
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
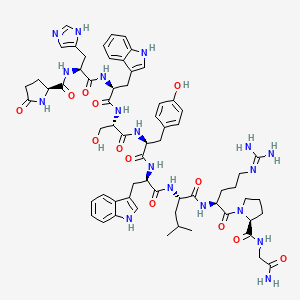

1. 6-d-tryptophan-luteinizing Hormone-releasing Factor (pig)
2. Ay 25650
3. Ay-25650
4. Ay25650
5. Cl 118532
6. Cl-118532
7. Cl118532
8. D-trp-6-lh-rh
9. Decapeptyl
10. Decapeptyl Depot
11. Decapeptyl Lp
12. Decapeptyl Trimestral
13. Embonate, Triptorelin
14. Gnrh, Trp(6)-
15. Lhrh, Trp(6)-
16. Lhrh, Tryptophyl(6)-
17. Pamoate, Triptorelin
18. Trelstar
19. Trimestral, Decapeptyl
20. Triptorelin Embonate
21. Triptorelin Pamoate
22. Wy 42462
23. Wy-42462
24. Wy42462
1. 57773-63-4
2. Triptoreline
3. Arvekap
4. (d-trp6)-gnrh
5. Decapeptyl
6. Triptorelina
7. Triptorelinum
8. Trelstar
9. Triptodur
10. Diphereline
11. (6-d-tryptophan)luteinizing Hormone-releasing Hormone
12. Cl 118532
13. Cl-118532
14. Pglu-his-trp-ser-tyr-d-trp-leu-arg-pro-gly-nh2
15. Ay-25650
16. Luteinizing Hormone-releasing Factor (pig), 6-d-tryptophan
17. Cl 118,532
18. Chebi:63633
19. Wy 42462
20. Ay 25650
21. Pamorelin
22. Salvacyl
23. Trelstar Depot
24. 5-oxo-l-prolyl-l-histidyl-l-tryptophyl-l-seryl-l-tyrosyl-d-tryptophyl-l-leucyl-l-arginyl-l-prolylglycinamide
25. Trelstar La
26. Luteinizing Hormone-releasing Factor (swine), 6-d-tryptophan-
27. Bim 21003
28. Wy 42422
29. Tryptorelin
30. Diferelin
31. D-tryptophan-lh-rh
32. Triptoreline [inn-french]
33. Triptorelinum [inn-latin]
34. Triptorelina [inn-spanish]
35. Trellasar
36. Moapar
37. Triptorelin [usan:inn:ban]
38. Decapeptyl Sr
39. Pamorelin La
40. Unii-9081y98w2v
41. Triptorelin (swine)
42. Wy-42462
43. D-trp6-lhrh
44. Debio-8206
45. Triptorelin (usan/inn)
46. (d-trp6)-lh-rh
47. Gtpl1177
48. Schembl5079698
49. Chembl1201334
50. Dtxsid2048375
51. Schembl22289009
52. Hms2090c17
53. Ex-a3857
54. Bim-21003
55. Akos030213249
56. 9081y98w2v
57. Cs-5745
58. Db06825
59. Ncgc00167301-01
60. Ncgc00167301-02
61. As-71149
62. Bn-52014
63. Hy-12551
64. 73t634
65. C75704
66. D06247
67. [d-trp6]-lh-rh, >=97% (hplc), Powder
68. Ab01275488-01
69. A936467
70. Pglu-his-trp-ser-tyr-d-trp-leu-arg-pro-glynh2
71. Q1992452
72. (s)-1-((3s,6s,9s,12s,15r,18s,21s)-3-((1h-imidazol-5-yl)methyl)-6,15-bis((1h-indol-3-yl)methyl)-21-(3-guanidinopropyl)-12-(4-hydroxybenzyl)-9-(hydroxymethyl)-18-isobutyl-1,4,7,10,13,16,19-heptaoxo-1-((s)-5-oxopyrrolidin-2-yl)-2,5,8,11,14,17,20-heptaazadocosan-22-oyl)-n-(2-amino-2-oxoethyl)pyrrolidine-2-carboxamide
73. Glycinamide, 5-oxo-l-prolyl-l-histidyl-l-tryptophyl-l-seryl-l-tyrosyl-d-tryptophyl-l-leucyl-l-arginyl-l-prolyl-
| Molecular Weight | 1311.4 g/mol |
|---|---|
| Molecular Formula | C64H82N18O13 |
| XLogP3 | -0.3 |
| Hydrogen Bond Donor Count | 17 |
| Hydrogen Bond Acceptor Count | 15 |
| Rotatable Bond Count | 33 |
| Exact Mass | 1310.63087474 g/mol |
| Monoisotopic Mass | 1310.63087474 g/mol |
| Topological Polar Surface Area | 490 Ų |
| Heavy Atom Count | 95 |
| Formal Charge | 0 |
| Complexity | 2710 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 9 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Trelstar |
| PubMed Health | Triptorelin (Injection) |
| Drug Classes | Antineoplastic Agent, Endocrine-Metabolic Agent |
| Drug Label | TRELSTAR is a white to slightly yellow lyophilized cake. When reconstituted, TRELSTAR has a milky appearance. It contains a pamoate salt of triptorelin, a synthetic decapeptide agonist analog of gonadotropin releasing hormone (GnRH). The chemical... |
