

Synopsis
Synopsis
0
VMF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
Annual Reports
NA
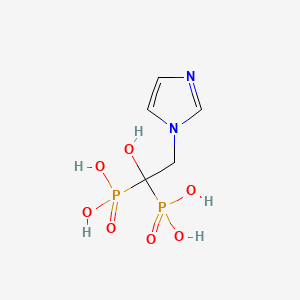

1. 2-(imidazol-1-yl)-1-hydroxyethylidene-1,1-bisphosphonic Acid
2. Cgp 42'446
3. Cgp 42446
4. Cgp 42446a
5. Cgp-42'446
6. Cgp-42446
7. Cgp42'446
8. Cgp42446
9. Zoledronate
10. Zoledronic Acid Anhydrous
11. Zometa
1. Zoledronate
2. 118072-93-8
3. Zometa
4. Reclast
5. Aclasta
6. (1-hydroxy-2-(1h-imidazol-1-yl)ethane-1,1-diyl)diphosphonic Acid
7. Cgp 42446
8. (1-hydroxy-2-imidazol-1-ylethylidene)diphosphonic Acid
9. Zoledronic Acid Anhydrous
10. Anhydrous Zoledronic Acid
11. (1-hydroxy-2-imidazol-1-yl-1-phosphonoethyl)phosphonic Acid
12. Phosphonic Acid, [1-hydroxy-2-(1h-imidazol-1-yl)ethylidene]bis-
13. Zol
14. [1-hydroxy-2-(1h-imidazol-1-yl)ethane-1,1-diyl]bis(phosphonic Acid)
15. Orazol
16. Zol 446
17. Zoledronic Acid (inn)
18. Cgp-42446
19. Reclast (tn)
20. Zometa (tn)
21. Chembl924
22. Zoledronic Acid Teva
23. Zoledronic Acid, Anhydrous
24. Nsc-721517
25. Zoledronic Acid Medac
26. Chebi:46557
27. [1-hydroxy-2-(1h-imidazol-1-yl)-1-phosphonoethyl]phosphonic Acid
28. 70hz18ph24
29. Ncgc00159521-02
30. (1-hydroxy-2-(1h-imidazol-1-yl)ethylidene)bisphosphonic Acid
31. Cgp-42446a
32. Zoledronate Hydrate
33. Zoledronic Acid [usan:inn:ban]
34. Phosphonic Acid, (1-hydroxy-2-(1h-imidazol-1-yl)ethylidene)bis-
35. Zoladrona Acid Mylan
36. Zoledronic
37. Zoledronic Acid Accord
38. Zoledronic Acid [inn]
39. Zomera
40. 1-hydroxy-2-(1h-imidazol-1-yl)ethane-1,1-diyldiphosphonic Acid
41. Bisphosphonate 3
42. Zometa (novartis)
43. Aclasta And Reclast
44. C5h10n2o7p2
45. [1-hydroxy-2-(1h-imidazol-1-yl)ethylidene]bisphosphonic Acid
46. Zoledronic-acid
47. Unii-70hz18ph24
48. Bph 91
49. [1-hydroxy-2-(1h-imidazol-1-yl)-ethylidene]bisphosphonic Acid
50. Dsstox_cid_22668
51. Dsstox_rid_80065
52. Zoledronic Acid, Zoledronate
53. Bidd:pxr0134
54. Dsstox_gsid_42668
55. Schembl19054
56. Zoledronic Acid [mi]
57. Bidd:gt0292
58. Zoledronic Acid (zoledronate)
59. Gtpl3177
60. Jmc515594 Compound 55
61. Dtxsid0042668
62. Bdbm12578
63. Cgp42446a
64. Zoledronic Acid [who-dd]
65. Hms2089o09
66. Bcp22750
67. Cgp-4244
68. Zinc3803652
69. Tox21_111739
70. Mfcd00867791
71. Nsc721517
72. S1314
73. Stl452893
74. Akos005145739
75. Ab07564
76. Ac-1092
77. Cs-1829
78. Db00399
79. Hs-0091
80. Nsc 721517
81. Ncgc00159521-03
82. Ncgc00159521-04
83. Ncgc00159521-05
84. Ncgc00159521-09
85. Ncgc00159521-18
86. Hy-13777
87. Cas-118072-93-8
88. Ft-0601384
89. Z0031
90. D08689
91. H11422
92. S00092
93. Ab01273947-01
94. Ab01273947-02
95. Ab01273947-03
96. Ab01273947_04
97. 072z938
98. A803876
99. Q218507
100. Sr-05000001436
101. Q-201946
102. Sr-05000001436-1
103. 1-hydroxy-2-(1-imidazolyl)ethane-1,1-diphosphonic Acid
104. Z1691545083
105. (1-hydroxy-2-(1h-imidazol-1-yl)ethane-1,1-diyl)diphosphonicacid
| Molecular Weight | 272.09 g/mol |
|---|---|
| Molecular Formula | C5H10N2O7P2 |
| XLogP3 | -4.3 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 4 |
| Exact Mass | 271.99632466 g/mol |
| Monoisotopic Mass | 271.99632466 g/mol |
| Topological Polar Surface Area | 153 Ų |
| Heavy Atom Count | 16 |
| Formal Charge | 0 |
| Complexity | 327 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 6 | |
