Testosterona
51. Testosteronum
52. Intrinsa
53. Striant
54. Andro 100
55. Testex
56. Testosterone Hydrate
57. Virormone
58. Beta Testosterone
59. Testro Aq
60. Malestrone (amps)
61. Malogen, Aquaspension Injection
62. Testosteronum [inn-latin]
63. Testosterona [inn-spanish]
64. Natesto
65. Vogelxo
66. Neo-hombreol F
67. Androst-4-en-17beta-ol-3-one
68. 17beta-hydroxy-4-androsten-3-one
69. 7-beta-hydroxyandrost-4-en-3-one
70. Col 1621
71. Delta4-androsten-17beta-ol-3-one
72. 17beta-hydroxyandrost-4-ene-3-one
73. 17-beta-hydroxyandrost-4-en-3-one
74. 4-androsten-17beta-ol-3-one
75. Testosterone Ciii
76. 17-hydroxy-(17beta)-androst-4-en-3-one
77. 17beta-hydroxy-delta(sup4)-androsten-3-one
78. Delta(sup 4)-androsten-17(beta)-ol-3-one
79. (+)-testosterone
80. 17-hydroxy-(17-beta)-androst-4-en-3-one
81. 17-beta-hydroxy-delta(sup 4)-androsten-3-one
82. Androst-4-en-3-one, 17beta-hydroxy-
83. Androst-4-en-3-one, 17-hydroxy-, (17beta)-
84. Androst-4-en-3-one, 17-beta-hydroxy-
85. Chebi:17347
86. (17beta)-17-hydroxyandrost-4-en-3-one
87. (8r,9s,10r,13s,14s,17s)-17-hydroxy-10,13-dimethyl-1,2,6,7,8,9,11,12,14,15,16,17-dodecahydrocyclopenta[a]phenanthren-3-one
