
Synopsis
Synopsis
0
VMF
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
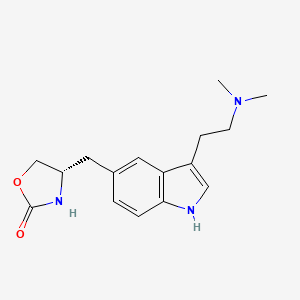

1. 311c90
2. 4-((3-(2-(dimethylamino)ethyl)-1h-indol-5-yl)methyl)-2-oxazolidinone
3. Ascotop
4. Flezol
5. Zomig
6. Zomigoro
1. 139264-17-8
2. Zomig
3. Zomigoro
4. 311c90
5. Zomigon
6. Flezol
7. Zomig-zmt
8. C16h21n3o2
9. Bw-311c90
10. (s)-4-((3-(2-(dimethylamino)ethyl)-1h-indol-5-yl)methyl)oxazolidin-2-one
11. Zolmitriptan (zomig)
12. (s)-4-[3-(2-dimethylamino-ethyl)-1h-indol-5-ylmethyl]-oxazolidin-2-one
13. Nsc-760383
14. Cvt-427
15. 2fs66th3yw
16. Chembl1185
17. (s)-4-((3-(2-(dimethylamino)ethyl)indol-5-yl)methyl)-2-oxazolidinone
18. Chebi:10124
19. (s)-4-((3-(2-(dimethylamino)ethyl)-1h-indol-5-yl)methyl)-2-oxazolidinone
20. (4s)-4-[[3-[2-(dimethylamino)ethyl]-1h-indol-5-yl]methyl]-1,3-oxazolidin-2-one
21. Zomig Nasal Spray
22. Zomig Zmt
23. Ascotopand
24. Ascotop
25. Zolmitriptan [usan]
26. 4-[[3-(2-dimethylaminoethyl)-1h-indol-5-yl]methyl]oxazolidin-2-one
27. (4s)-4-[[3-[2-(dimethylamino)ethyl]-1h-indol-5-yl]methyl]-2-oxazolidinone
28. (s)-4-[[3-[2-(dimethylamino)ethyl]-1h-indol-5-yl]methyl]-2-oxazolidinone
29. (4s)-4-({3-[2-(dimethylamino)ethyl]-1h-indol-5-yl}methyl)-1,3-oxazolidin-2-one