|---|---|
| Drug Name | Reclast |
| Drug Label | Reclast contains zoledronic acid, a bisphosphonic acid which is an inhibitor of osteoclastic bone resorption. Zoledronic acid is designated chemically as (1-Hydroxy-2-imidazol-1-yl-phosphonoethyl) phosphonic acid monohydrate and its structural formul... |
| Active Ingredient | Zoledronic acid |
| Dosage Form | Injectable |
| Route | injection; Iv (infusion) |
| Strength | eq 5mg base/100ml; 5mg/100ml |
| Market Status | Prescription |
| Company | Novartis Pharms; Novartis |
| 2 of 6 | |
|---|---|
| Drug Name | Zoledronic acid |
| PubMed Health | Zoledronic Acid (Injection) |
| Drug Classes | Calcium Regulator |
| Drug Label | Zoledronic Acid Injection contains zoledronic acid, a bisphosphonic acid which is an inhibitor of osteoclastic bone resorption. Zoledronic acid is designated chemically as (1-Hydroxy-2-imidazol-1-yl-phosphonoethyl) phosphonic acid monohydrate and its... |
| Active Ingredient | Zoledronic acid |
| Dosage Form | Injectable |
| Route | injection; Iv (infusion); Injection; iv (infusion) |
| Strength | eq 5mg base/100ml; 5mg/100ml; 4mmg; eq 4mg base/100ml; eq 4mg base/5ml; 4mg/100ml; 4mg/5ml(0.8mg/ml); eq 4mg base/vial |
| Market Status | Tentative Approval; Prescription |
| Company | Pharmaceutics; Hospira; Gland Pharma; Teva Parenteral; Apotex; Hikma Farmaceutica; Usv North America; Acs Dobfar Info Sa; Pharmaforce; Cipla; Sun Pharma Global; Emcure Pharms; Pharms; Dr Reddys Labs; Agila Speclts; Actavis; Akorn |
| 3 of 6 | |
|---|---|
| Drug Name | Zometa |
| PubMed Health | Zoledronic Acid (Injection) |
| Drug Classes | Calcium Regulator |
| Drug Label | Zometa contains zoledronicacid, a bisphosphonic acid which is an inhibitor of osteoclastic bone resorption. Zoledronicacid is designated chemically as (1-Hydroxy-2-imidazol-1-yl-phosphonoethyl) phosphonic acid monohydrate and its structural formu... |
| Active Ingredient | Zoledronic acid |
| Dosage Form | Injectable |
| Route | Iv (infusion); iv (infusion) |
| Strength | eq 4mg base/100ml; eq 4mg base/5ml; eq 4mg base/vial |
| Market Status | Prescription |
| Company | Novartis |
| 4 of 6 | |
|---|---|
| Drug Name | Reclast |
| Drug Label | Reclast contains zoledronic acid, a bisphosphonic acid which is an inhibitor of osteoclastic bone resorption. Zoledronic acid is designated chemically as (1-Hydroxy-2-imidazol-1-yl-phosphonoethyl) phosphonic acid monohydrate and its structural formul... |
| Active Ingredient | Zoledronic acid |
| Dosage Form | Injectable |
| Route | injection; Iv (infusion) |
| Strength | eq 5mg base/100ml; 5mg/100ml |
| Market Status | Prescription |
| Company | Novartis Pharms; Novartis |
| 5 of 6 | |
|---|---|
| Drug Name | Zoledronic acid |
| PubMed Health | Zoledronic Acid (Injection) |
| Drug Classes | Calcium Regulator |
| Drug Label | Zoledronic Acid Injection contains zoledronic acid, a bisphosphonic acid which is an inhibitor of osteoclastic bone resorption. Zoledronic acid is designated chemically as (1-Hydroxy-2-imidazol-1-yl-phosphonoethyl) phosphonic acid monohydrate and its... |
| Active Ingredient | Zoledronic acid |
| Dosage Form | Injectable |
| Route | injection; Iv (infusion); Injection; iv (infusion) |