88. Neo-testis
89. Testoderm Tts
90. 3xmk78s47o
91. Androst-4-en-3-one, 17-hydroxy-, (17b)-
92. Nsc-9700
93. Testopel Pellets
94. Cdb 111c
95. Testosterone And Its Esters
96. Halotensin
97. Fortesta
98. Livensa
99. Nasobol
100. Axiron
101. Tefina
102. Androst-4-en-3-one, 17-hydroxy-, (17.beta.)-
103. Compleotrt
104. Cp 601b
105. 17.beta.-hydroxyandrost-4-en-3-one
106. (8alpha,10alpha,13alpha,14beta,17alpha)-17-hydroxyandrost-4-en-3-one
107. Androderm (tn)
108. Androgel 1%
109. 17.beta.-testosterone
110. Androgel (tn)
111. Smr000058344
112. Ccris 574
113. Striant (tn)
114. Bio-t-gel
115. Aa 2500
116. Testim (tn)
117. Androgel 1.62%
118. Hsdb 3398
119. Nsc 9700
120. Einecs 200-370-5
121. Testosterone (jan/usp)
122. Androst-4-en-3-on-17b-ol
123. Chembl386630
124. Component Of Duogen (salt/mix)
125. Component Of Tostrex (salt/mix)
126. Component Of Di-met (salt/mix)
127. Unii-3xmk78s47o
128. Component Of Intrinsa (salt/mix)
129. Epitestosteron
130. Lumitestosteron
131. 4-androsten-17.beta.-ol-3-one
132. 4-androsten-3-one-17.beta.-ol
133. Tostrelle
134. Tostrex
135. Viatrel
136. Component Of Di-genik (salt/mix)
137. 4-androsten-3-one, 17b-hydroxy-
138. Testosterone [usp:inn:ban]
139. 9b-testosterone
140. Component Of Mal-o-fem (salt/mix)
141. Testosterone [androgenic Steroids, Anabolic]
142. 4-androstene-17beta-ol-3-one
143. 17b-hydroxy-4-androsten-3-one
144. Delta4-androsten-17b-ol-3-one
145. Lpcn 1021
146. Testosterone, 1
147. 3kdm
148. 17b-testosterone
149. 17-.beta.-hydroxyandrost-4-en-3-one
150. Ncgc00091018-01
151. 8-iso-testosterone
152. Androst-4-en-3-one, 17-hydroxy-, (17-beta)-
153. (+-)-testosterone
154. Axiron (tn)
155. Cmc_13449
156. 13-iso-testosterone
157. Androlan
158. Mpp10
159. 9b,10a-testosterone
160. 17.beta.-hydroxy-.delta.4-androsten-3-one
161. Android-t
162. 17-hydroxyandrost-4-en-3-one, (17.beta.)-
163. (+-)-retrotestosterone
164. Testosterone, >=98%
165. Testosterone [mi]
166. Dsstox_cid_2371
167. Testosterone [inn]
168. Testosterone [jan]
169. Epitope Id:135865
170. Ec 200-370-5
171. Testosterone [hsdb]
172. Testosterone [inci]
173. Testosterone Ep Impurity C
174. (+-)-8-iso-testosterone
175. Schembl8452
176. Dsstox_rid_76563
177. Testosterone [vandf]
178. Dsstox_gsid_22371
179. Testosterone [mart.]
180. 4-androsten-3-one-17b-ol
181. Mls000563091
182. Mls001032098
183. Mls001306401
184. Mls001424262
185. Mls002174283
186. Bidd:er0555
187. Testosterone [who-dd]
188. D4-androsten-17b-ol-3-one
189. Androst-4-en-17b-ol-3-one
190. Bdbm8885
191. Gtpl2858
192. Androst-4-ene-17b-ol-3-one
193. Testosterone [ema Epar]
194. Dtxsid8022371
195. 17-hydroxy-d4-androsten-3-one
196. 17-hydroxy-androst-4-en-3-one
197. 17beta-hydroxy-4-androsten-3one
198. Testosterone [green Book]
199. Testosterone, Cell Culture Tested
200. 17b-hydroxy-androst-4-en-3-on
201. 17b-hydroxy-d4-androsten-3-one
202. 17b-hydroxyandrost-4-ene-3-one
203. 1i37
204. 17a-hydroxy-androst-4-en-3-one
205. 17b-hydroxy-androst-4-en-3-one
206. Hms2052n11
207. Hms2272b03
208. Hms2272p03
209. Testosterone [ep Impurity]
210. Testosterone [orange Book]
211. Testosterone [ep Monograph]
212. (8r,9s,10r,13s,14s,17s)-17-hydroxy-10,13-dimethyl-6,7,8,9,10,11,12,13,14,15,16,17-dodecahydro-1h-cyclopenta[a]phenanthren-3(2h)-one
213. 17-hydroxy-10,13-dimethyl-1,2,6,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-cyclopenta[a]phenanthren-3-one
214. Testosterone [usp Monograph]
215. Tox21_200689
216. Hy-15554a
217. Lmst02020002
218. Rb3046
219. 17b-hydroxy-8a-androst-4-en-3-one
220. Rac-17b-hydroxy-androst-4-en-3-one
221. Akos015894897
222. Zinc118912393
223. 17a-hydroxy-13a-androst-4-en-3-one
224. 17a-hydroxy-14b-androst-4-en-3-one
225. 17b-hydroxy-13a-androst-4-en-3-one
226. Ccg-101189
227. Cs-1415
228. Db00624
229. Nc00439
230. Cas-58-22-0
231. Testosterone 1.0 Mg/ml In Acetonitrile
232. Testosterone 100 Microg/ml In Methanol
233. (17?)-17-hydroxyandrost-4-en-3-one
234. 17b-hydroxy-(8a)-androst-4-en-3-one
235. 17b-hydroxy-(9b)-androst-4-en-3-one
236. Ncgc00258243-01
237. (17b)-17-hydroxy-androst-4-en-3-one
238. 17a-hydroxy-(13a)-androst-4-en-3-one
239. 17b-hydroxy-(10a)-androst-4-en-3-one
240. 17b-hydroxy-(13a)-androst-4-en-3-one
241. Ac-14899
242. Cpd000058344
243. Smr001261453
244. Testosterone 1000 Microg/ml In Methanol
245. Testosterone, Purum, >=99.0% (hplc)
246. Testosterone 100 Microg/ml In Acetonitrile
247. Rac-17b-hydroxy-(13a)androst-4-en-3-one
248. Rac-17b-hydroxy-(8a)-androst-4-en-3-one
249. T0027
250. 17b-hydroxy-(8a,10a)-androst-4-en-3-one
251. 17b-hydroxy-(9b,10a)-androst-4-en-3-one
252. Bim-0061761.0001
253. C00535
254. D00075
255. Rac-17b-hydroxy-(9b,10a)androst-4-en-3-one
256. S00309
257. Ab00973630-03
258. Testosterone, Vetranal(tm), Analytical Standard
259. 003t654
260. Androst-4-en-3-one, 17-hydroxy, (17.beta.)-
261. Q-101251
262. Q1318776
263. B5dee83f-632b-48a1-a0ed-a51e7f13df2e
264. Testosterone Enantate Impurity D [ep Impurity]
265. Testosterone Propionate Impurity C [ep Impurity]
