
Synopsis
Synopsis
0
JDMF
0
NDC API
0
VMF
0
FDA Orange Book

0
Canada

0
Australia

0
South Africa

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
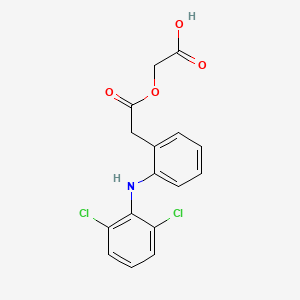

1. 2-((2,6-dichlorophenyl)amino)phenylacetoxyacetic Acid
2. Airtal
3. Airtal Difucrem
4. Aital
5. Beofenac
6. Biofenac
7. Bristaflam
8. Clanza Cr
9. Falcol
10. Falcol Difucrem
11. Gerbin
12. Gerbin Difucrem
13. Preservex
14. Sanein
1. 89796-99-6
2. Preservex
3. Aceclofenaco
4. Aceclofenacum
5. Aceclofenac Betadex
6. Chebi:31159
7. 2-[2-[2-(2,6-dichloroanilino)phenyl]acetyl]oxyacetic Acid
8. Mfcd00864296
9. Airtal
10. Rpk779r03h
11. 2-(2-(2-((2,6-dichlorophenyl)amino)phenyl)acetoxy)acetic Acid
12. Ncgc00016957-01
13. Glycolic Acid [o-(2,6-dichloroanilino)phenyl]acetate Ester
14. 2-(2-(2-(2,6-dichlorophenylamino)phenyl)acetoxy)acetic Acid
15. Glycolic Acid, (o-(2,6-dichloroanilino)phenyl)acetate (ester)
16. 2-((2,6-dichlorophenyl)amino)benzeneacetic Acid Carboxymethyl Ester
17. 2-[(2,6-dichlorophenyl)amino]benzeneacetic Acid Carboxymethyl Ester
18. Cas-89796-99-6
19. Dsstox_cid_25522
20. Dsstox_rid_80928
21. Dsstox_gsid_45522
22. 2-[(2',6'-dichlorophenyl)amino]phenylacetoxyacetic Acid
23. Aceclofenacum [latin]
24. Aceclofenaco [spanish]
25. Hifenac
26. 2-[(2-{2-[(2,6-dichlorophenyl)amino]phenyl}acetyl)oxy]acetic Acid
27. Benzeneacetic Acid, 2-((2,6-dichlorophenyl)amino)-, Carboxymethyl Ester
28. Smr000718629
29. Sr-01000802972
30. Aceclofenac [inn:ban]
31. Brn 4884476
32. Unii-rpk779r03h
33. Cincofen
34. [({2-[(2,6-dichlorophenyl)amino]phenyl}acetyl)oxy]acetic Acid
35. Clanza
36. Benzeneacetic Acid, 2-[(2,6-dichlorophenyl)amino]-, Carboxymethyl Ester
37. (2-{2-[(2,6-dichlorophenyl)amino]phenyl}acetoxy)acetic Acid
38. Aceclofenac (tn)
39. Prestwick_772
40. 2-(o-(2,6-dichloranilino)phenylacetoxy)essigsaeure
41. Pr-82/3
42. Aceclofenac (jan/inn)
43. Aceclofenac [mi]
44. Prestwick0_000175
45. Prestwick1_000175
46. Prestwick2_000175
47. Prestwick3_000175
48. Aceclofenac [inn]
49. Aceclofenac [jan]
50. Aceclofenac [mart.]
51. Schembl25734
52. Aceclofenac [who-dd]
53. Bspbio_000069
54. Mls001032069
55. Mls001304028
56. Mls002154226
57. Chembl93645
58. Spbio_001990
59. Bpbio1_000077
60. Dtxsid7045522
61. Aceclofenac, >=98% (hplc)
62. Aceclofenac [ep Monograph]
63. Aceclofenac For Peak Identification
64. Hms1568d11
65. Hms2090g07
66. Hms2095d11
67. Hms2231b03
68. Hms3371a10
69. Hms3712d11
70. Hms3873i03
71. Bcp11932
72. Hy-b0634
73. Zinc3805798
74. Tox21_110710
75. Aceclofenac Betadex [who-dd]
76. Bbl010788
77. Bdbm50109016
78. S4835
79. Stk594349
80. Akos005516194
81. Tox21_110710_1
82. Ab07468
83. Ac-5282
84. Aceclofenac 1.0 Mg/ml In Acetonitrile
85. Ccg-213835
86. Db06736
87. Ks-5033
88. Ncgc00016957-02
89. Ncgc00016957-03
90. Ncgc00016957-05
91. Ba164135
92. Aceclofenac 100 Microg/ml In Acetonitrile
93. Ft-0621715
94. D01545
95. 796a996
96. Q481757
97. 2-(2,6-dichlorophenylamine)phenylacetoxyacetic Acid
98. Sr-01000802972-2
99. Sr-01000802972-3
100. 2-[2-(2,6-dichloroanilino)phenylacetoxy]acetic Acid
101. Brd-k68538666-001-03-2
102. Aceclofenac For Peak Identification, European Pharmacopoeia (ep) Reference Standard
| Molecular Weight | 354.2 g/mol |
|---|---|
| Molecular Formula | C16H13Cl2NO4 |
| XLogP3 | 4.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 7 |
| Exact Mass | 353.0221633 g/mol |
| Monoisotopic Mass | 353.0221633 g/mol |
| Topological Polar Surface Area | 75.6 Ų |
| Heavy Atom Count | 23 |
| Formal Charge | 0 |
| Complexity | 411 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Aceclofenac is indicated for the relief of pain and inflammation in osteoarthritis, rheumatoid arthritis and ankylosing spondylitis.
Aceclofenac is a NSAID that inhibits both isoforms of COX enzyme, a key enzyme involved in the inflammatory cascade. COX-1 enzyme is a constitutive enzyme involved in prostacyclin production and protective functions of gastric mucosa whereas COX-2 is an inducible enzyme involved in the production of inflammatory mediators in response to inflammatory stimuli. Aceclofenac displays more selectivity towards COX-2 (IC50 of 0.77uM) than COX-1 (IC50 of >100uM), which promotes its gastric tolerance compared to other NSAIDs. The primary metabolite, 4'-hydroxyaceclofenac, also minimally inhibits COX-2 with IC50 value of 36uM. Although the mode of action of aceclofenac is thought to mainly arise from the inhibition of synthesis of prostaglandins (PGE2), aceclofenac also inhibits the production of inflammatory cytokines, interleukins (IL-1, IL-6), and tumor necrosis factors (TNF). It is also reported that aceclofenac also affects the cell adhesion molecules from neutrophils. Aceclofenac also targets the synthesis of glycosaminoglycan and mediates chrondroprotective effects.