| Strength | eq 5mg base/100ml; 5mg/100ml; 4mmg; eq 4mg base/100ml; eq 4mg base/5ml; 4mg/100ml; 4mg/5ml(0.8mg/ml); eq 4mg base/vial |
| Market Status | Tentative Approval; Prescription |
| Company | Pharmaceutics; Hospira; Gland Pharma; Teva Parenteral; Apotex; Hikma Farmaceutica; Usv North America; Acs Dobfar Info Sa; Pharmaforce; Cipla; Sun Pharma Global; Emcure Pharms; Pharms; Dr Reddys Labs; Agila Speclts; Actavis; Akorn |
| 6 of 6 | |
|---|---|
| Drug Name | Zometa |
| PubMed Health | Zoledronic Acid (Injection) |
| Drug Classes | Calcium Regulator |
| Drug Label | Zometa contains zoledronicacid, a bisphosphonic acid which is an inhibitor of osteoclastic bone resorption. Zoledronicacid is designated chemically as (1-Hydroxy-2-imidazol-1-yl-phosphonoethyl) phosphonic acid monohydrate and its structural formu... |
| Active Ingredient | Zoledronic acid |
| Dosage Form | Injectable |
| Route | Iv (infusion); iv (infusion) |
| Strength | eq 4mg base/100ml; eq 4mg base/5ml; eq 4mg base/vial |
| Market Status | Prescription |
| Company | Novartis |
Zoledronic acid is indicated to treat hypercalcemia of malignancy, multiple myeloma, bone metastases from solid tumors, osteoporosis in men and postmenopausal women, glucocorticoid induced osteoporosis, and Paget's disease of bone in men and women. Zoledronic acid is also indicated for the prevention of osteoporosis in post menopausal women and glucocorticoid induced osteoporosis.
Prevention of skeletal-related events and treatment of tumour-induced hypercalcaemia.
Prevention of skeletal-related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in adult patients with advanced malignancies involving bone.
Treatment of adult patients with tumour-induced hypercalcaemia.
* 4 mg / 5 ml and 4 mg / 100 ml: :
- Prevention of skeletal-related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in adult patients with advanced malignancies involving bone.
- Treatment of adult patients with tumour-induced hypercalcaemia (TIH).
* 5 mg / 100 ml: :
Treatment of osteoporosis:
- in post-menopausal women;
- in men;
at increased risk of fracture, including those with a recent low-trauma hip fracture.
Treatment of osteoporosis associated with long-term systemic glucocorticoid therapy:
- in post-menopausal women;
- in men;
at increased risk of fracture.
Treatment of Paget's disease of the bone in adults.
Prevention of skeletal related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in adult patients with advanced malignancies involving bone.
Treatment of adult patients with tumour-induced hypercalcaemia (TIH).
- Prevention of skeletal related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in adult patients with advanced malignancies involving bone;
- treatment of adult patients with tumour-induced hypercalcaemia (TIH).
Treatment of osteoporosis:
- in post-menopausal women;
- in men;
at increased risk of fracture, including those with a recent low-trauma hip fracture.
Treatment of osteoporosis associated with long-term systemic glucocorticoid therapy in post-menopausal women and in men at increased risk of fracture.
Treatment of Paget's disease of the bone.