266. Testosterone, European Pharmacopoeia (ep) Reference Standard
267. Testosterone Solution, 1.0 Mg/ml In 1,2-dimethoxyethane, Drug Standard
268. Testosterone; 4-androsten-17?-ol-3-one; 17?-hydroxy-4-androsten-3-one
269. Testosterone For Impurity D Identification, European Pharmacopoeia (ep) Reference Standard
270. Testosterone For System Suitability, European Pharmacopoeia (ep) Reference Standard
271. (1s,2r,10r,11s,14s,15s)-14-hydroxy-2,15-dimethyltetracyclo[8.7.0.0;{2,7}.0;{11,15}]heptadec-6-en-5-one
272. 1050678-68-6
273. 17?-hydroxyandrost-4-en-3-one; Epitestosterone; 17-epi-testosterone; 17?-cis-testosterone; 4-androstene-17?-ol-3-one; Isotestosterone
274. Testosterone Solution, 1.0 Mg/ml In Acetonitrile, Ampule Of 1 Ml, Certified Reference Material
1. Testosterone Acetate
2. Testosterone Decanoate
3. Testosterone Isocaproate
4. Testosterone Laurate
5. Testosterone Dodecanoate
6. Retandrol
7. Testosterone Hydrocinnamate
8. Testosterone Phenpropionate
9. Testosterone Phenylpropionate
| Molecular Weight | 288.4 g/mol |
|---|---|
| Molecular Formula | C19H28O2 |
| XLogP3 | 3.3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | 288.208930132 g/mol |
| Monoisotopic Mass | 288.208930132 g/mol |
| Topological Polar Surface Area | 37.3 Ų |
| Heavy Atom Count | 21 |
| Formal Charge | 0 |
| Complexity | 508 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 6 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 14 | |
|---|---|
| Drug Name | Androderm |
| PubMed Health | Testosterone |
| Drug Classes | Androgen, Endocrine-Metabolic Agent |
| Drug Label | ANDRODERM (testosterone transdermal system) is designed to delivertestosteronecontinuously for 24 hours following application to intact, non-scrotal skin (e.g., back, abdomen, thighs, upper arms). Two strengths of ANDRODERM are available that del... |
| Active Ingredient | Testosterone |
| Dosage Form | Film, extended release |
| Route | Transdermal |
| Strength | 2mg/24hr; 4mg/24hr |
| Market Status | Prescription |
| Company | Watson Labs |
| 2 of 14 | |
|---|---|
| Drug Name | Androgel |
| PubMed Health | Testosterone (On the skin) |
| Drug Classes | Endocrine-Metabolic Agent |
| Drug Label | AndroGel (testosterone gel) 1% is a clear, colorle hydroalcoholic gel containing testosterone. The active pharmacologic ingredient in AndroGel 1% is testosterone, an androgen. Testosterone USP is a white to practically white crystalline powder chem... |
| Active Ingredient | Testosterone |
| Dosage Form | Gel; Gel, metered |
| Route | Transdermal |
| Strength | 1.62% (20.25mg/1.25gm actuation); 1.62% (20.25mg/1.25gm packet); 12.5mg/1.25gm actuation; 50mg/5gm packet; 1.62% (40.5mg/2.5gm packet); 25mg/2.5gm packet |
| Market Status | Prescription |
| Company | Abbvie |
| 3 of 14 | |
|---|---|
| Drug Name | Axiron |
| PubMed Health | Testosterone |
| Drug Classes | Androgen, Endocrine-Metabolic Agent |
| Drug Label | AXIRON (testosterone) topical solution is a clear, colorle, single phase solution containing 30 mg of testosterone in 1.5 mL of AXIRON solution for topical administration through the axilla. The active pharmacologic ingredient in AXIRON is testoste... |
| Active Ingredient | Testosterone |
| Dosage Form | Solution, metered |
| Route | Transdermal |
| Strength | 30mg/1.5ml actuation |
| Market Status | Prescription |
| Company | Eli Lilly And |
| 4 of 14 | |
|---|---|
| Drug Name | Fortesta |
| PubMed Health | Testosterone |
| Drug Classes | Androgen, Endocrine-Metabolic Agent |
| Drug Label | FORTESTA is a clear, colorle, odorle, gel containing testosterone. FORTESTA is available in a metered-dose pump. Each pump actuation provides 10 mg of testosterone and each container is capable of dispensing 120 pump actuations. One pump actuatio... |
| Active Ingredient | Testosterone |
| Dosage Form | Gel, metered |
| Route | Transdermal |
| Strength | 10mg/0.5gm actuation |
| Market Status | Prescription |
| Company | Endo Pharms |
| 5 of 14 | |
|---|---|
| Drug Name | Striant |
| PubMed Health | Testosterone |
| Drug Classes | Androgen, Endocrine-Metabolic Agent |
| Drug Label | Striant (testosterone buccal system) is designed to adhere to the gum or inner cheek. It provides a controlled and sustained release of testosterone through the buccal mucosa as the buccal system gradually hydrates. Insertion of Striant twice a d... |
| Active Ingredient | Testosterone |
| Dosage Form | Tablet, extended release |
| Route | Buccal |
| Strength | 30mg |
| Market Status | Prescription |
| Company | Auxilium Pharms |
| 6 of 14 | |
|---|---|
| Drug Name | Testim |
| Drug Label | Structure Image... |
| Active Ingredient | Testosterone |
| Dosage Form | Gel |
| Route | Transdermal |
| Strength | 1% (50mg/5gm packet) |
| Market Status | Prescription |
| Company | Auxilium Pharms |
| 7 of 14 | |
|---|---|
| Drug Name | Testosterone |
| Drug Label | Structure Image... |
| Active Ingredient | Testosterone |
| Dosage Form | Gel; Gel, metered |
| Route | Transdermal |