Anti-Inflammatory Agents, Non-Steroidal
Anti-inflammatory agents that are non-steroidal in nature. In addition to anti-inflammatory actions, they have analgesic, antipyretic, and platelet-inhibitory actions. They act by blocking the synthesis of prostaglandins by inhibiting cyclooxygenase, which converts arachidonic acid to cyclic endoperoxides, precursors of prostaglandins. Inhibition of prostaglandin synthesis accounts for their analgesic, antipyretic, and platelet-inhibitory actions; other mechanisms may contribute to their anti-inflammatory effects. (See all compounds classified as Anti-Inflammatory Agents, Non-Steroidal.)
M01AB16
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
M - Musculo-skeletal system
M01 - Antiinflammatory and antirheumatic products
M01A - Antiinflammatory and antirheumatic products, non-steroids
M01AB - Acetic acid derivatives and related substances
M01AB16 - Aceclofenac
M - Musculo-skeletal system
M02 - Topical products for joint and muscular pain
M02A - Topical products for joint and muscular pain
M02AA - Antiinflammatory preparations, non-steroids for topical use
M02AA25 - Aceclofenac
Absorption
Aceclofenac is rapidly and completely absorbed from the gastrointestinal tract and circulates mainly as unchanged drug following oral administration. Peak plasma concentrations are reached around 1.25 to 3 hours post-ingestion, and the drug penetrates into the synovial fluid where the concentration may reach up to 60% of that in the plasma. There is no accumulation in regular dosing, with similar maximum plasma concentration (Cmax) and time to reach peak plasma concentration (Tmax) after single and multiple doses.
Route of Elimination
The main route of elimination is via the urine where the elimination accounts for 70-80% of clearance of the drug. Approximately two thirds of the administered dose is excreted via the urine, mainly as glucuronidated and hydroxylated forms of aceclofenac. About 20% of the dose is excreted into feces.
Volume of Distribution
The volume of distribution is approximately 25 L.
Clearance
The mean clearance rate is approximately 5 L/h.
4'-hydroxyaceclofenac is the main metabolite detected in plasma however other minor metabolites include diclofenac, 5-hydroxyaceclofenac, 5-hydroxydiclofenac, and 4'-hydroxydiclofenac. It is probable that the metabolism of aceclofenac is mediated by CYP2C9.
Aceclofenac has known human metabolites that include 4'-hydroxy-aceclofenac, 5-hydroxy-aceclofenac, and diclofenac.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
The mean plasma elimination half-life is approximately 4 hours.
Through COX-2 inhibition, aceclofenac downregulates the production of various inflammatory mediators including prostaglandin E2 (PGE2), IL-1, and TNF from the arachidonic acid (AA) pathway. Inhibition of IL-6 is thought to be mediated by diclofenac converted from aceclofenac. Suppressed action of inflammatory cytokines decreases the production of reactive oxygen species. Aceclofenac is shown to decreased production of nitrous oxide in human articular chondrocytes. In addition, aceclofenac interferes with neutrophil adhesion to endothelium by decreasing the expression of L-selectin (CD62L), which is a cell adhesion molecule expressed on lymphocytes. Aceclofenac is proposed to stimulate the synthesis of glycosaminoglycan in human osteoarthritic cartilage which may be mediated through its inhibitory action on IL-1 production and activity. The chrondroprotective effects are generated by 4'-hydroxyaceclofenac which suppresses IL-1 mediated production of promatrix metalloproteinase-1 and metalloproteinase-3 and interferes with the release of proteoglycan from chrondrocytes.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
57