30. 2-oxazolidinone, 4-((3-(2-(dimethylamino)ethyl)-1h-indol-5-yl)methyl)-, (s)-
31. Dsstox_cid_25933
32. Dsstox_rid_81232
33. Dsstox_gsid_45933
34. 311-c-90
35. Zipton
36. Zolmitriptane
37. Zolmitriptanum
38. Zominat
39. Rapimelt
40. (4s)-4-((3-(2-(dimethylamino)ethyl)-1h-indol-5-yl)methyl)-2-oxazolidinone
41. Smr000449310
42. Cas-139264-17-8
43. Zomig (tn)
44. Zolmitriptan Rapidfilm
45. Unii-2fs66th3yw
46. 4-((3-(2-(dimethylamino)ethyl)-1h-indol-5-yl)methyl)-2-oxazolidinone
47. Zolmitriptan [usan:inn:ban]
48. Ncgc00095155-01
49. Mfcd00871503
50. Ks-5072
51. Spectrum2_000728
52. Zolmitriptan [mi]
53. Zolmitriptan [inn]
54. Zolmitriptan [jan]
55. Gtpl60
56. (4s)-4-[[3-(2-dimethylaminoethyl)-1h-indol-5-yl]methyl]-1,3-oxazolidin-2-one
57. Zolmitriptan [vandf]
58. Schembl33336
59. Zolmitriptan [mart.]
60. Mls000758208
61. Mls001424172
62. Bidd:gt0040
63. Spectrum1505281
64. Zolmitriptan [usp-rs]
65. Zolmitriptan [who-dd]
66. Spbio_000656
67. Zolmitriptan (jan/usp/inn)
68. Dtxsid8045933
69. Zinc15515
70. Zolmitriptan, >=98% (hplc)
71. Hms1922b04
72. Hms2052k17
73. Hms2093o14
74. Hms2235i22
75. Hms3714h11
76. Hms3884a06
77. Pharmakon1600-01505281
78. Zolmitriptan [orange Book]
79. Zolmitriptan [ep Monograph]
80. Act04392
81. Bcp10513
82. Hy-b0229
83. Tox21_111455
84. Zolmitriptan [usp Monograph]
85. Bdbm50033383
86. Ccg-38993
87. Nsc760383
88. S1649
89. Akos015850706
90. Tox21_111455_1
91. Db00315
92. Nc00382
93. Nsc 760383
94. (s)-n,n-dimethyl-2-[5-(2-oxo-1,3-oxazolidin-4-ylmethyl)-1h-indol-3-yl]ethylamine
95. Ncgc00263884-02
96. Ncgc00263884-03
97. Ncgc00263884-08
98. Bz164590
99. Bw-311c90;311c90
100. Sbi-0206762.p001
101. B2261
102. Sw197762-2
103. Z0024
104. 64z178
105. C07218
106. D00415
107. Ab00698244-05
108. Ab00698244_06
109. Q218820
110. Sr-05000001693
111. J-007257
112. Sr-05000001693-1
113. Zolmitriptan, United States Pharmacopeia (usp) Reference Standard
114. (4s)-4-[[3-[2-(dimethylamino)ethyl]-1h-indol-5-yl]methyl-2-oxazolidinone
115. (s)-4-{3-[2-(dimethylamino)ethyl]-1h-indol-5-ylmethyl}oxazolidin-2-one
116. (4s)-4-((3-(2-(dimethylamino)ethyl) -1h-indol-5-yl)methyl)-2-oxazolidinone
117. (s) -n,n-dimethyl-2- [5- (2-oxo-1,3-oxazolidin-4-ylmethyl) -1h -indol-3-yl]ethylamine
118. (s)-4-({3-[2-(dimethylamino)ethyl]-1h-indol-5-yl}methyl)-1,3-oxazolidin-2-one
119. (s)-n,n-dimethyl-2-[5-(2-oxo-1,3-oxazolidin-4-ylmethyl)-1h -indol-3-yl]ethylamine
120. 2-oxazolidinone, 4-[[3-[2-(dimethylamino)ethyl]-1h-indol-5-yl]methyl]-, (4s)-
121. 139264-25-8
122. Zmt
| Molecular Weight | 287.36 g/mol |
|---|---|
| Molecular Formula | C16H21N3O2 |
| XLogP3 | 2.2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 5 |
| Exact Mass | 287.16337692 g/mol |
| Monoisotopic Mass | 287.16337692 g/mol |
| Topological Polar Surface Area | 57.4 Ų |
| Heavy Atom Count | 21 |
| Formal Charge | 0 |
| Complexity | 375 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 6 | |
|---|---|
| Drug Name | Zolmitriptan |
| PubMed Health | Zolmitriptan |
| Drug Classes | Antimigraine |
| Drug Label | ZOMIG (zolmitriptan) Tablets and ZOMIG-ZMT (zolmitriptan) Orally Disintegrating Tablets contain zolmitriptan, which is a selective 5-hydroxytryptamine 1B/1D (5-HT1B/1D) receptor agonist. Zolmitriptan is chemically designated as (S)-4-[[3-[2-(dime... |
| Active Ingredient | Zolmitriptan |
| Dosage Form | Tablet; Tablet, orally disintegrating |
| Route | Oral |
| Strength | 2.5mg; 5mg |
| Market Status | Prescription |
| Company | Mylan Pharms; Apotex; Teva Pharms Usa; Invagen Pharms; Sun Pharma Global; Glenmark Generics; Zydus Pharms Usa |
| 2 of 6 | |
|---|---|
| Drug Name | Zomig |
| PubMed Health | Zolmitriptan (Into the nose) |
| Drug Classes | Antimigraine |