| Strength | 12.5mg/1.25gm actuation; 50mg/5gm packet; 25mg/2.5gm packet |
| Market Status | Prescription |
| Company | Teva Pharms; Perrigo Israel |
| 8 of 14 | |
|---|---|
| Drug Name | Androderm |
| PubMed Health | Testosterone |
| Drug Classes | Androgen, Endocrine-Metabolic Agent |
| Drug Label | ANDRODERM (testosterone transdermal system) is designed to delivertestosteronecontinuously for 24 hours following application to intact, non-scrotal skin (e.g., back, abdomen, thighs, upper arms). Two strengths of ANDRODERM are available that del... |
| Active Ingredient | Testosterone |
| Dosage Form | Film, extended release |
| Route | Transdermal |
| Strength | 2mg/24hr; 4mg/24hr |
| Market Status | Prescription |
| Company | Watson Labs |
| 9 of 14 | |
|---|---|
| Drug Name | Androgel |
| PubMed Health | Testosterone (On the skin) |
| Drug Classes | Endocrine-Metabolic Agent |
| Drug Label | AndroGel (testosterone gel) 1% is a clear, colorle hydroalcoholic gel containing testosterone. The active pharmacologic ingredient in AndroGel 1% is testosterone, an androgen. Testosterone USP is a white to practically white crystalline powder chem... |
| Active Ingredient | Testosterone |
| Dosage Form | Gel; Gel, metered |
| Route | Transdermal |
| Strength | 1.62% (20.25mg/1.25gm actuation); 1.62% (20.25mg/1.25gm packet); 12.5mg/1.25gm actuation; 50mg/5gm packet; 1.62% (40.5mg/2.5gm packet); 25mg/2.5gm packet |
| Market Status | Prescription |
| Company | Abbvie |
| 10 of 14 | |
|---|---|
| Drug Name | Axiron |
| PubMed Health | Testosterone |
| Drug Classes | Androgen, Endocrine-Metabolic Agent |
| Drug Label | AXIRON (testosterone) topical solution is a clear, colorle, single phase solution containing 30 mg of testosterone in 1.5 mL of AXIRON solution for topical administration through the axilla. The active pharmacologic ingredient in AXIRON is testoste... |
| Active Ingredient | Testosterone |
| Dosage Form | Solution, metered |
| Route | Transdermal |
| Strength | 30mg/1.5ml actuation |
| Market Status | Prescription |
| Company | Eli Lilly And |
| 11 of 14 | |
|---|---|
| Drug Name | Fortesta |
| PubMed Health | Testosterone |
| Drug Classes | Androgen, Endocrine-Metabolic Agent |
| Drug Label | FORTESTA is a clear, colorle, odorle, gel containing testosterone. FORTESTA is available in a metered-dose pump. Each pump actuation provides 10 mg of testosterone and each container is capable of dispensing 120 pump actuations. One pump actuatio... |
| Active Ingredient | Testosterone |
| Dosage Form | Gel, metered |
| Route | Transdermal |
| Strength | 10mg/0.5gm actuation |
| Market Status | Prescription |
| Company | Endo Pharms |
| 12 of 14 | |
|---|---|
| Drug Name | Striant |
| PubMed Health | Testosterone |
| Drug Classes | Androgen, Endocrine-Metabolic Agent |
| Drug Label | Striant (testosterone buccal system) is designed to adhere to the gum or inner cheek. It provides a controlled and sustained release of testosterone through the buccal mucosa as the buccal system gradually hydrates. Insertion of Striant twice a d... |
| Active Ingredient | Testosterone |
| Dosage Form | Tablet, extended release |
| Route | Buccal |
| Strength | 30mg |
| Market Status | Prescription |
| Company | Auxilium Pharms |
| 13 of 14 | |
|---|---|
| Drug Name | Testim |
| Drug Label | Structure Image... |
| Active Ingredient | Testosterone |
| Dosage Form | Gel |
| Route | Transdermal |
| Strength | 1% (50mg/5gm packet) |
| Market Status | Prescription |
| Company | Auxilium Pharms |
| 14 of 14 | |
|---|---|
| Drug Name | Testosterone |
| Drug Label | Structure Image... |
| Active Ingredient | Testosterone |
| Dosage Form | Gel; Gel, metered |
| Route | Transdermal |
| Strength | 12.5mg/1.25gm actuation; 50mg/5gm packet; 25mg/2.5gm packet |
| Market Status | Prescription |
| Company | Teva Pharms; Perrigo Israel |
Androgens
National Library of Medicine's Medical Subject Headings. Testosterone. Online file (MeSH, 2016). Available from, as of November 28, 2016: https://www.nlm.nih.gov/mesh/2016/mesh_browser/MBrowser.html
/CLINICAL TRIALS/ ClinicalTrials.gov is a registry and results database of publicly and privately supported clinical studies of human participants conducted around the world. The Web site is maintained by the National Library of Medicine (NLM) and the National Institutes of Health (NIH). Each ClinicalTrials.gov record presents summary information about a study protocol and includes the following: Disease or condition; Intervention (for example, the medical product, behavior, or procedure being studied); Title, description, and design of the study; Requirements for participation (eligibility criteria); Locations where the study is being conducted; Contact information for the study locations; and Links to relevant information on other health Web sites, such as NLM's MedlinePlus for patient health information and PubMed for citations and abstracts for scholarly articles in the field of medicine. Testosterone is included in the database.
