Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
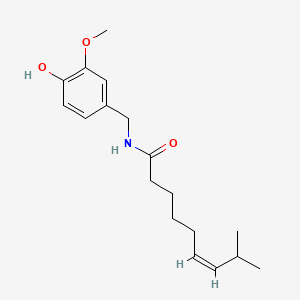

1. Civamide
1. 25775-90-0
2. Civamide
3. Cis-capsaicin
4. (z)-capsaicin
5. (z)-n-(4-hydroxy-3-methoxybenzyl)-8-methylnon-6-enamide
6. Civanex
7. (z)-n-[(4-hydroxy-3-methoxyphenyl)methyl]-8-methylnon-6-enamide
8. (6z)-n-[(4-hydroxy-3-methoxyphenyl)methyl]-8-methylnon-6-enamide
9. 15ox67p384
10. Brn 4261852
11. Zucapsaicin [usan:inn]
12. Smr000449294
13. (z)-8-methyl-n-vanillyl-6-nonenamide
14. Sr-01000597644
15. Zuacta
16. Tnp00277
17. Unii-15ox67p384
18. 6-nonenamide, N-[(4-hydroxy-3-methoxyphenyl)methyl]-8-methyl-, (6z)-
19. Cas-404-86-4
20. Tocris-0462
21. Tocris-0463
22. Cpd000449294
23. 6-noneamide, 8-methyl-n-vanillyl-, (z)-
24. N-((4-hydroxy-3-methoxyphenyl)methyl)-8-methyl-6-nonenamide (z)-
25. Zucapsaicin [inn]
26. Zucapsaicin (usan/inn)
27. Zucapsaicin [usan]
28. Schembl41210
29. Zucapsaicin [who-dd]
30. Mls000758306
31. Mls001423959
32. Chembl313971
33. Gtpl11576
34. Chebi:135952
35. Dtxsid101014453
36. Hms2051o07
37. Hms2235o20
38. Hy-b1583
39. Zinc4468952
40. 6-nonenamide, N-((4-hydroxy-3-methoxyphenyl)methyl)-8-methyl-, (z)-
41. Bdbm50519720
42. Mfcd00209942
43. S6409
44. Akos015851907
45. Ccg-100774
46. Db09120
47. Nc00024
48. Ng-0020
49. Ncgc00016441-01
50. Ncgc00016441-02
51. Ncgc00016441-03
52. Ncgc00017337-01
53. Ncgc00024601-01
54. Ncgc00024601-02
55. Ac-31789
56. Cs-0013480
57. D06388
58. 775c900
59. Q8074969
60. Sr-01000597644-1
61. Sr-01000597644-2
| Molecular Weight | 305.4 g/mol |
|---|---|
| Molecular Formula | C18H27NO3 |
| XLogP3 | 3.6 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 9 |
| Exact Mass | 305.19909372 g/mol |
| Monoisotopic Mass | 305.19909372 g/mol |
| Topological Polar Surface Area | 58.6 Ų |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 341 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Indicated to be used in conjunction with oral COX-2 inhibitors or NSAIDs for the relief of severe pain in adult patients with osteoarthritis of the knee, not controlled with oral COX-2 inhibitors or NSAIDs alone, for a duration of no more than three months.
Zucapsaicin mediates an antinociceptive action via acting as an agonist at TRPV1. TRPV1 play an important physiological role of transducing chemical, mechanical and thermal stimuli as well as pain transduction, and participate in pain modulation and perception. They are mainly distributed in C sensory nerve fibers as well as A fibers to transmit sensory information involving inflammatory and neuropathic pain, and activation of these channels releasesomatostatin, calcitonin gene-related peptide (CGRP) and other neuropeptides (neurokinin A, kassinin), leading to neurogenic inflammation. Zucapsaicin is also reported to affect the peptidergic afferent neurons via a desensitization mechanism to decrease the levels of dorsal root ganglia and sciatic calcitonin gene-related peptide (CGRP) and substance P (SP).
M - Musculo-skeletal system
M02 - Topical products for joint and muscular pain
M02A - Topical products for joint and muscular pain
M02AB - Capsaicin and similar agents
M02AB02 - Zucapsaicin
Absorption
Zucapsaicin displays low systemic absorption and localizes at the area of application. In animal studies, systemic absorption is 0.075%.
Route of Elimination
In rat studies, zucapsaicin and its metabolites are slowly excreted into urine and feces (up to 2/3), with minimal elimination via exhalation following dermal administration.
In vitro studies demonstrates weak to moderate inhibitiory effects on various cytochrome P450 enzymes, although not clinically significant due to low systemic absorption.
In rats, the elimination half life of zucapsaicin and its metabolites is approximately 7 to 11 hours.
Zucapsaicin excites and desensitizes C-fibers via agonist at TRPV1 on nociceptive neurons. It binds to intracellular sites and initially stimulates the channels, causing burning sensation. Activation of TRPV1 results in calcium influx and sodium, which leads to cell depolarization. Hypersensitization by zucapsaicin is then followed by reduced sensitivity and persistent desensitization (tachyphylaxis) of the channels via various pathways. Densentiziation is thought to be dependent on intraceullar levels of calcium. Decreased TRPV1 channel action and release of inflammatory neuropeptides induces an analgesic effect, and pain relief. Zucapsaicin activates calcineurin and calcium-dependent protein kinase C isoforms which results in phosphorylation of TRPV1. Phosphorylation of TRPV1 enhances reponsivity to zucapsaicin by potentiating capsaicin- or proton-evoked responses and reducing the temperature threshold for TRPV1 activation. Studies suggest that zucapsaicin is involved in activation of phospholipase C with the subsequent phosphatidylinositol 4,5-biphosphate (PIP2) hydrolysis, which results in TRPV1 inactivation. Tachyphylaxis or persistent desensitization is reversible, and involves the downregulation of proalgesic substances (such as SP) and upregulation of analgesic peptides.
ABOUT THIS PAGE
26
PharmaCompass offers a list of Zucapsaicin API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Zucapsaicin manufacturer or Zucapsaicin supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Zucapsaicin manufacturer or Zucapsaicin supplier.
PharmaCompass also assists you with knowing the Zucapsaicin API Price utilized in the formulation of products. Zucapsaicin API Price is not always fixed or binding as the Zucapsaicin Price is obtained through a variety of data sources. The Zucapsaicin Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Zucapsaicin manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Zucapsaicin, including repackagers and relabelers. The FDA regulates Zucapsaicin manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Zucapsaicin API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Zucapsaicin supplier is an individual or a company that provides Zucapsaicin active pharmaceutical ingredient (API) or Zucapsaicin finished formulations upon request. The Zucapsaicin suppliers may include Zucapsaicin API manufacturers, exporters, distributors and traders.
Zucapsaicin Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Zucapsaicin GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Zucapsaicin GMP manufacturer or Zucapsaicin GMP API supplier for your needs.
A Zucapsaicin CoA (Certificate of Analysis) is a formal document that attests to Zucapsaicin's compliance with Zucapsaicin specifications and serves as a tool for batch-level quality control.
Zucapsaicin CoA mostly includes findings from lab analyses of a specific batch. For each Zucapsaicin CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Zucapsaicin may be tested according to a variety of international standards, such as European Pharmacopoeia (Zucapsaicin EP), Zucapsaicin JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Zucapsaicin USP).
