
Synopsis
Synopsis
0
CEP/COS
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
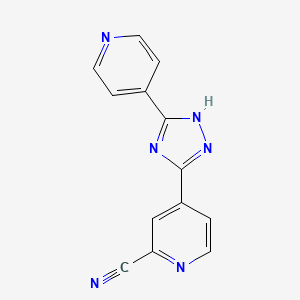

1. 4-(5-pyridin-4-yl-1h-(1,2,4) Triazol-3-yl)pyridine-2-carbonitrile
2. Fyx-051
1. 577778-58-6
2. Fyx 051
3. Fyx-051
4. 4-(5-pyridin-4-yl-1h-1,2,4-triazol-3-yl)pyridine-2-carbonitrile
5. Topiroxostat [inn]
6. 2-pyridinecarbonitrile, 4-[5-(4-pyridinyl)-1h-1,2,4-triazol-3-yl]-
7. 5-(2-cyano-4-pyridyl)-3-(4-pyridyl)-1,2,4-triazole
8. Chembl1078685
9. 4-(5-(pyridin-4-yl)-1h-1,2,4-triazol-3-yl)picolinonitrile
10. 4-(5-(pyridin-4-yl)-1h-1,2,4-triazol-3-yl)pyridine-2-carbonitrile
11. Mfcd17167057
12. 0j877412jv
13. 4-[5-(pyridin-4-yl)-1h-1,2,4-triazol-3-yl]pyridine-2-carbonitrile
14. Topiloric
15. Uriadec
16. 4-(3-(pyridin-4-yl)-1h-1,2,4-triazol-5-yl)picolinonitrile
17. Unii-0j877412jv
18. Uriadec (tn)
19. Topiroxostat (jan/inn)
20. Topiroxostat [mi]
21. Topiroxostat [jan]
22. Schembl860420
23. Topiroxostat [who-dd]
24. Schembl18056618
25. Gtpl10592
26. Dtxsid80206462
27. Bcp09957
28. Ex-a1197
29. 4-(5-pyridin-4-yl-1h-(1,2,4) Triazol-3-yl)pyridine-2-carbonitrile
30. 4-[5-(4-pyridyl)-1h-1,2,4-triazol-3-yl]pyridine-2-carbonitrile
31. Bdbm50267750
32. Bdbm50311275
33. S3719
34. Zinc13536586
35. 4-[5-pyridin-4-yl-1h-[1,2,4]triazol-3-yl]-pyridine-2-carbonitrile
36. Akos026750465
37. Akos026751485
38. (fyx-051)"
39. Ccg-266938
40. Cs-2033
41. Db01685
42. Ncgc00378610-01
43. Ncgc00378610-02
44. Ac-30740
45. As-56359
46. Hy-14874
47. Sy116097
48. Ft-0700966
49. A14144
50. D09786
51. Q15725832
| Molecular Weight | 248.24 g/mol |
|---|---|
| Molecular Formula | C13H8N6 |
| XLogP3 | 1.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Exact Mass | 248.08104428 g/mol |
| Monoisotopic Mass | 248.08104428 g/mol |
| Topological Polar Surface Area | 91.1 Ų |
| Heavy Atom Count | 19 |
| Formal Charge | 0 |
| Complexity | 344 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Indicated for the treatment of gout and hyperurcemia in Japan.
Topiroxostat reduces the synthesis of uric acid by competitively inhibiting xanthine oxidase in a selective and time-dependent manner. It serves to reduce the concentration of insoluble urates and uric acid in tissues, plasma and urine. Topiroxostat is not reported to cause QT prolongation.
Enzyme Inhibitors
Compounds or agents that combine with an enzyme in such a manner as to prevent the normal substrate-enzyme combination and the catalytic reaction. (See all compounds classified as Enzyme Inhibitors.)
Absorption
The time to reach peak plasma concentration of 229.9 ng/mL was 0.67 hour following a single oral dose of 20mg topiroxostat. The oral bioavailability in male rats was 69.6% after oral administration of a single dose of 1mg/kg.
Route of Elimination
Urinary excretion and fecal excretion of radiolabeled topiroxostat are 30.4% and 40.9% of total dose of 1mg/kg administered to rats, respectively. Within 24 h after a single oral administration of 120mg of topiroxostat, the main metabolites of topiroxostat, N-oxide, N1-gluculonide, and N2-gluculonide, are excreted into urine about 4.8, 43.3, and 16.1 % of the dose, respectively. Unchanged topiroxostat and the hydroxide metabolite was 0.1% or less.
Volume of Distribution
The distribution of 14C-topiroxostat (20, 200, and 2000 ng/mL) in human blood cells was 6.7% to 12.8%.
Clearance
The apparent total body clearance rate is 89.5 L/h and the renal clearance rate is 17.4 mL/h following a single oral dose of 20mg topiroxostat.