Prevention of skeletal related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in adult patients with advanced malignancies involving bone.
Treatment of adult patients with tumour-induced hypercalcaemia (TIH).
Treatment of osteoporosis:
- in post-menopausal women;
- in men;
at increased risk of fracture including those with a recent low-trauma hip fracture.
Treatment of osteoporosis associated with long-term systemic glucocorticoid therapy:
- in post-menopausal women;
- in men;
at increased risk of fracture.
Treatment of Pagets disease of the bone in adults.
- Prevention of skeletal related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in patients with advanced malignancies involving bone;
- treatment of tumour-induced hypercalcaemia (TIH);
- prevention of skeletal related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in patients with advanced malignancies involving bone;
- treatment of tumour-induced hypercalcaemia (TIH);
- prevention of skeletal related events (pathological fractures, spinal compression, radiation or surgery to bone, or tumour-induced hypercalcaemia) in adult patients with advanced malignancies involving bone;
- treatment of adult patients with tumour-induced hypercalcaemia (TIH).
Treatment of osteoporosis
- in post-menopausal women
- in adult men
at increased risk of fracture, including those with recent low-trauma hip fracture.
Treatment of osteoporosis associated with long-term systemic glucocorticoid therapy
- in post-menopausal women
- in adult men
at increased risk of fracture.
Treatment of Paget's disease of the bone in adults.
Osteogenesis imperfecta, Prevention of fracture and bone loss in postmenopausal women with early-stage breast cancer treated with aromatase inhibitors, Prevention of skeletal related events in patients with advanced malignancies involving bone, Tumour-induced hypercalcaemia
Treatment of osteoporosis, Treatment of Pagets disease of the bone
Zoledronic acid is a third generation, nitrogen containing bisphosphonate that inhibits osteoclast function and prevents bone resorption. The therapeutic window is wide as patients are unlikely to suffer severe effects from overdoses and the duration of action is long. Patients should be counselled regarding the risk of electrolyte deficiencies, renal impairment, osteonecrosis of the jaw, atypical femoral fractures, bronchoconstriction, hepatic impairment, hypocalcemia, and embryo-fetal toxicity.
Bone Density Conservation Agents
Agents that inhibit BONE RESORPTION and/or favor BONE MINERALIZATION and BONE REGENERATION. They are used to heal BONE FRACTURES and to treat METABOLIC BONE DISEASES such as OSTEOPOROSIS. (See all compounds classified as Bone Density Conservation Agents.)
M05BA08
M05BA08
M05BA08
M05BA08
M05BA08
M05BA08
M05BA08
M05BA08
M05BA08
M05BA08
M05BA08
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
M - Musculo-skeletal system
M05 - Drugs for treatment of bone diseases
M05B - Drugs affecting bone structure and mineralization
M05BA - Bisphosphonates
M05BA08 - Zoledronic acid
Absorption
A 4mg intravenous dose reaches a Cmax of 37078.5ng/mL, with a Tmax of 0.3170.014h, and an AUC of 788181ng\*h/mL. A 5mg intravenous dose reaches a Cmax of 47176.1ng/mL, with a Tmax of 0.3680.005h, and an AUC of 917226ng\*h/mL.