| Drug Label | ZOMIG (zolmitriptan) Tablets and ZOMIG-ZMT (zolmitriptan) Orally Disintegrating Tablets contain zolmitriptan, which is a selective 5-hydroxytryptamine 1B/1D (5-HT1B/1D) receptor agonist. Zolmitriptan is chemically designated as (S)-4-[[3-[2-(dime... |
| Active Ingredient | Zolmitriptan |
| Dosage Form | Tablet; Spray |
| Route | Oral; Nasal |
| Strength | 2.5mg; 2.5mg/spray; 5mg; 5mg/spray |
| Market Status | Prescription |
| Company | Ipr; Astrazeneca |
| 3 of 6 | |
|---|---|
| Drug Name | Zomig-zmt |
| PubMed Health | Zolmitriptan (By mouth) |
| Drug Classes | Antimigraine |
| Drug Label | ZOMIG (zolmitriptan) Tablets and ZOMIG-ZMT (zolmitriptan) Orally Disintegrating Tablets contain zolmitriptan, which is a selective 5-hydroxytryptamine 1B/1D (5-HT1B/1D) receptor agonist. Zolmitriptan is chemically designated as (S)-4-[[3-[2-(dime... |
| Active Ingredient | Zolmitriptan |
| Dosage Form | Tablet, orally disintegrating |
| Route | Oral |
| Strength | 2.5mg; 5mg |
| Market Status | Prescription |
| Company | Astrazeneca |
| 4 of 6 | |
|---|---|
| Drug Name | Zolmitriptan |
| PubMed Health | Zolmitriptan |
| Drug Classes | Antimigraine |
| Drug Label | ZOMIG (zolmitriptan) Tablets and ZOMIG-ZMT (zolmitriptan) Orally Disintegrating Tablets contain zolmitriptan, which is a selective 5-hydroxytryptamine 1B/1D (5-HT1B/1D) receptor agonist. Zolmitriptan is chemically designated as (S)-4-[[3-[2-(dime... |
| Active Ingredient | Zolmitriptan |
| Dosage Form | Tablet; Tablet, orally disintegrating |
| Route | Oral |
| Strength | 2.5mg; 5mg |
| Market Status | Prescription |
| Company | Mylan Pharms; Apotex; Teva Pharms Usa; Invagen Pharms; Sun Pharma Global; Glenmark Generics; Zydus Pharms Usa |
| 5 of 6 | |
|---|---|
| Drug Name | Zomig |
| PubMed Health | Zolmitriptan (Into the nose) |
| Drug Classes | Antimigraine |
| Drug Label | ZOMIG (zolmitriptan) Tablets and ZOMIG-ZMT (zolmitriptan) Orally Disintegrating Tablets contain zolmitriptan, which is a selective 5-hydroxytryptamine 1B/1D (5-HT1B/1D) receptor agonist. Zolmitriptan is chemically designated as (S)-4-[[3-[2-(dime... |
| Active Ingredient | Zolmitriptan |
| Dosage Form | Tablet; Spray |
| Route | Oral; Nasal |
| Strength | 2.5mg; 2.5mg/spray; 5mg; 5mg/spray |
| Market Status | Prescription |
| Company | Ipr; Astrazeneca |
| 6 of 6 | |
|---|---|
| Drug Name | Zomig-zmt |
| PubMed Health | Zolmitriptan (By mouth) |
| Drug Classes | Antimigraine |
| Drug Label | ZOMIG (zolmitriptan) Tablets and ZOMIG-ZMT (zolmitriptan) Orally Disintegrating Tablets contain zolmitriptan, which is a selective 5-hydroxytryptamine 1B/1D (5-HT1B/1D) receptor agonist. Zolmitriptan is chemically designated as (S)-4-[[3-[2-(dime... |
| Active Ingredient | Zolmitriptan |
| Dosage Form | Tablet, orally disintegrating |
| Route | Oral |
| Strength | 2.5mg; 5mg |
| Market Status | Prescription |
| Company | Astrazeneca |
Zolmitriptan is indicated for the acute treatment of migraine with or without auras in patients aged 18 and over.
FDA Label
Zolmitriptan, like other triptans, is a serotonin (5-hydroxytryptamine; 5-HT) receptor agonist, with enhanced specificity for the 5-HT1B and 5-HT1D receptor subtypes. It is through the downstream effects of 5-HT1B/1D activation that triptans are proposed to provide acute relief of migraines. Zolmitriptan is also a vasoconstrictor, leading to possible adverse cardiovascular effects such as myocardial ischemia/infarction, arrhythmias, cerebral and subarachnoid hemorrhage, stroke, gastrointestinal ischemia, and peripheral vasospastic reactions. In addition, chest/throat/neck/jaw pain, tightness, and/or pressure has been reported, along with the possibility of medication overuse headaches and serotonin syndrome. Patients with phenylketonuria should be advised that ZOMIG-ZMT contains phenylalanine.
