

Synopsis
Synopsis
0
CEP/COS
0
VMF
0
FDA Orange Book

0
Canada

0
Australia

0
South Africa

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
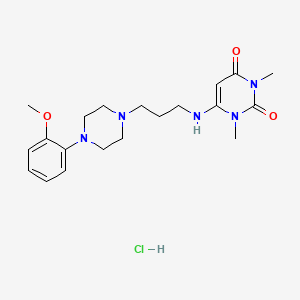

1. Urapidil Hcl
2. 64887-14-5
3. Urapidil (hydrochloride)
4. Pvu92pzo12
5. Ncgc00094502-01
6. Ebrantil (tn)
7. Dsstox_cid_25812
8. Dsstox_rid_81149
9. Dsstox_gsid_45812
10. 6-[3-[4-(2-methoxyphenyl)piperazin-1-yl]propylamino]-1,3-dimethylpyrimidine-2,4-dione;hydrochloride
11. 6-((3-(4-(2-methoxyphenyl)piperazin-1-yl)propyl)amino)-1,3-dimethylpyrimidine-2,4(1h,3h)-dione Hydrochloride
12. 6-[[3-[4-(2-methoxyphenyl)-1-piperazinyl]propyl]amino]-1,3-dimethyl-2,4(1h,3h)-pyrimidinedione Monohydrochloride
13. Smr000058525
14. Sr-01000075295
15. Cas-64887-14-5
16. Unii-pvu92pzo12
17. Urapidilhydrochloride
18. Urapidil Hydrochloride,(s)
19. Urapidil Hydrochloride, Solid
20. Mls000758309
21. Mls001424033
22. C20h29n5o3.hcl
23. Schembl1321654
24. Chembl1256716
25. Dtxsid2045812
26. Hy-b0354a
27. Hms1570f12
28. Pharmakon1600-01503100
29. Bcp23389
30. Tox21_111286
31. Tox21_501265
32. Mfcd00078601
33. Nsc758226
34. S2025
35. Akos015994621
36. Tox21_111286_1
37. Ac-8089
38. Ccg-100798
39. H61u751
40. Hs-0064
41. Lp01265
42. Nc00048
43. Nsc-758226
44. Urapidil Hydrochloride [who-dd]
45. Ncgc00016066-14
46. Ncgc00261950-01
47. Eu-0101265
48. Ft-0675734
49. Sw197244-4
50. U-100
51. C76033
52. D08662
53. Sr-01000075295-1
54. Sr-01000075295-3
55. Sr-01000075295-9
56. Q27286781
57. Sr-01000075295-10
58. 6[[3-[4-(o-methoxyphenyl)-1-piperazinyl]propyl]amino]-1,3-dimethyluracil Hydrochloride
59. 2,4(1h,3h)-pyrimidinedione, 6-((3-(4-(2-methoxyphenyl)-1-piperazinyl)propyl)amino)-1,3-dimethyl-, Monohydrochloride
60. 6-(3-(4-(2-methoxyphenyl)piperazin-1-yl)propylamino)-1,3-dimethylpyrimidine-2,4(1h,3h)-dione Hydrochloride
61. 6-[[3-[4-(2-methoxyphenyl)-1-pipera Zinyl]propyl]amino]-1,3-dimethyl-2,4(1h,3h)-pyrimi Dinedione Hydrochloride
62. 6-[[3-[4-(2-methoxyphenyl)-1-piperazinyl]propyl]amino]-1,3-dimethyl-2,4(1h,3h)-pyrimidinedione Hydrochloride
| Molecular Weight | 423.9 g/mol |
|---|---|
| Molecular Formula | C20H30ClN5O3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 7 |
| Exact Mass | 423.2037175 g/mol |
| Monoisotopic Mass | 423.2037175 g/mol |
| Topological Polar Surface Area | 68.4 Ų |
| Heavy Atom Count | 29 |
| Formal Charge | 0 |
| Complexity | 588 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
 Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38214
Submission : 2023-03-29
Status : Active
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 4382
Submission : 1981-12-07
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF

Registration Number : 304MF10127
Registrant's Address : VIA F. BARACCA 19-24060 SAN PAOLO D'ARGON (BERGAMO) ITALY
Initial Date of Registration : 2022-08-31
Latest Date of Registration : --

Registrant Name : Samo Pharmaceutical Co., Ltd.
Registration Date : 2020-06-24
Registration Number : 20200624-211-J-564
Manufacturer Name : Erregierre SpA
Manufacturer Address : Via Francesco. Baracca, 19-24060 SAN PAOLO D'ARGON (BG), Italy




Date of Issue : 2019-07-08
Valid Till : 2022-07-08
Written Confirmation Number : WC-0204
Address of the Firm :



 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
 Ever Pharma, highly specialized CDMO in producing complex injectables including high potency & controlled substances & suspensions.
Ever Pharma, highly specialized CDMO in producing complex injectables including high potency & controlled substances & suspensions.
Regulatory Info :
Registration Country : Austria
Brand Name : Tachyben
Dosage Form : Solution for Injection
Dosage Strength : 50MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Austria
 Ever Pharma, highly specialized CDMO in producing complex injectables including high potency & controlled substances & suspensions.
Ever Pharma, highly specialized CDMO in producing complex injectables including high potency & controlled substances & suspensions.
Regulatory Info :
Registration Country : Austria
Brand Name : Tachyben
Dosage Form : Solution for Injection
Dosage Strength : 25MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Austria
 Ever Pharma, highly specialized CDMO in producing complex injectables including high potency & controlled substances & suspensions.
Ever Pharma, highly specialized CDMO in producing complex injectables including high potency & controlled substances & suspensions.
Regulatory Info :
Registration Country : Austria
Brand Name : Tachyben
Dosage Form : Solution for Injection
Dosage Strength : 100MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Austria
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Turkey
Brand Name :
Dosage Form : Solution for Injection
Dosage Strength : 25MG/5ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Turkey

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Capsule
Dosage Strength : 30MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Capsule
Dosage Strength : 90MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Urapidil
Dosage Form : Urapidil 50Mg 10Ml 5 Units Parenteral Use
Dosage Strength : 5 EV vials 50 mg 10 ml
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country :
Brand Name :
Dosage Form : Prolonged Release Capsule
Dosage Strength : 90MG
Packaging : 30, 50, 60 (2x 30),90 (3x 30) and 100 (2x 50) capsules
Approval Date :
Application Number :
Regulatory Info :
Registration Country :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : France
Brand Name :
Dosage Form : Capsule
Dosage Strength : 30MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : France

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : France
Brand Name :
Dosage Form : Capsule
Dosage Strength : 90MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : France

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
We have 1 companies offering Urapidil
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