Route of Elimination
Zoledronic acid is 39 16% eliminated in the urine as the unmetabolized parent drug.
Clearance
Zoledronic acid has a renal clearance of 3.7 2.0 L/h.
Zoledronic acid is not metabolized _in vivio_.
Zoledronic acid has a terminal elimination half life of 146 hours.
Bisphosphonates are taken into the bone where they bind to hydroxyapatite. Bone resorption by osteoclasts causes local acidification, releasing the bisphosphonate, which is taken into the osteoclast by fluid-phase endocytosis. Endocytic vesicles become acidified, releasing bisphosphonates into the cytosol of osteoclasts where they act. Osteoclasts mediate resorption of bone. When osteoclasts bind to bone they form podosomes, ring structures of F-actin. Etidronic acid also inhibits V-ATPases in the osteoclast, though the exact subunits are unknown, preventing F-actin from forming podosomes. Disruption of the podosomes causes osteoclasts to detach from bones, preventing bone resorption. Nitrogen containing bisphosphonates such as zoledronate are known to induce apoptosis of hematopoietic tumor cells by inhibiting the components of the mevalonate pathway farnesyl diphosphate synthase, farnesyl diphosphate, and geranylgeranyl diphosphate. These components are essential for post-translational prenylation of GTP-binding proteins like Rap1. The lack of prenylation of these proteins interferes with their function, and in the case of Rap1, leads to apoptosis. zoledronate also activated caspases which further contribute to apoptosis.
 Polpharma is a Polish CDMO of APIs and a significant European API producer, delivering products to companies worldwide.
Polpharma is a Polish CDMO of APIs and a significant European API producer, delivering products to companies worldwide.
GDUFA
DMF Review : Complete
Rev. Date : 2016-06-03
Pay. Date : 2015-08-27
DMF Number : 24306
Submission : 2010-10-13
Status : Active
Type : II
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 21344
Submission : 2008-02-18
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2013-02-15
Pay. Date : 2013-01-28
DMF Number : 24469
Submission : 2010-12-24
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 19749
Submission : 2006-08-24
Status : Inactive
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2013-05-16
Pay. Date : 2013-05-02
DMF Number : 20052
Submission : 2006-12-16
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2013-01-31
Pay. Date : 2012-12-31
DMF Number : 19523
Submission : 2006-06-15
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 18964
Submission : 2005-11-18
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 20574
Submission : 2007-05-31
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2012-11-23
Pay. Date : 2012-11-13
DMF Number : 19701
Submission : 2006-08-22
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 21099
Submission : 2007-12-03
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Certificate Number : R0-CEP 2022-203 - Rev 00
Status : Valid
Issue Date : 2023-02-10
Type : Chemical
Substance Number : 2743
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : CEP 2020-205 - Rev 03
Status : Valid
Issue Date : 2024-09-03
Type : Chemical
Substance Number : 2743