PharmaCompass offers a list of Aceclofenac API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Aceclofenac manufacturer or Aceclofenac supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Aceclofenac manufacturer or Aceclofenac supplier.
PharmaCompass also assists you with knowing the Aceclofenac API Price utilized in the formulation of products. Aceclofenac API Price is not always fixed or binding as the Aceclofenac Price is obtained through a variety of data sources. The Aceclofenac Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Zerodol manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Zerodol, including repackagers and relabelers. The FDA regulates Zerodol manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Zerodol API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Zerodol manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Zerodol supplier is an individual or a company that provides Zerodol active pharmaceutical ingredient (API) or Zerodol finished formulations upon request. The Zerodol suppliers may include Zerodol API manufacturers, exporters, distributors and traders.
click here to find a list of Zerodol suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Zerodol DMF (Drug Master File) is a document detailing the whole manufacturing process of Zerodol active pharmaceutical ingredient (API) in detail. Different forms of Zerodol DMFs exist exist since differing nations have different regulations, such as Zerodol USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Zerodol DMF submitted to regulatory agencies in the US is known as a USDMF. Zerodol USDMF includes data on Zerodol's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Zerodol USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Zerodol suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Zerodol Drug Master File in Korea (Zerodol KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Zerodol. The MFDS reviews the Zerodol KDMF as part of the drug registration process and uses the information provided in the Zerodol KDMF to evaluate the safety and efficacy of the drug.
After submitting a Zerodol KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Zerodol API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Zerodol suppliers with KDMF on PharmaCompass.
A Zerodol CEP of the European Pharmacopoeia monograph is often referred to as a Zerodol Certificate of Suitability (COS). The purpose of a Zerodol CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Zerodol EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Zerodol to their clients by showing that a Zerodol CEP has been issued for it. The manufacturer submits a Zerodol CEP (COS) as part of the market authorization procedure, and it takes on the role of a Zerodol CEP holder for the record. Additionally, the data presented in the Zerodol CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Zerodol DMF.
A Zerodol CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Zerodol CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Zerodol suppliers with CEP (COS) on PharmaCompass.
A Zerodol written confirmation (Zerodol WC) is an official document issued by a regulatory agency to a Zerodol manufacturer, verifying that the manufacturing facility of a Zerodol active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Zerodol APIs or Zerodol finished pharmaceutical products to another nation, regulatory agencies frequently require a Zerodol WC (written confirmation) as part of the regulatory process.
click here to find a list of Zerodol suppliers with Written Confirmation (WC) on PharmaCompass.
Zerodol Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Zerodol GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Zerodol GMP manufacturer or Zerodol GMP API supplier for your needs.
A Zerodol CoA (Certificate of Analysis) is a formal document that attests to Zerodol's compliance with Zerodol specifications and serves as a tool for batch-level quality control.
Zerodol CoA mostly includes findings from lab analyses of a specific batch. For each Zerodol CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Zerodol may be tested according to a variety of international standards, such as European Pharmacopoeia (Zerodol EP), Zerodol JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Zerodol USP).