| Active Ingredient | Triptorelin pamoate |
| Dosage Form | Injectable |
| Route | Intramuscular |
| Strength | eq 11.25mg base/vial; eq 3.75mg base/vial; eq 22.5mg base/vial |
| Market Status | Prescription |
| Company | Watson Labs |
| 2 of 2 | |
|---|---|
| Drug Name | Trelstar |
| PubMed Health | Triptorelin (Injection) |
| Drug Classes | Antineoplastic Agent, Endocrine-Metabolic Agent |
| Drug Label | TRELSTAR is a white to slightly yellow lyophilized cake. When reconstituted, TRELSTAR has a milky appearance. It contains a pamoate salt of triptorelin, a synthetic decapeptide agonist analog of gonadotropin releasing hormone (GnRH). The chemical... |
| Active Ingredient | Triptorelin pamoate |
| Dosage Form | Injectable |
| Route | Intramuscular |
| Strength | eq 11.25mg base/vial; eq 3.75mg base/vial; eq 22.5mg base/vial |
| Market Status | Prescription |
| Company | Watson Labs |
Triptorelin is indicated for the palliative treatment of advanced prostate cancer.
FDA Label
For the synchronisation of ovulation in weaned sows to enable a single fixed-time artificial insemination.
The first administration of triptorelin is followed by a transient surge of follicle stimulating hormone (FSH), luteinizing hormone (LH), estradiol,and testosterone. The time, peak and decline of testosterone in the body varies depending on the dose administered. This initial surge is often responsible for worsening of prostate cancer symptoms such as urethral or bladder outlet obstruction, bone pain, spinal cord injury and hematuria in the early stages. A sustained decrease in FSH and LH, and significant reduction of testicular steroidogenesis is usually seen 2-4 weeks post-initiation of therapy. This result is a reduction of serum testosterone to levels which are typically seen in surgically castrated men. Ultimately, tissues and functions that require these hormones become inactive. The effects of triptorelin can usually be reversed once the drug is discontinued.
Antineoplastic Agents, Hormonal
Antineoplastic agents that are used to treat hormone-sensitive tumors. Hormone-sensitive tumors may be hormone-dependent, hormone-responsive, or both. A hormone-dependent tumor regresses on removal of the hormonal stimulus, by surgery or pharmacological block. Hormone-responsive tumors may regress when pharmacologic amounts of hormones are administered regardless of whether previous signs of hormone sensitivity were observed. The major hormone-responsive cancers include carcinomas of the breast, prostate, and endometrium; lymphomas; and certain leukemias. (From AMA Drug Evaluations Annual 1994, p2079) (See all compounds classified as Antineoplastic Agents, Hormonal.)
Luteolytic Agents
Chemical compounds that cause LUTEOLYSIS or degeneration of the CORPUS LUTEUM. (See all compounds classified as Luteolytic Agents.)
QH01CA97
L - Antineoplastic and immunomodulating agents
L02 - Endocrine therapy
L02A - Hormones and related agents
L02AE - Gonadotropin releasing hormone analogues
L02AE04 - Triptorelin
Absorption
Following IV administration of triptorelin, triptorelin is completely absorbed.
Route of Elimination
Elimination of triptorelin involves both the kidneys and the liver.
Volume of Distribution
After a single IV dose of 0.5mg, the volume of distribution of triptorelin peptide in healthy males was 30 - 33L.
Clearance
In healthy male volunteers, total clearance of triptorelin was 211.9 mL/min.