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : CEP 2021-181 - Rev 00
Status : Valid
Issue Date : 2023-10-27
Type : Chemical
Substance Number : 2743

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R0-CEP 2019-349 - Rev 01
Status : Valid
Issue Date : 2022-08-11
Type : Chemical
Substance Number : 2743

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R0-CEP 2021-177 - Rev 00
Status : Valid
Issue Date : 2022-08-26
Type : Chemical
Substance Number : 2743

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R0-CEP 2019-281 - Rev 00
Status : Valid
Issue Date : 2021-04-07
Type : Chemical
Substance Number : 2743

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R0-CEP 2021-269 - Rev 00
Status : Valid
Issue Date : 2023-07-24
Type : Chemical
Substance Number : 2743

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R0-CEP 2020-018 - Rev 01
Status : Valid
Issue Date : 2022-01-14
Type : Chemical
Substance Number : 2743


Certificate Number : CEP 2018-191 - Rev 03
Status : Valid
Issue Date : 2024-07-23
Type : Chemical
Substance Number : 2743


Certificate Number : CEP 2019-314 - Rev 01
Status : Valid
Issue Date : 2024-05-30
Type : Chemical
Substance Number : 2743

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]  Polpharma is a Polish CDMO of APIs and a significant European API producer, delivering products to companies worldwide.
Polpharma is a Polish CDMO of APIs and a significant European API producer, delivering products to companies worldwide.
Registration Number : 225MF10034
Registrant's Address : 19 Pellinska Str. 83-200 Starogard Gdanski POLAND
Initial Date of Registration : 2013-02-20
Latest Date of Registration : 2017-06-15
Zoledronic Acid Hydrate "Teva"
Registration Number : 225MF10045
Registrant's Address : Neot-Hovav Eco-Industrial Park, Emek Sara P. O. Box 2049 Be'er Sheva 8412316, Israel
Initial Date of Registration : 2013-03-05
Latest Date of Registration : 2020-08-24

Registration Number : 225MF10022
Registrant's Address : Viale dei Laboratori 54, 20092 Cinisello Balsamo (Milano), Italy
Initial Date of Registration : 2013-02-06
Latest Date of Registration : 2013-02-06

Registration Number : 225MF10031
Registrant's Address : 48, Kandivli Industrial Estate, Kandivli (W), Mumbai 400 067, India
Initial Date of Registration : 2013-02-13
Latest Date of Registration : 2013-02-13

Registration Number : 225MF10053
Registrant's Address : Natco House, Road No. 2, Banjara Hills,Hyderabad 500034, Telangana, India
Initial Date of Registration : 2013-03-12
Latest Date of Registration : 2014-05-19

Registration Number : 225MF10012
Registrant's Address : 2-1-1 Marunouchi, Chiyoda-ku, Tokyo
Initial Date of Registration : 2013-01-30
Latest Date of Registration : 2013-11-14

Registration Number : 225MF10047
Registrant's Address : No. 1, Nan-Ke 8th Road, Shan-Hua, Tainan 74144 Taiwan
Initial Date of Registration : 2013-03-11
Latest Date of Registration : 2013-03-11

Registration Number : 227MF10043
Registrant's Address : Osaka Prefecture, Osaka City, Nishi Ward, Awaza 1-12-18
Initial Date of Registration : 2015-02-09
Latest Date of Registration : 2015-02-09

Registration Number : 225MF10058
Registrant's Address : No. 1191, Sec. 1, Chung Shan Rd. , Tachia, Taichung, Taiwan, R. O. C.
Initial Date of Registration : 2013-03-12
Latest Date of Registration : 2020-01-07

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Polpharma is a Polish CDMO of APIs and a significant European API producer, delivering products to companies worldwide.
Polpharma is a Polish CDMO of APIs and a significant European API producer, delivering products to companies worldwide.
NDC Package Code : 12658-0608
Start Marketing Date : 1995-12-06
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
NDC Package Code : 55111-813
Start Marketing Date : 2008-02-18
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
NDC Package Code : 62147-0125
Start Marketing Date : 2010-01-04
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
NDC Package Code : 63415-0532
Start Marketing Date : 2023-07-12
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 53104-7622
Start Marketing Date : 2015-01-01
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (25kg/25kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 14593-836
Start Marketing Date : 2022-02-14
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (5kg/5kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 68554-0037
Start Marketing Date : 1995-12-06
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 63850-7104
Start Marketing Date : 2016-01-20
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 65129-1144
Start Marketing Date : 2006-04-14
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 82920-012
Start Marketing Date : 2022-08-10
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
RLD : No
TE Code :
Brand Name : ZOLEDRONIC ACID
Dosage Form : INJECTABLE;INTRAVENOUS
Dosage Strength : EQ 4MG BASE/100ML
Approval Date : 2018-11-19
Application Number : 204344
RX/OTC/DISCN : DISCN
RLD : No
TE Code :
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code :
Brand Name : ZOLEDRONIC ACID
Dosage Form : INJECTABLE;INTRAVENOUS
Dosage Strength : EQ 4MG BASE/5ML
Approval Date : 2013-03-04
Application Number : 91170
RX/OTC/DISCN : DISCN
RLD : No
TE Code :
RLD : No
TE Code : AP
Brand Name : ZOLEDRONIC ACID
Dosage Form : INJECTABLE;INTRAVENOUS
Dosage Strength : EQ 4MG BASE/5ML
Approval Date : 2014-09-04
Application Number : 202923
RX/OTC/DISCN : RX
RLD : No
TE Code : AP
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AP
Brand Name : ZOLEDRONIC ACID
Dosage Form : INJECTABLE;INTRAVENOUS
Dosage Strength : EQ 4MG BASE/5ML
Approval Date : 2013-03-12
Application Number : 201783
RX/OTC/DISCN : RX
RLD : No
TE Code : AP

