30 Sep 2024
// BSEINDIA
11 Apr 2025
// REUTERS
11 Apr 2025
// REUTERS
Latest Content by PharmaCompass
 KEY PRODUCTS
KEY PRODUCTS
With Virupaksha, you get a quality product with on-time delivery.
About
CPhI WW FrankfurtCPhI WW Frankfurt
Industry Trade Show
Booth #11.0F34
28-30 October, 2025
German Wound CongressGerman Wound Congress
Industry Trade Show
Not Confirmed
07 April-09 May, 2025
Industry Trade Show
Not Confirmed
08 April-11 May, 2025
CONTACT DETAILS




Events
Webinars & Exhibitions
CPhI WW FrankfurtCPhI WW Frankfurt
Industry Trade Show
Booth #11.0F34
28-30 October, 2025
German Wound CongressGerman Wound Congress
Industry Trade Show
Not Confirmed
07 April-09 May, 2025
Industry Trade Show
Not Confirmed
08 April-11 May, 2025
CORPORATE CONTENT #SupplierSpotlight
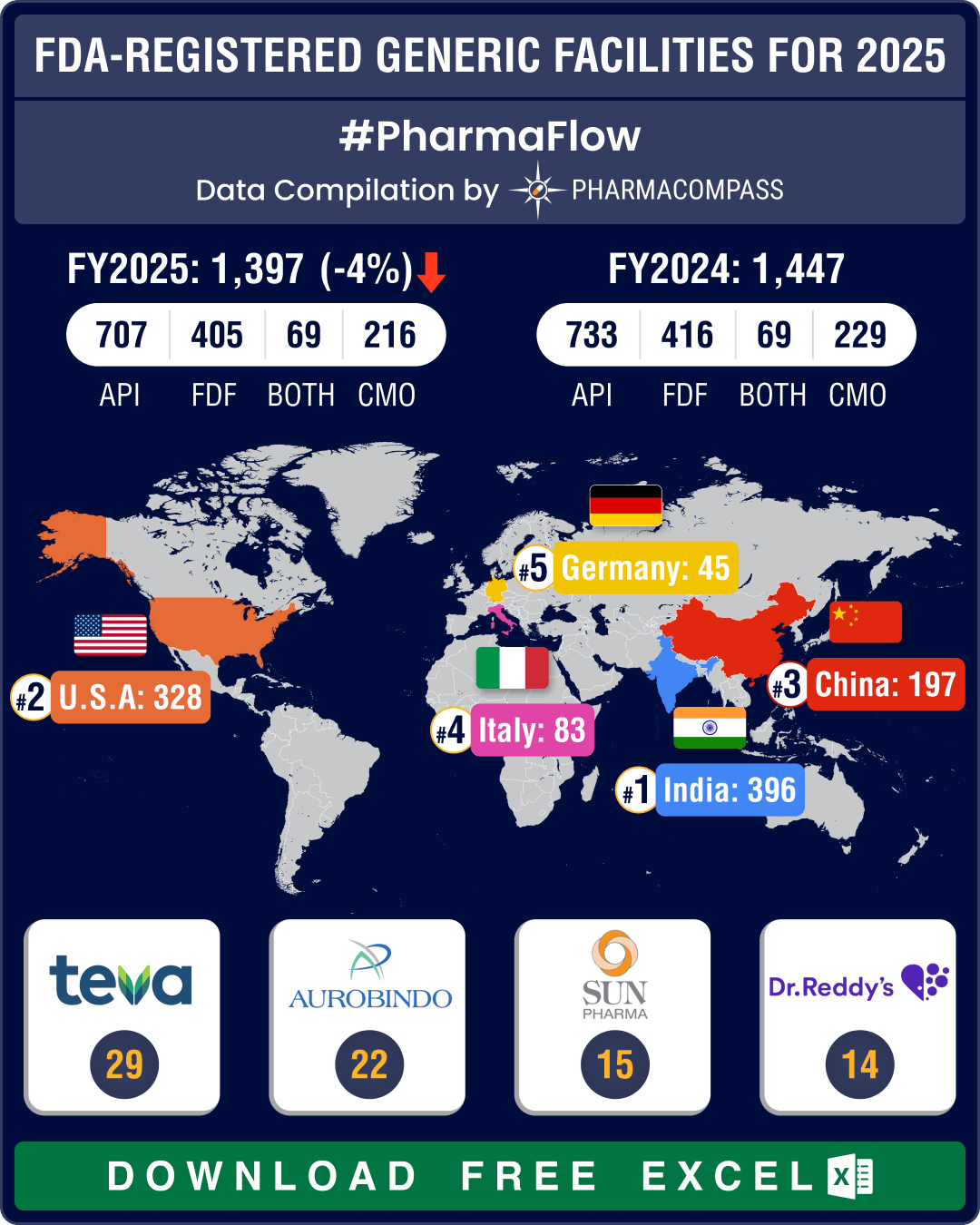
https://www.pharmacompass.com/radio-compass-blog/chinese-fda-registered-generic-facilities-gain-steam-india-maintains-lead-with-396-facilities

30 Sep 2024
// BSEINDIA
https://www.bseindia.com/xml-data/corpfiling/AttachHis/1C45CF51-65B3-4DE7-B64C-CE5E996BBAD7-191527.pdf
Inspections and registrations
ABOUT THIS PAGE
Virupaksha Organics is a supplier offers 39 products (APIs, Excipients or Intermediates).