Serotonin 5-HT1 Receptor Agonists
Endogenous compounds and drugs that specifically stimulate SEROTONIN 5-HT1 RECEPTORS. Included under this heading are agonists for one or more of the specific 5-HT1 receptor subtypes. (See all compounds classified as Serotonin 5-HT1 Receptor Agonists.)
N02CC03
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
N - Nervous system
N02 - Analgesics
N02C - Antimigraine preparations
N02CC - Selective serotonin (5ht1) agonists
N02CC03 - Zolmitriptan
Absorption
Zolmitriptan tablets have a mean absolute oral bioavailability of approximately 40%, with food having no effect on the rate or extent of absorption. The dosing kinetics are linear over a range of 2.5 to 50 mg with 75% of the eventual Cmax being attained within 1 hour of dosing. The median Tmax for the tablet form is 1.5 hours, while for the orally disintegrating tablet form, it is 3 hours. The AUC across studies was in the range of 84.4-173.8 ng/mL*h while the Cmax was between 16 and 25.2 ng/mL. Zolmitriptan administered as a nasal spray is detected in the plasma within 2-5 minutes, compared to 10-15 minutes for the tablet form; the faster kinetics likely reflect fast absorption across the nasal mucosa. The bioavailability compared to the tablet is 102%, and plasma zolmitriptan concentration is maintained for 4-6 hours after intranasal delivery. The active N-desmethyl metabolite of zolmitriptan has a mean plasma concentration that is roughly two-thirds of zolmitriptan, regardless of dosage route or concentration.
Route of Elimination
Zolmitriptan is primarily excreted in urine (approximately 65%) and feces (approximately 30%). Within urine, the most common form is the indole acetic acid metabolite (31%), followed by the N-oxide (7%), and N-desmethyl (4%) metabolites; the majority of zolmitriptan recovered in feces remains unchanged.
Volume of Distribution
Zolmitriptan has a volume of distribution between 7 and 8.4 L/kg.
Clearance
Zolmitriptan has a clearance of 31.5 mL/min/kg for oral tablets and 25.9 mL/min/kg for nasal administration; one-sixth of the clearance is renal.
Zolmitriptan is metabolized in the liver, and studies using cytochrome P450 inhibitors like [cimetidine] suggest that it is likely metabolized by CYP1A2, as well as by monoamine oxidase (MAO). Zolmitriptan metabolism results in three major metabolites: an active N-desmethyl metabolite (183C91) as well as inactive N-oxide (1652W92) and indole acetic acid (2161W92) metabolites.
Zolmitriptan has known human metabolites that include N-Desmethylzolmitriptan and Zolmitriptan N-oxide.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
Zolmitriptan has a mean elimination half-life of approximately three hours, while its active N-desmethyl metabolite has a slightly longer (approximately 3.5 hours) half-life.
Migraines are complex physiological events characterized by unilateral throbbing headaches combined with photophobia and other aversions to sensory input. Migraine attacks are generally divided into phases: the premonitory phase, which typically involves irritability, fatigue, yawning, and stiff neck; the headache phase, which lasts for between four and 72 hours; and the postdrome phase, which lasts for up to a day following resolution of pain and whose symptoms are similar to those of the premonitory phase. In addition, neurological deficits, collectively termed migraine aura, may precede the headache phase. The underlying pathophysiology of migraines is a matter of active research but involves both neurological and vascular components. The head pain associated with migraine is thought to be a consequence of activation of the nociceptive nerves comprising the trigeminocervical complex (TCC). Terminals of nociceptive nerves that innervate the dura matter release vasoactive peptides, such as calcitonin gene-related peptide (CGRP), resulting in cranial vasodilation. Finally, when present, migraine aura appears to correlate with a transient wave(s) of cortical depolarization, termed cortical spreading depression (CSD). Triptans, including zolmitriptan, are proposed to act in three ways. The main mechanism is through modulation of nociceptive nerve signalling in the central nervous system through 5-HT1B/1D receptors throughout the TCC and associated areas of the brain. In addition, triptans can enhance vasoconstriction, both through direct 5-HT1B-mediated dilation of cranial blood vessels, as well as through 5-HT1D-mediated suppression of CGRP release. Although triptans are classically described solely in terms of their effects on 5-HT1B/1D receptors, they also act as 5-HT1F agonists as well. This 5-HT subtype is also found throughout the TCC, but is not present appreciably in cerebral vasculature; the significance of triptan-mediated 5-HT1F activation is currently not well described. Additionally, CSD that initiates in the ipsilateral parietal region may exert its effects in a manner that relies on 5-HT1B/1D receptor activation, suggesting that triptans may have some effect on CSD-mediated symptoms.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
22
PharmaCompass offers a list of Zolmitriptan API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Zolmitriptan manufacturer or Zolmitriptan supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Zolmitriptan manufacturer or Zolmitriptan supplier.