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AP
Brand Name : ZOLEDRONIC ACID
Dosage Form : INJECTABLE;INTRAVENOUS
Dosage Strength : EQ 4MG BASE/5ML
Approval Date : 2017-12-29
Application Number : 207341
RX/OTC/DISCN : RX
RLD : No
TE Code : AP

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AP
Brand Name : ZOLEDRONIC ACID
Dosage Form : INJECTABLE;INTRAVENOUS
Dosage Strength : EQ 4MG BASE/5ML
Approval Date : 2013-08-05
Application Number : 202930
RX/OTC/DISCN : RX
RLD : No
TE Code : AP

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD :
TE Code :
Brand Name : ZOLEDRONIC ACID
Dosage Form : INJECTABLE;IV (INFUSION)
Dosage Strength : EQ 4MG BASE/100ML
Approval Date :
Application Number : 205749
RX/OTC/DISCN :
RLD :
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AP
Brand Name : ZOLEDRONIC ACID
Dosage Form : INJECTABLE;INTRAVENOUS
Dosage Strength : EQ 5MG BASE/100ML
Approval Date : 2013-04-05
Application Number : 202837
RX/OTC/DISCN : RX
RLD : No
TE Code : AP

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AP
Brand Name : ZOLEDRONIC ACID
Dosage Form : INJECTABLE;INTRAVENOUS
Dosage Strength : EQ 4MG BASE/5ML
Approval Date : 2014-11-24
Application Number : 91493
RX/OTC/DISCN : RX
RLD : No
TE Code : AP

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD :
TE Code :
Brand Name : ZOLEDRONIC ACID
Dosage Form : INJECTABLE; IV (INFUSION)
Dosage Strength : EQ 4MG BASE/VIAL
Approval Date :
Application Number : 78576
RX/OTC/DISCN :
RLD :
TE Code :