Topiroxostat is mainly inactivated by hepatic metabolism. 2-hydroxy topiroxostat is formed from primary hydroxylation of the drug by xanthine oxidase and still retains an inhibitory activity on the enzyme. Topiroxostat N-oxide is another major metabolite that can be detected in plasma and urine. It is determined that the N-oxide and hydroxide metabolites are pyridine N-oxide and pyridine 2 (or 6)-hydroxide, respectively. Topiroxostat is mainly inactivated by hepatic metabolism where it undergoes glucuronidation. The metabolism of topiroxostat to N1-and N2-glucuronide conjugates is mainly mediated by UGT1A1, 1A7, and 1A9, with UGT1A9 being the most predominant.
FYX-051 has known human metabolites that include (2S,3S,4S,5R)-6-[3-(2-cyanopyridin-4-yl)-5-pyridin-4-yl-1,2,4-triazol-1-yl]-3,4,5-trihydroxyoxane-2-carboxylic acid and 4-[2-[(3R,4S,5S,6S)-6-carboxy-3,4,5-trihydroxyoxan-2-yl]-5-pyridin-4-yl-1,2,4-triazol-3-yl]pyridine-2-carbonitrilium.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
The mean half life of topiroxostat after a single oral dose of 20mg topiroxostat is 5 hours under fasting condition. The complex of molybdenum (IV)- topiroxostat has an approximate half life of 20.4 hours.
Uric acid synthesis depends on the action of xanthine oxidase activity in the conversion of hypoxanthine to xanthine, followed by the conversion of xanthine to uric acid. Xanthine oxidase consists of a molybdenum ion as cofactor in the active center that has different redox states upon substrate binding. When a substrate such as hypoxanthine or xanthine binds, xanthine oxidase hydroxylates it and molybdenum ion is reduced from hexavalent, Mo(VI), to tetravalent form, Mo(IV). Molybdenum ion is reoxidized into hexavalent state once the hydroxylated substrate, xanthine or uric acid, dissociates from the active site. Topiroxostat is shown to interact with multiple amino acid residues of the solvent channel and additionally forms a reaction intermediate by covalent binding with molybdenum (IV) ion via an oxygen atom. It also forms hydrogen bonds with molybdenum (VI) ion, suggesting that it has multiple inhibition modes to xanthine oxidase. Enhanced binding interactions to xanthine oxidase achieves delayed dissociation of topiroxostat from the enzyme. 2-hydroxy-topiroxostat, the metabolite formed by primary hydroxylation of topiroxostat by xanthine oxidase, also causes time and concentration-dependent inhibition of the enzyme. Topiroxostat is shown to inhibit ATP-binding cassette transporter G2 (ABCG2) in vitro, which is a membrane protein responsible for recovering uric acid in the kidneys and secreting uric acid from the intestines.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
82
PharmaCompass offers a list of Topiroxostat API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Topiroxostat manufacturer or Topiroxostat supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Topiroxostat manufacturer or Topiroxostat supplier.
PharmaCompass also assists you with knowing the Topiroxostat API Price utilized in the formulation of products. Topiroxostat API Price is not always fixed or binding as the Topiroxostat Price is obtained through a variety of data sources. The Topiroxostat Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Topiroxostat manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Topiroxostat, including repackagers and relabelers. The FDA regulates Topiroxostat manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Topiroxostat API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Topiroxostat manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Topiroxostat supplier is an individual or a company that provides Topiroxostat active pharmaceutical ingredient (API) or Topiroxostat finished formulations upon request. The Topiroxostat suppliers may include Topiroxostat API manufacturers, exporters, distributors and traders.
click here to find a list of Topiroxostat suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Topiroxostat DMF (Drug Master File) is a document detailing the whole manufacturing process of Topiroxostat active pharmaceutical ingredient (API) in detail. Different forms of Topiroxostat DMFs exist exist since differing nations have different regulations, such as Topiroxostat USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Topiroxostat DMF submitted to regulatory agencies in the US is known as a USDMF. Topiroxostat USDMF includes data on Topiroxostat's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Topiroxostat USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Topiroxostat suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Topiroxostat Drug Master File in Japan (Topiroxostat JDMF) empowers Topiroxostat API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Topiroxostat JDMF during the approval evaluation for pharmaceutical products. At the time of Topiroxostat JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Topiroxostat suppliers with JDMF on PharmaCompass.
Topiroxostat Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Topiroxostat GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Topiroxostat GMP manufacturer or Topiroxostat GMP API supplier for your needs.
A Topiroxostat CoA (Certificate of Analysis) is a formal document that attests to Topiroxostat's compliance with Topiroxostat specifications and serves as a tool for batch-level quality control.
Topiroxostat CoA mostly includes findings from lab analyses of a specific batch. For each Topiroxostat CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Topiroxostat may be tested according to a variety of international standards, such as European Pharmacopoeia (Topiroxostat EP), Topiroxostat JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Topiroxostat USP).