PharmaCompass also assists you with knowing the Zolmitriptan API Price utilized in the formulation of products. Zolmitriptan API Price is not always fixed or binding as the Zolmitriptan Price is obtained through a variety of data sources. The Zolmitriptan Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Zolmitriptan manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Zolmitriptan, including repackagers and relabelers. The FDA regulates Zolmitriptan manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Zolmitriptan API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Zolmitriptan manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Zolmitriptan supplier is an individual or a company that provides Zolmitriptan active pharmaceutical ingredient (API) or Zolmitriptan finished formulations upon request. The Zolmitriptan suppliers may include Zolmitriptan API manufacturers, exporters, distributors and traders.
click here to find a list of Zolmitriptan suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Zolmitriptan DMF (Drug Master File) is a document detailing the whole manufacturing process of Zolmitriptan active pharmaceutical ingredient (API) in detail. Different forms of Zolmitriptan DMFs exist exist since differing nations have different regulations, such as Zolmitriptan USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Zolmitriptan DMF submitted to regulatory agencies in the US is known as a USDMF. Zolmitriptan USDMF includes data on Zolmitriptan's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Zolmitriptan USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Zolmitriptan suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Zolmitriptan Drug Master File in Japan (Zolmitriptan JDMF) empowers Zolmitriptan API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Zolmitriptan JDMF during the approval evaluation for pharmaceutical products. At the time of Zolmitriptan JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Zolmitriptan suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Zolmitriptan Drug Master File in Korea (Zolmitriptan KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Zolmitriptan. The MFDS reviews the Zolmitriptan KDMF as part of the drug registration process and uses the information provided in the Zolmitriptan KDMF to evaluate the safety and efficacy of the drug.
After submitting a Zolmitriptan KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Zolmitriptan API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Zolmitriptan suppliers with KDMF on PharmaCompass.
A Zolmitriptan CEP of the European Pharmacopoeia monograph is often referred to as a Zolmitriptan Certificate of Suitability (COS). The purpose of a Zolmitriptan CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Zolmitriptan EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Zolmitriptan to their clients by showing that a Zolmitriptan CEP has been issued for it. The manufacturer submits a Zolmitriptan CEP (COS) as part of the market authorization procedure, and it takes on the role of a Zolmitriptan CEP holder for the record. Additionally, the data presented in the Zolmitriptan CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Zolmitriptan DMF.
A Zolmitriptan CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Zolmitriptan CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Zolmitriptan suppliers with CEP (COS) on PharmaCompass.
A Zolmitriptan written confirmation (Zolmitriptan WC) is an official document issued by a regulatory agency to a Zolmitriptan manufacturer, verifying that the manufacturing facility of a Zolmitriptan active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Zolmitriptan APIs or Zolmitriptan finished pharmaceutical products to another nation, regulatory agencies frequently require a Zolmitriptan WC (written confirmation) as part of the regulatory process.
click here to find a list of Zolmitriptan suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Zolmitriptan as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Zolmitriptan API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Zolmitriptan as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Zolmitriptan and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Zolmitriptan NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Zolmitriptan suppliers with NDC on PharmaCompass.
Zolmitriptan Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Zolmitriptan GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Zolmitriptan GMP manufacturer or Zolmitriptan GMP API supplier for your needs.
A Zolmitriptan CoA (Certificate of Analysis) is a formal document that attests to Zolmitriptan's compliance with Zolmitriptan specifications and serves as a tool for batch-level quality control.
Zolmitriptan CoA mostly includes findings from lab analyses of a specific batch. For each Zolmitriptan CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Zolmitriptan may be tested according to a variety of international standards, such as European Pharmacopoeia (Zolmitriptan EP), Zolmitriptan JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Zolmitriptan USP).