NIH/NLM; ClinicalTrials.Gov. Available from, as of February 1, 2017: https://clinicaltrials.gov/ct2/results?term=testosterone&Search=Search
In males, testosterone is used for the management of congenital or acquired primary hypogonadism such as that resulting from orchiectomy or from testicular failure caused by cryptorchidism, bilateral torsion, orchitis, or vanishing testis syndrome. Testosterone also is used in males for the management of congenital or acquired hypogonadotropic hypogonadism such as that resulting from idiopathic gonadotropin or gonadotropin-releasing hormone (luteinizing hormone releasing hormone) deficiency or from pituitary-hypothalamic injury caused by tumors, trauma, or radiation. If any of these conditions occur before puberty, androgen replacement therapy will be necessary during adolescence for the development of secondary sexual characteristics and prolonged therapy will be required to maintain these characteristics. Prolonged androgen therapy also is required to maintain sexual characteristics in other males who develop testosterone deficiency after puberty. /Included in US product labeling/
American Society of Health-System Pharmacists 2016; Drug Information 2016. Bethesda, MD. 2016, p. 3131
When the diagnosis is well established, testosterone may be used to stimulate puberty in carefully selected males with delayed puberty. These males usually have a family history of delayed puberty that is not caused by a pathologic disorder. Brief treatment with conservative doses of an androgen may occasionally be justified in these males if they do not respond to psychologic support. Because androgens may adversely affect bone maturation in these prepubertal males, this potential risk should be fully discussed with the patient and his parents prior to initiation of androgen therapy. If androgen therapy is initiated in these prepubertal males, radiographs of the hand and wrist should be obtained at 6-month intervals to determine the effect of therapy on the epiphyseal centers. Testosterone is designated an orphan drug by the FDA for use in this condition. /Included in US product labeling/
American Society of Health-System Pharmacists 2016; Drug Information 2016. Bethesda, MD. 2016, p. 3132
For more Therapeutic Uses (Complete) data for Testosterone (17 total), please visit the HSDB record page.
/BOXED WARNING/ WARNING: SECONDARY EXPOSURE TO TESTOSTERONE. Virilization has been reported in children who were secondarily exposed to testosterone gel. Children should avoid contact with unwashed or unclothed application sites in men using testosterone gel. Healthcare providers should advise patients to strictly adhere to recommended instructions for use.
NIH; DailyMed. Current Medication Information for Androgel (Testosterone Gel) (Updated: January 2015). Available from, as of April 5, 2017: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=a8253ce7-77df-49d4-8077-816cf4dc234f
Cardiovascular events, including MI or stroke, have been reported during postmarketing experience with testosterone transdermal system (Androderm). Testosterone should be used with caution in patients at high risk for cardiovascular disease (e.g., older men, those with diabetes mellitus or obesity). Patients should be advised to immediately report symptoms suggestive of MI or stroke (e.g., chest pain, shortness of breath, unilateral weakness, difficulty talking) to their clinician.
American Society of Health-System Pharmacists 2016; Drug Information 2016. Bethesda, MD. 2016, p. 3135-6
Venous thromboembolic events, including deep-vein thrombosis (DVT) and pulmonary embolism (PE), have been reported during postmarketing experience with testosterone preparations, including testosterone transdermal system (Androderm). Patients reporting symptoms of pain, edema, warmth, and erythema in a lower extremity or presenting with acute shortness of breath should be evaluated for possible DVT or PE, respectively. If venous thromboembolism is suspected, testosterone therapy should be discontinued and appropriate evaluation and management should be initiated.
American Society of Health-System Pharmacists 2016; Drug Information 2016. Bethesda, MD. 2016, p. 3136
Testosterone should be used with caution in patients with cardiac, renal, and/or hepatic dysfunction since edema may occur as a result of sodium and water retention. Edema, with or without congestive heart failure, may be a serious complication in patients with preexisting cardiac, renal, and/or hepatic disease. If edema occurs during testosterone therapy and it is considered a serious complication, the drug should be discontinued; diuretic therapy may also be necessary.
American Society of Health-System Pharmacists 2016; Drug Information 2016. Bethesda, MD. 2016, p. 3136
For more Drug Warnings (Complete) data for Testosterone (34 total), please visit the HSDB record page.
Testosterone is indicated to treat primary hypogonadism and hypogonadotropic hypogonadism.
FDA Label
Intrinsa is indicated for the treatment of hypoactive sexual desire disorder (HSDD) in bilaterally oophorectomised and hysterectomised (surgically induced menopause) women receiving concomitant estrogen therapy.