The metabolism of triptorelin in humans is not well understood; however, metabolism likely does not involve hepatic enzymes such as cytochrome P450. Whether or not triptorelin affects, or how it affects other metabolizing enzymes is also poorly understood. Triptorelin has no identified metabolites.
The pharmacokinetics of triptorelin follows a 3 compartment model. The half lives are estimated to be 6 minutes, 45 minutes, and 3 hours respectively.
Triptorelin is a synthetic agonist analog of gonadotropin releasing hormone (GnRH). Animal studies comparing triptorelin to native GnRH found that triptorelin had 13 fold higher releasing activity for luteinizing hormone, and 21-fold higher releasing activity for follicle-stimulating hormone.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
55
PharmaCompass offers a list of Triptorelin API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Triptorelin manufacturer or Triptorelin supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Triptorelin manufacturer or Triptorelin supplier.
PharmaCompass also assists you with knowing the Triptorelin API Price utilized in the formulation of products. Triptorelin API Price is not always fixed or binding as the Triptorelin Price is obtained through a variety of data sources. The Triptorelin Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Watson Brand of Triptorelin Embonate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Watson Brand of Triptorelin Embonate, including repackagers and relabelers. The FDA regulates Watson Brand of Triptorelin Embonate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Watson Brand of Triptorelin Embonate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Watson Brand of Triptorelin Embonate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Watson Brand of Triptorelin Embonate supplier is an individual or a company that provides Watson Brand of Triptorelin Embonate active pharmaceutical ingredient (API) or Watson Brand of Triptorelin Embonate finished formulations upon request. The Watson Brand of Triptorelin Embonate suppliers may include Watson Brand of Triptorelin Embonate API manufacturers, exporters, distributors and traders.
click here to find a list of Watson Brand of Triptorelin Embonate suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Watson Brand of Triptorelin Embonate DMF (Drug Master File) is a document detailing the whole manufacturing process of Watson Brand of Triptorelin Embonate active pharmaceutical ingredient (API) in detail. Different forms of Watson Brand of Triptorelin Embonate DMFs exist exist since differing nations have different regulations, such as Watson Brand of Triptorelin Embonate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Watson Brand of Triptorelin Embonate DMF submitted to regulatory agencies in the US is known as a USDMF. Watson Brand of Triptorelin Embonate USDMF includes data on Watson Brand of Triptorelin Embonate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Watson Brand of Triptorelin Embonate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Watson Brand of Triptorelin Embonate suppliers with USDMF on PharmaCompass.
A Watson Brand of Triptorelin Embonate written confirmation (Watson Brand of Triptorelin Embonate WC) is an official document issued by a regulatory agency to a Watson Brand of Triptorelin Embonate manufacturer, verifying that the manufacturing facility of a Watson Brand of Triptorelin Embonate active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Watson Brand of Triptorelin Embonate APIs or Watson Brand of Triptorelin Embonate finished pharmaceutical products to another nation, regulatory agencies frequently require a Watson Brand of Triptorelin Embonate WC (written confirmation) as part of the regulatory process.
click here to find a list of Watson Brand of Triptorelin Embonate suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Watson Brand of Triptorelin Embonate as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Watson Brand of Triptorelin Embonate API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Watson Brand of Triptorelin Embonate as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Watson Brand of Triptorelin Embonate and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Watson Brand of Triptorelin Embonate NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Watson Brand of Triptorelin Embonate suppliers with NDC on PharmaCompass.
Watson Brand of Triptorelin Embonate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Watson Brand of Triptorelin Embonate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Watson Brand of Triptorelin Embonate GMP manufacturer or Watson Brand of Triptorelin Embonate GMP API supplier for your needs.
A Watson Brand of Triptorelin Embonate CoA (Certificate of Analysis) is a formal document that attests to Watson Brand of Triptorelin Embonate's compliance with Watson Brand of Triptorelin Embonate specifications and serves as a tool for batch-level quality control.
Watson Brand of Triptorelin Embonate CoA mostly includes findings from lab analyses of a specific batch. For each Watson Brand of Triptorelin Embonate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Watson Brand of Triptorelin Embonate may be tested according to a variety of international standards, such as European Pharmacopoeia (Watson Brand of Triptorelin Embonate EP), Watson Brand of Triptorelin Embonate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Watson Brand of Triptorelin Embonate USP).
