

Synopsis
Synopsis
0
CEP/COS
0
VMF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
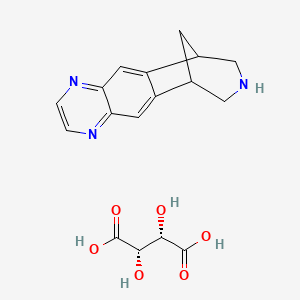

1. 6,7,8,9-tetrahydro-6,10-methano-6h-pyrazino(2,3-h)benzazepine
2. Champix
3. Varenicline
4. Varenicline Tartrate
1. Varenicline Tartrate
2. 375815-87-5
3. (2s,3s)-2,3-dihydroxybutanedioic Acid; 5,8,14-triazatetracyclo[10.3.1.0^{2,11}.0^{4,9}]hexadeca-2(11),3,5,7,9-pentaene
4. (2s,3s)-2,3-dihydroxybutanedioic Acid;5,8,14-triazatetracyclo[10.3.1.02,11.04,9]hexadeca-2,4,6,8,10-pentaene
5. Akos026750167
6. As-35136
7. A21286
8. Z1642292115
| Molecular Weight | 361.3 g/mol |
|---|---|
| Molecular Formula | C17H19N3O6 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 3 |
| Exact Mass | 361.12738533 g/mol |
| Monoisotopic Mass | 361.12738533 g/mol |
| Topological Polar Surface Area | 153 Ų |
| Heavy Atom Count | 26 |
| Formal Charge | 0 |
| Complexity | 388 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 2 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Chantix |
| PubMed Health | Varenicline (By mouth) |
| Drug Classes | Smoking Ceation Agent |
| Drug Label | CHANTIX tablets contain varenicline (as the tartrate salt), which is a partial agonist selective for 42 nicotinic acetylcholine receptor subtypes.Varenicline, as the tartrate salt, is a powder which is a white to off-white to slightly yellow soli... |
| Active Ingredient | Varenicline tartrate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 0.5mg base; eq 1mg base |
| Market Status | Prescription |
| Company | Pfizer |
| 2 of 2 | |
|---|---|
| Drug Name | Chantix |
| PubMed Health | Varenicline (By mouth) |
| Drug Classes | Smoking Ceation Agent |
| Drug Label | CHANTIX tablets contain varenicline (as the tartrate salt), which is a partial agonist selective for 42 nicotinic acetylcholine receptor subtypes.Varenicline, as the tartrate salt, is a powder which is a white to off-white to slightly yellow soli... |
| Active Ingredient | Varenicline tartrate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 0.5mg base; eq 1mg base |
| Market Status | Prescription |
| Company | Pfizer |
Nicotinic Agonists
Drugs that bind to and activate nicotinic cholinergic receptors (RECEPTORS, NICOTINIC). Nicotinic agonists act at postganglionic nicotinic receptors, at neuroeffector junctions in the peripheral nervous system, and at nicotinic receptors in the central nervous system. Agents that function as neuromuscular depolarizing blocking agents are included here because they activate nicotinic receptors, although they are used clinically to block nicotinic transmission. (See all compounds classified as Nicotinic Agonists.)
Smoking Cessation Agents
Substances that facilitate the cessation of tobacco smoking. (See all compounds classified as Smoking Cessation Agents.)

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
Patent Expiration Date : 2035-10-19
US Patent Number : 9597284
Drug Substance Claim :
Drug Product Claim :
Application Number : 213978
Patent Use Code : U-1900
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-10-19

Patent Expiration Date : 2035-10-19
US Patent Number : 11903942
Drug Substance Claim :
Drug Product Claim : Y
Application Number : 213978
Patent Use Code :
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-10-19

Patent Expiration Date : 2035-10-19
US Patent Number : 10456396
Drug Substance Claim :
Drug Product Claim : Y
Application Number : 213978
Patent Use Code : U-1900
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-10-19

Patent Expiration Date : 2035-10-19
US Patent Number : 9504645
Drug Substance Claim :
Drug Product Claim : Y
Application Number : 213978
Patent Use Code :
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-10-19

Patent Expiration Date : 2035-10-19
US Patent Number : 11224598
Drug Substance Claim :
Drug Product Claim :
Application Number : 213978
Patent Use Code : U-1900
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-10-19

Patent Expiration Date : 2035-10-19
US Patent Number : 11903941
Drug Substance Claim :
Drug Product Claim :
Application Number : 213978
Patent Use Code : U-1900
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-10-19

Patent Expiration Date : 2035-10-19
US Patent Number : 11903943
Drug Substance Claim :
Drug Product Claim :
Application Number : 213978
Patent Use Code : U-1900
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-10-19

Patent Expiration Date : 2035-10-19
US Patent Number : 11911380
Drug Substance Claim :
Drug Product Claim :
Application Number : 213978
Patent Use Code : U-1900
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-10-19

Patent Expiration Date : 2035-10-19
US Patent Number : 9504644
Drug Substance Claim :
Drug Product Claim :
Application Number : 213978
Patent Use Code : U-1900
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-10-19

Patent Expiration Date : 2035-10-19
US Patent Number : 9532944
Drug Substance Claim :
Drug Product Claim :
Application Number : 213978
Patent Use Code : U-1900
Delist Requested :
Patent Use Description :
Patent Expiration Date : 2035-10-19

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