Livensa is indicated for the treatment of hypoactive sexual desire disorder (HSDD) in bilaterally oophorectomised and hysterectomised (surgically induced menopause) women receiving concomitant estrogen therapy.
Treatment of dry eye disease
Treatment of male hypogonadism
Hypoactive sexual desire disorder
Hypoactive sexual desire disorder
Treatment of male hypogonadism
Testosterone antagonizes the androgen receptor to induce gene expression that causes the growth and development of masculine sex organs and secondary sexual characteristics. The duration of action of testosterone is variable from patient to patient with a half life of 10-100 minutes. The therapeutic index is wide considering the normal testosterone levels in an adult man range from 300-1000ng/dL. Counsel patients regarding the risk of secondary exposure of testosterone topical products to children.
Androgens
Compounds that interact with ANDROGEN RECEPTORS in target tissues to bring about the effects similar to those of TESTOSTERONE. Depending on the target tissues, androgenic effects can be on SEX DIFFERENTIATION; male reproductive organs, SPERMATOGENESIS; secondary male SEX CHARACTERISTICS; LIBIDO; development of muscle mass, strength, and power. (See all compounds classified as Androgens.)
G03BA03
G03BA03
G03BA03
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
G - Genito urinary system and sex hormones
G03 - Sex hormones and modulators of the genital system
G03B - Androgens
G03BA - 3-oxoandrosten (4) derivatives
G03BA03 - Testosterone
Absorption
A single 100mg topical dose of testosterone has an AUC of 104255521ng\*h/dL and a Cmax of 573284ng/dL. Testosterone is approximately 10% bioavailable topically.
Route of Elimination
90% of an intramuscular dose is eliminated in urine, mainly as glucuronide and sulfate conjugates. 6% is eliminated in feces, mostly as unconjugated metabolites.
Volume of Distribution
The volume of distribution of testosterone in elderly men is 80.3624.51L.
Clearance
The mean metabolic clearance in middle aged men is 81264L/day.
Testosterone is absorbed systemically through the skin following topical application as a gel or transdermal system. Following topical application of a hydroalcoholic gel formulation of testosterone (AndroGel, Testim) to the skin, the gel quickly dries on the skin surface, which serves as a reservoir for sustained release of the hormone into systemic circulation. Approximately 10% of a testosterone dose applied topically to the skin as a 1% gel is absorbed percutaneously into systemic circulation. The manufacturer of AndroGel states that increases in serum testosterone concentrations were apparent within 30 minutes of topical application of a 100-mg testosterone dose of the 1% gel, with physiologic concentrations being achieved in most patients within 4 hours (pretreatment concentrations were not described); percutaneous absorption continues for the entire 24-hour dosing interval. Serum testosterone concentrations approximate steady-state levels by the end of the initial 24 hours and are at steady state by the second or third day of dosing of the 1% gel. With daily topical application of the 1% gel (AndroGel), serum testosterone concentrations 30, 90, and 180 days after initiating treatment generally are maintained in the eugonadal range. Administration of 10 or 5 g of AndroGel daily results in average daily serum testosterone concentrations of 794 or 566 ng/dL, respectively, at day 30. Following discontinuance of such topical therapy, serum testosterone concentrations remain within the normal range for 24-48 hours but return to pretreatment levels by the fifth day after the last application.
American Society of Health-System Pharmacists 2016; Drug Information 2016. Bethesda, MD. 2016, p. 3137
With topical application of a transdermal preparation, the extent of percutaneous testosterone absorption varies according to the site of application, possibly secondary to regional differences in skin permeability, cutaneous blood flow, and/or degree of adhesion between the transdermal system and skin. In one study in which transdermal systems were applied to the abdomen, back, chest, shin, thigh, or upper arm, serum hormone profiles were qualitatively similar with each site, but steady-state serum concentrations showed significant differences, decreasing in order with the back, thigh, upper arm, abdomen, chest, and shin. Application of Androderm transdermal systems to the abdomen, back, thighs, or upper arms results in achievement of similar serum testosterone concentration profiles, and these sites are recommended as optimal for rotation of application sites during chronic therapy. Daily nighttime (at approximately 10 p.m.) application of Androderm transdermal system results in a serum testosterone concentration profile that mimics the endogenous diurnal pattern in healthy young men. In one study, showering 3 hours after application of Androderm decreased peak plasma concentrations of testosterone by 0.4% compared with not showering 3 hours after application of the transdermal system. In addition, showering 3 hours after transdermal system application did not substantially alter the systemic exposure of testosterone.
American Society of Health-System Pharmacists 2016; Drug Information 2016. Bethesda, MD. 2016, p. 3137-8
Following topical application of transdermal systems of testosterone, the hormone is absorbed percutaneously into systemic circulation. Although interindividual variation in percutaneous testosterone absorption occurs, serum testosterone concentrations achieved with recommended dosages of transdermal systems of the drug generally reach the normal range during the first day of dosing and are maintained during continuous dosing without accumulation. Average daily serum testosterone concentrations in patients receiving Androderm reportedly are 498 ng/dL at steady state. Mean ratios of testosterone to DHT are within the normal range.