Find a price of Fexofenadine Hydrochloride bulk with DMF, CEP, JDMF, WC offered by Virupaksha Organics
Find a price of Fluconazole bulk with DMF, CEP, JDMF, WC offered by Virupaksha Organics
Find a price of Escitalopram Oxalate bulk with DMF, CEP, WC offered by Virupaksha Organics
Find a price of Tramadol Hydrochloride bulk with DMF, CEP, WC offered by Virupaksha Organics
Find a price of Bilastine bulk with DMF, JDMF offered by Virupaksha Organics
Find a price of Citalopram Hydrobromide bulk with DMF, CEP offered by Virupaksha Organics
Find a price of Crisaborole bulk with DMF, WC offered by Virupaksha Organics
Find a price of Fexofenadine Hydrochloride bulk with JDMF, WC offered by Virupaksha Organics
Find a price of Tapentadol bulk with CEP, WC offered by Virupaksha Organics
Find a price of Cinnarizine bulk with WC offered by Virupaksha Organics
Find a price of Dextromethorphan Hydrobromide bulk with DMF offered by Virupaksha Organics
Find a price of Efinaconazole bulk with DMF offered by Virupaksha Organics
Find a price of Fexofenadine Hydrochloride bulk with JDMF offered by Virupaksha Organics
Find a price of Fluconazole bulk with DMF offered by Virupaksha Organics
Find a price of Sofosbuvir bulk with WC offered by Virupaksha Organics
Find a price of Tramadol Hydrochloride bulk with WC offered by Virupaksha Organics
Find a price of 1-(4-(BENZYLOXY)-3-(HYDROXYMETHYL)PHENYL)-2-(TERT-BUTYLAMINO) ETHAN-1-OL bulk with DMF offered by Virupaksha Organics
Find a price of Amisulpride bulk offered by Virupaksha Organics
Find a price of Bempedoic Acid bulk offered by Virupaksha Organics
Find a price of Capsaicin bulk offered by Virupaksha Organics
Find a price of Chlorthalidone bulk offered by Virupaksha Organics
Find a price of Dapagliflozin bulk offered by Virupaksha Organics
Find a price of Dapagliflozin Propanediol Monohydrate bulk offered by Virupaksha Organics
Find a price of Efinaconazole bulk offered by Virupaksha Organics
Find a price of Fluconazole bulk offered by Virupaksha Organics
Find a price of Flurbiprofen bulk offered by Virupaksha Organics
Find a price of Isavuconazonium Sulfate bulk offered by Virupaksha Organics
Find a price of Linagliptin bulk offered by Virupaksha Organics
Find a price of Luliconazole bulk offered by Virupaksha Organics
Find a price of Omeprazole bulk offered by Virupaksha Organics
Find a price of Sacubitril-Valsartan bulk offered by Virupaksha Organics
Find a price of Salbutamol Sulphate bulk offered by Virupaksha Organics
Find a price of Siponimod bulk offered by Virupaksha Organics
Find a price of Tafamidis bulk offered by Virupaksha Organics
Find a price of Tafamidis Meglumine bulk offered by Virupaksha Organics
Find a price of Tavaborole bulk offered by Virupaksha Organics
Find a price of Tramadol Hydrochloride bulk offered by Virupaksha Organics
Find a price of Vonoprazan Fumarate bulk offered by Virupaksha Organics
Find a price of DEXTROMETHORPHAN HYDRIODIDE bulk offered by Virupaksha Organics