
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Europe

0
Australia

0
South Africa

DRUG PRODUCT COMPOSITIONS
Regulatory FDF Prices
NA
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
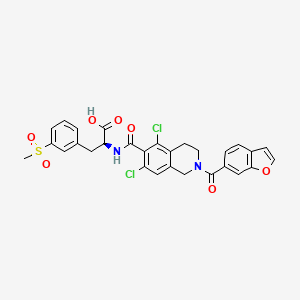

1. (2s)-2-(((2-(benzofuran-6-ylcarbonyl)-5,7-dichloro-1,2,3,4-tetrahydroisoquinolin-6- Yl)carbonyl)amino)-3-(3-(methylsulfonyl)phenyl)propanoic Acid
2. L-phenylalanine, N-((2-(6-benzofuranylcarbonyl)-5,7-dichloro-1,2,3,4-tetrahydro-6-isoquinolinyl)carbonyl)-3-(methylsulfonyl)-
3. Lifitegrast Ophthalmic Solution
4. Sar 1118
5. Sar-1118
6. Shp-606
7. Shp606
8. Xiidra
1. 1025967-78-5
2. Xiidra
3. Sar 1118
4. Sar-1118
5. Shp606
6. Shp-606
7. (s)-2-(2-(benzofuran-6-carbonyl)-5,7-dichloro-1,2,3,4-tetrahydroisoquinoline-6-carboxamido)-3-(3-(methylsulfonyl)phenyl)propanoic Acid
8. Chembl2048028
9. 038e5l962w
10. (2s)-2-(((2-(benzofuran-6-ylcarbonyl)-5,7-dichloro-1,2,3,4-tetrahydroisoquinolin-6- Yl)carbonyl)amino)-3-(3-(methylsulfonyl)phenyl)propanoic Acid
11. L-phenylalanine, N-((2-(6-benzofuranylcarbonyl)-5,7-dichloro-1,2,3,4-tetrahydro-6-isoquinolinyl)carbonyl)-3-(methylsulfonyl)-
12. L-phenylalanine, N-[[2-(6-benzofuranylcarbonyl)-5,7-dichloro-1,2,3,4-tetrahydro-6-isoquinolinyl]carbonyl]-3-(methylsulfonyl)-
13. Lifitegrast [usan:inn]
14. Unii-038e5l962w
15. Sar1118
16. Xiidra (tn)
17. Lifitegrast [mi]
18. Lifitegrast [inn]
19. Lifitegrast (usan/inn)
20. Lifitegrast [usan]
21. Lifitegrast; Sar 1118
22. Lifitegrast [who-dd]
23. Lifitegrast Ophthalmic Solution
24. Gtpl7533
25. Schembl2632068
26. Amy4450
27. Dtxsid60145345
28. Lifitegrast [orange Book]
29. Chebi:133023
30. Ex-a2582
31. Bdbm50386331
32. Mfcd28502439
33. S3714
34. Zinc84668739
35. Ccg-270245
36. Cs-6264
37. Db11611
38. Compound 1g [pmid: 24900456]
39. Ac-32534
40. Ds-20052
41. Hy-19344
42. D10374
43. A900838
44. Q23044263
45. (2s)-2-[[2-(1-benzofuran-6-carbonyl)-5,7-dichloro3,4-dihydro-1h-isoquinoline-6-carbonyl]amino]-3-(3-methylsulfonylphenyl)propanoic Acid
46. N-[[2-(6-benzofuranylcarbonyl)-5,7-dichloro-1,2,3,4-tetrahydro-6-isoquinolinyl]carbonyl]-3-(methylsulfonyl)-l-phenylalanine
47. N-[2-(1-benzofuran-6-carbonyl)-5,7-dichloro-1,2,3,4-tetrahydroisoquinoline-6-carbonyl]-3-(methanesulfonyl)-l-phenylalanine
| Molecular Weight | 615.5 g/mol |
|---|---|
| Molecular Formula | C29H24Cl2N2O7S |
| XLogP3 | 4.7 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 7 |
| Exact Mass | 614.0681277 g/mol |
| Monoisotopic Mass | 614.0681277 g/mol |
| Topological Polar Surface Area | 142 Ų |
| Heavy Atom Count | 41 |
| Formal Charge | 0 |
| Complexity | 1100 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
For the treatment of signs and symptoms of keratoconjunctivitis sicca (dry eye syndrome).
FDA Label
Treatment of dry eye disease
Lifitegrast addresses both the symptoms and the resulting ocular surface damage by interfering with ocular inflammatory cycle. Lifitegrast is a lymphocyte functionassociated antigen-1 antagonist through direct competitive antagonism and sequentially inhibits the T-cell recruitment, activation, and proinflammatory cytokine release associated with dry eye syndrome.
Ophthalmic Solutions
Sterile solutions that are intended for instillation into the eye. It does not include solutions for cleaning eyeglasses or CONTACT LENS SOLUTIONS. (See all compounds classified as Ophthalmic Solutions.)
S - Sensory organs
S01 - Ophthalmologicals
S01X - Other ophthalmologicals
S01XA - Other ophthalmologicals
S01XA25 - Lifitegrast
Absorption
The mean peak plasma concentration (Cmax) of 1.70ng/mL was reached within 15 minutes of application. Quantifiable trough plasma concentrations ranged from 0.55 ng/mL to 3.74 ng/mL. Observations show limited systemic exposure that produces significant clinical outcomes.
Route of Elimination
Not possible to perform mass balance study to determine the main route of elimination.
Clearance
Not possible to calculate clearance rate based on plasma concentrations of lifitegrast, but reported to be relatively fast in rat I.V. pharmacokinetics study. It is predicted that lifitegrast is cleared via nasal and subsequently gastrointestinal tract.
Based on the findings of an in vitro metabolism study using fresh human hepatocytes, lifitegrast does not appear to undergo significant metabolism.
Not possible to calculate plasma elimination half-life based on plasma concentrations of lifitegrast, but reported to be relatively short in rat I.V. pharmacokinetics study.
Lifitegrast binds to the integrin lymphocyte function-associated antigen-1 (LFA-1), a cell surface protein found on leukocytes and blocks the interaction of LFA-1 with its cognate ligand intercellular adhesion molecule-1 (ICAM-1). ICAM-1 may be overexpressed in corneal and conjunctival tissues in dry eye disease. LFA-1/ICAM-1 interaction can contribute to the formation of an immunological synapse resulting in T-cell proliferation/activation and migration to target tissues. In vitro studies demonstrated that lifitegrast may inhibit T-cell adhesion to ICAM-1 in a human T-cell line and may inhibit secretion of inflammatory cytokines, inflammatory mediators, chemokines, TNF-, and IL-1 in human peripheral blood mononuclear cells.
Related Excipient Companies

Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
61
PharmaCompass offers a list of Lifitegrast API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Lifitegrast manufacturer or Lifitegrast supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Lifitegrast manufacturer or Lifitegrast supplier.
PharmaCompass also assists you with knowing the Lifitegrast API Price utilized in the formulation of products. Lifitegrast API Price is not always fixed or binding as the Lifitegrast Price is obtained through a variety of data sources. The Lifitegrast Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Xiidra manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Xiidra, including repackagers and relabelers. The FDA regulates Xiidra manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Xiidra API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Xiidra manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Xiidra supplier is an individual or a company that provides Xiidra active pharmaceutical ingredient (API) or Xiidra finished formulations upon request. The Xiidra suppliers may include Xiidra API manufacturers, exporters, distributors and traders.
click here to find a list of Xiidra suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Xiidra DMF (Drug Master File) is a document detailing the whole manufacturing process of Xiidra active pharmaceutical ingredient (API) in detail. Different forms of Xiidra DMFs exist exist since differing nations have different regulations, such as Xiidra USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Xiidra DMF submitted to regulatory agencies in the US is known as a USDMF. Xiidra USDMF includes data on Xiidra's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Xiidra USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Xiidra suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Xiidra Drug Master File in Korea (Xiidra KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Xiidra. The MFDS reviews the Xiidra KDMF as part of the drug registration process and uses the information provided in the Xiidra KDMF to evaluate the safety and efficacy of the drug.
After submitting a Xiidra KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Xiidra API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Xiidra suppliers with KDMF on PharmaCompass.
A Xiidra written confirmation (Xiidra WC) is an official document issued by a regulatory agency to a Xiidra manufacturer, verifying that the manufacturing facility of a Xiidra active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Xiidra APIs or Xiidra finished pharmaceutical products to another nation, regulatory agencies frequently require a Xiidra WC (written confirmation) as part of the regulatory process.
click here to find a list of Xiidra suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Xiidra as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Xiidra API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Xiidra as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Xiidra and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Xiidra NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Xiidra suppliers with NDC on PharmaCompass.
Xiidra Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Xiidra GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Xiidra GMP manufacturer or Xiidra GMP API supplier for your needs.
A Xiidra CoA (Certificate of Analysis) is a formal document that attests to Xiidra's compliance with Xiidra specifications and serves as a tool for batch-level quality control.
Xiidra CoA mostly includes findings from lab analyses of a specific batch. For each Xiidra CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Xiidra may be tested according to a variety of international standards, such as European Pharmacopoeia (Xiidra EP), Xiidra JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Xiidra USP).