American Society of Health-System Pharmacists 2016; Drug Information 2016. Bethesda, MD. 2016, p. 3137
Esterification of testosterone generally results in less polar compounds. The enanthate ester of testosterone is absorbed slowly from the lipid tissue phase at the IM injection site, achieving peak serum concentrations about 72 hours after IM injection; thus, this preparation has a prolonged duration of action (i.e., up to 2-4 weeks) following IM administration. Because IM injection of testosterone esters causes local irritation, the rate of absorption may be erratic. /Testosterone esters/
American Society of Health-System Pharmacists 2016; Drug Information 2016. Bethesda, MD. 2016, p. 3137
For more Absorption, Distribution and Excretion (Complete) data for Testosterone (9 total), please visit the HSDB record page.
Testosterone is metabolized to 17-keto steroids through two different pathways. The major active metabolites are estradiol and dihydrotestosterone (DHT). Testosterone can be hydroxylated at a number of positions by CYP3A4, CYP2B6, CYP2C9, and CYP2C19; glucuronidated by UGT2B17; sulfated; converted to estradiol by aromatase; converted to dihydrotestosterone (DHT) by 5-reductase; metabolized to androstenedione by CYP3A4, CYP2C9, and CYP2C19; or converted to DHT glucuronide. Androstenedione undergoes metabolism by aromatase to form estrone, which undergoes a reversible reaction to form estradiol. Androstenedione can also be converted to 5-androstanedione by 5-reductase, which can be further metabolized to 5-androsterone. DHT can be glucuronidated or sulfated, or metabolized to 5-androstanediol, androstane-3,17-diol, or androstane-3,17-diol. DHT can also be reversibly converted to 5-androstanedione.
Extensive reductive metabolism of testosterone occurs not only in the liver, but also in a variety of extrahepatic tissues, especially in target organs of the sex hormones; the ultimately effective physiological androgen is formed in the target tissues. Testosterone metabolism occurs not only in the prostate and seminal vesicles but also in rat uterus, rabbit placenta, rodent testis and primate brain. In rats, the small intestine is also capable of metabolizing testosterone.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V21 536 (1979)
It is transformed to 5-alpha-dehydrotestosterone in target organs such as the prostate, sebaceous glands and seminal vesicles; only the latter compound binds to the androgen-receptor site in these target organs.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V21 536 (1979)
Large quantitative differences in testosterone metabolism are evident between female and male rats. The reason for this phenomenon is that many steroid-metabolizing enzymes in rats are either androgen- or estrogen-dependent; the sex hormones thus act in an inductive or a repressive manner.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V21 536 (1979)
Esters of testosterone, such as the propionate, the heptanoate, the cypionate, the valerate, the isovalerate, the enanthate and the undecanoate, are partially cleaved in vivo to release the parent compound. This has been demonstrated by oral administration of testosterone undecanoate in oily solution to rats: most of the compound is converted within the intestinal wall, the first step being partial splitting off of the fatty acid moiety. The non-metabolized portion, however, and the metabolite 5-alpha-dihydrotestosterone undecanoate, are absorbed via the lymphatic system and made available for androgenic action to the organism. /Testosterone esters/
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V21 537 (1979)
For more Metabolism/Metabolites (Complete) data for Testosterone (6 total), please visit the HSDB record page.
Testosterone has known human metabolites that include 15-Hydroxytestosterone, 15beta-Hydroxytestosterone, 16-Hydroxytestosterone, 16beta-Hydroxytestosterone, 2alpha-Hydroxytestosterone, 2beta-Hydroxytestosterone, 6alpha-Hydroxytestosterone, Androstenedione, and Testosterone sulfate.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
The half life of testosterone is highly variable, ranging from 10-100 minutes.
The plasma half-life of testosterone reportedly ranges from 10-100 minutes. The plasma half-life of testosterone cypionate after IM injection is approximately 8 days. Following removal of an Androderm transdermal system, plasma testosterone concentrations decline with an apparent half-life of approximately 70 minutes ... .
American Society of Health-System Pharmacists 2016; Drug Information 2016. Bethesda, MD. 2016, p. 3138
The androgen receptor exists in the cytoplasm bound to the heat shock proteins HSP90, HSP70, and other chaperones. After binding to an androgen, the androgen receptor dissociates from HSP90 and undergoes a conformational change to slow the rate of dissociation from the androgen receptor. The androgen-receptor complex is transported into the nucleus where it binds to DNA and recruits other transcriptional regulators to form a pre-initiation complex and eventually induce expression of specific genes. Testosterone and its active metabolite dihydrotestosterone (DHT) antagonize the androgen receptor to develop masculine sex organs including the prostate, seminal vesicles, penis, and scrotum. Antagonism of the androgen receptor is also responsible for the development of secondary sexual characteristics including facial and body hair, enlargement of the larynx, thickening of the vocal cords, and changes in muscle and fat distribution.