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Ethypharm is an international Pharma company with European roots manufacturing and commercializing essential drugs all over the world.
Ethypharm is an international Pharma company with European roots manufacturing and commercializing essential drugs all over the world.
Regulatory Info :
Registration Country : France
Brand Name :
Dosage Form : Solution for Infusion
Dosage Strength : 5MG/100ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : France
 Ethypharm is an international Pharma company with European roots manufacturing and commercializing essential drugs all over the world.
Ethypharm is an international Pharma company with European roots manufacturing and commercializing essential drugs all over the world.
Packaging :
Regulatory Info :
Dosage : Solution for Infusion
Dosage Strength : 5MG/100ML
Brand Name :
Approval Date :
Application Number :
Registration Country : France
 Ethypharm is an international Pharma company with European roots manufacturing and commercializing essential drugs all over the world.
Ethypharm is an international Pharma company with European roots manufacturing and commercializing essential drugs all over the world.
Regulatory Info :
Registration Country : France
Brand Name :
Dosage Form : Solution for Infusion
Dosage Strength : 4MG/100ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : France
 Ethypharm is an international Pharma company with European roots manufacturing and commercializing essential drugs all over the world.
Ethypharm is an international Pharma company with European roots manufacturing and commercializing essential drugs all over the world.
Packaging :
Regulatory Info :
Dosage : Solution for Infusion
Dosage Strength : 4MG/100ML
Brand Name :
Approval Date :
Application Number :
Registration Country : France
 Ethypharm is an international Pharma company with European roots manufacturing and commercializing essential drugs all over the world.
Ethypharm is an international Pharma company with European roots manufacturing and commercializing essential drugs all over the world.
Regulatory Info :
Registration Country : France
Brand Name :
Dosage Form : Vial
Dosage Strength : 4MG/5ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : France
 Ethypharm is an international Pharma company with European roots manufacturing and commercializing essential drugs all over the world.
Ethypharm is an international Pharma company with European roots manufacturing and commercializing essential drugs all over the world.
Packaging :
Regulatory Info :
Dosage : Vial
Dosage Strength : 4MG/5ML
Brand Name :
Approval Date :
Application Number :
Registration Country : France
Regulatory Info :
Registration Country : Iran
Brand Name : Zobis
Dosage Form : Vial
Dosage Strength : 4MG/5ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Iran
Packaging :
Regulatory Info :
Dosage : Vial
Dosage Strength : 4MG/5ML
Brand Name : Zobis
Approval Date :
Application Number :
Registration Country : Iran
Regulatory Info :
Registration Country : Iran
Brand Name : Clastoz
Dosage Form : Vial
Dosage Strength : 5MG/100ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Iran
Packaging :
Regulatory Info :
Dosage : Vial
Dosage Strength : 5MG/100ML
Brand Name : Clastoz
Approval Date :
Application Number :
Registration Country : Iran
 Naprod is a top player in Oncology & Anesthesia FDF through excellent innovation, discovery of new processes & high-quality production.
Naprod is a top player in Oncology & Anesthesia FDF through excellent innovation, discovery of new processes & high-quality production.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Lyophilised Injection
Dosage Strength : 4MG/Vial
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Naprod is a top player in Oncology & Anesthesia FDF through excellent innovation, discovery of new processes & high-quality production.
Naprod is a top player in Oncology & Anesthesia FDF through excellent innovation, discovery of new processes & high-quality production.
Packaging :
Regulatory Info :
Dosage : Lyophilised Injection
Dosage Strength : 4MG/Vial
Brand Name :
Approval Date :
Application Number :
Registration Country : India
 Naprod is a top player in Oncology & Anesthesia FDF through excellent innovation, discovery of new processes & high-quality production.
Naprod is a top player in Oncology & Anesthesia FDF through excellent innovation, discovery of new processes & high-quality production.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Liquid Injection
Dosage Strength : 4MG/5ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Naprod is a top player in Oncology & Anesthesia FDF through excellent innovation, discovery of new processes & high-quality production.
Naprod is a top player in Oncology & Anesthesia FDF through excellent innovation, discovery of new processes & high-quality production.
Packaging :
Regulatory Info :
Dosage : Liquid Injection
Dosage Strength : 4MG/5ML
Brand Name :
Approval Date :
Application Number :
Registration Country : India
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Iran
Brand Name : Zobis
Dosage Form : Injectable
Dosage Strength : 4MG/5ML
Packaging : Vial
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Iran

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging : Vial
Regulatory Info :
Dosage : Injectable
Dosage Strength : 4MG/5ML
Brand Name : Zobis
Approval Date :
Application Number :
Registration Country : Iran

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : India
Brand Name :
Dosage Form : Lyo.Inj
Dosage Strength : 4MG
Packaging :
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info : Generic
Dosage : Lyo.Inj
Dosage Strength : 4MG
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Egypt
Brand Name : Osteozomet
Dosage Form : Intravenous Infusion
Dosage Strength : 4MG/5ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Egypt

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Intravenous Infusion
Dosage Strength : 4MG/5ML
Brand Name : Osteozomet
Approval Date :
Application Number :
Registration Country : Egypt

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Dosage Form : Tablet, Orodispersible Tablet
Grade : Oral
Category : API Stability Enhancers, Chewable & Orodispersible Aids, Fillers, Diluents & Binders
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
We have 9 companies offering Zoledronic Acid
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