Low-grade chronic inflammation is commonly found in patients with polycystic ovary syndrome (PCOS) who exhibit hyperandrogenism or hyperandrogenemia. Clinical studies have shown that hyperandrogenemia is closely correlated with low-grade chronic inflammation. However, the mechanism underlying this correlation remains unclear. Recent studies have suggested that adipocytes increase the production of proinflammatory mediators such as interleukin-6 (IL-6) and macrophage chemotactic protein-1 (MCP-1) when the inflammatory signal transduction cascade system is activated by external stimuli. The present study aimed to evaluate the effects of testosterone on the innate signalling and expression of proinflammatory mediators in 3T3-L1 adipocytes, which were or were not induced by lipopolysaccharide (LPS). The effects of testosterone on the expression of proinflammatory mediators, nuclear factor-kappaB (NF-kappaB), and extracellular signal-regulated kinase 1/2 (ERK1/2) signalling pathways were investigated using an enzyme-linked immunosorbent assay, reverse transcriptase-polymerase chain reaction, western blot analysis and an electrophoresis mobility shift assay. Testosterone induces IL-6 and MCP-1, and enhances LPS-induction of IL-6 and MCP-1. However, the effects are not simply additive, testosterone significantly enhanced the effects of LPS-induced inflammation factors. Testosterone induces the phosphorylation of ERK1/2 and NF-kappaB. The effect of testosterone on the expression of IL-6 and MCP-1 is inhibited by PD98059, an ERK1/2 inhibitor, and PDTC, an NF-kappaB inhibitor. The results indicate that testosterone enhances LPS-induced IL-6 and MCP-1 expression by activating the ERK1/2/NF-kappaB signalling pathways in 3T3-L1 adipocytes.
PMID:25738264 Su C et al; Mol Med Rep 12 (1): 696-704 (2015)
Androgens reportedly stimulate the production of erythrocytes, apparently by enhancing the production of erythropoietic stimulating factor.
American Society of Health-System Pharmacists 2016; Drug Information 2016. Bethesda, MD. 2016, p. 3137

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
90
PharmaCompass offers a list of Testosterone API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Testosterone manufacturer or Testosterone supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Testosterone manufacturer or Testosterone supplier.
PharmaCompass also assists you with knowing the Testosterone API Price utilized in the formulation of products. Testosterone API Price is not always fixed or binding as the Testosterone Price is obtained through a variety of data sources. The Testosterone Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Testosterone manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Testosterone, including repackagers and relabelers. The FDA regulates Testosterone manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Testosterone API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Testosterone manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Testosterone supplier is an individual or a company that provides Testosterone active pharmaceutical ingredient (API) or Testosterone finished formulations upon request. The Testosterone suppliers may include Testosterone API manufacturers, exporters, distributors and traders.
click here to find a list of Testosterone suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Testosterone DMF (Drug Master File) is a document detailing the whole manufacturing process of Testosterone active pharmaceutical ingredient (API) in detail. Different forms of Testosterone DMFs exist exist since differing nations have different regulations, such as Testosterone USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Testosterone DMF submitted to regulatory agencies in the US is known as a USDMF. Testosterone USDMF includes data on Testosterone's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Testosterone USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Testosterone suppliers with USDMF on PharmaCompass.
A Testosterone CEP of the European Pharmacopoeia monograph is often referred to as a Testosterone Certificate of Suitability (COS). The purpose of a Testosterone CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Testosterone EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Testosterone to their clients by showing that a Testosterone CEP has been issued for it. The manufacturer submits a Testosterone CEP (COS) as part of the market authorization procedure, and it takes on the role of a Testosterone CEP holder for the record. Additionally, the data presented in the Testosterone CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Testosterone DMF.
A Testosterone CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Testosterone CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Testosterone suppliers with CEP (COS) on PharmaCompass.
A Testosterone written confirmation (Testosterone WC) is an official document issued by a regulatory agency to a Testosterone manufacturer, verifying that the manufacturing facility of a Testosterone active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Testosterone APIs or Testosterone finished pharmaceutical products to another nation, regulatory agencies frequently require a Testosterone WC (written confirmation) as part of the regulatory process.
click here to find a list of Testosterone suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Testosterone as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Testosterone API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Testosterone as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Testosterone and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Testosterone NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Testosterone suppliers with NDC on PharmaCompass.
Testosterone Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Testosterone GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Testosterone GMP manufacturer or Testosterone GMP API supplier for your needs.
A Testosterone CoA (Certificate of Analysis) is a formal document that attests to Testosterone's compliance with Testosterone specifications and serves as a tool for batch-level quality control.
Testosterone CoA mostly includes findings from lab analyses of a specific batch. For each Testosterone CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Testosterone may be tested according to a variety of international standards, such as European Pharmacopoeia (Testosterone EP), Testosterone JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Testosterone USP).
