
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
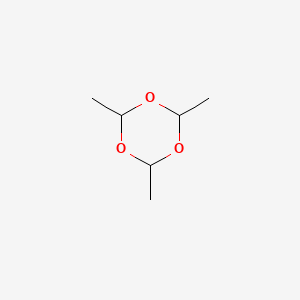

1. 2,4,6-trimethyl-1,3,5-trioxane
2. 123-63-7
3. Paracetaldehyde
4. Acetaldehyde Trimer
5. Paral
6. Paraldehyd
7. Paraacetaldehyde
8. Elaldehyde
9. S-trimethyltrioxymethylene
10. Paraldeide
11. Triacetaldehyde
12. 2,4,6-trimethyl-s-trioxane
13. 1,3,5-trioxane, 2,4,6-trimethyl-
14. 1,3,5-trimethyl-2,4,6-trioxane
15. Acetaldehyde, Trimer
16. P-acetaldehyde
17. Pcho
18. Nsc 9799
19. 2,4,6-trimethyl-1,3,5-trioxacyclohexane
20. Rcra Waste Number U182
21. Paraldehyde Enema (bpc 1973)
22. Paraldehyde Draught (bpc 1973)
23. Trimethyl Trioxane
24. 2,4,6-trimethyl-1,3,5-trioxaan
25. 2,4,6-trimetil-1,3,5-triossano
26. Paraldehyde (usp)
27. Paraldehyde [usp]
28. Cis-2,4,6-trimethyl-1,3,5-trioxane
29. Paral (tn)
30. S-trioxane, 2,4,6-trimethyl-
31. Nsc-9799
32. 2,4,6-trimethyl-1,3,5-trioxan
33. S6m3ybg8qa
34. Fema No. 4010
35. Chebi:27909
36. 1499-02-1
37. Ncgc00159367-02
38. Ncgc00159367-03
39. Ncgc00159367-04
40. Dsstox_cid_3419
41. Dsstox_rid_77020
42. Dsstox_gsid_23419
43. Paraldehyd [german]
44. Paraldehyde [usan]
45. Paraldeide [italian]
46. Triacetaldehyde [french]
47. Cas-123-63-7
48. Hsdb 3375
49. Einecs 204-639-8
50. Unii-s6m3ybg8qa
51. Un1264
52. Rcra Waste No. U182
53. Brn 0080142
54. Paraldehyde Civ
55. Ai3-03115
56. 2,4,6-trimethyl-1,3,5-trioxaan [dutch]
57. Dea No. 2585
58. 2,4,6-trimetil-1,3,5-triossano [italian]
59. Aldehyde (para)
60. Paraldehyd(german)
61. Mfcd00036208
62. S-trimethyltrioxymethane
63. Paraldehyde, >=97%
64. Paraldehyde [un1264] [flammable Liquid]
65. Paraldehyde [mi]
66. Paraldehyde [fhfi]
67. Paraldehyde [hsdb]
68. 2,6-trimethyl-s-trioxane
69. Ec 204-639-8
70. Paraldehyde [vandf]
71. Paraldehyde [mart.]
72. S-trioxane,4,6-trimethyl-
73. Schembl22870
74. Paraldehyde [who-dd]
75. 5-19-09-00112 (beilstein Handbook Reference)
76. Zinc1886
77. Chembl1410743
78. Dtxsid9023419
79. Schembl20072682
80. Nsc9799
81. Paraldehyde Civ [usp-rs]
82. 1,5-trimethyl-2,4,6-trioxane
83. 2,6-trimethyl-1,3,5-trioxaan
84. 2,6-trimethyl-1,3,5-trioxane
85. 2,6-trimetil-1,3,5-triossano
86. Dtxsid301271154
87. Paraldehyde [ep Monograph]
88. Paraldehyde, >=97.0% (gc)
89. Paraldehyde [usp Monograph]
90. Tox21_111607
91. Tox21_111608
92. Tox21_301299
93. 1,5-trioxane, 2,4,6-trimethyl-
94. Bbl011471
95. Stl146583
96. Akos000120195
97. Tox21_111607_1
98. Db09117
99. Un 1264
100. Wln: T6o Co Eotj B1 D1 F1
101. 2,6-trimethyl-1,3,5-trioxacyclohexane
102. Ncgc00257534-01
103. Vs-02956
104. Ft-0650173
105. P0019
106. Paraldehyde [un1264] [flammable Liquid]
107. Paraldehyde, Lonza Quality, >=99.5% (gc)
108. C07834
109. D00705
110. A805128
111. Q424342
112. J-523868
113. F1908-0170
114. (2alpha,4alpha,6alpha)-2,4,6-trimethyl-1,3,5-trioxane
115. Paraldehyde, United States Pharmacopeia (usp) Reference Standard
116. 26893-98-1
| Molecular Weight | 132.16 g/mol |
|---|---|
| Molecular Formula | C6H12O3 |
| XLogP3 | 0.7 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Exact Mass | 132.078644241 g/mol |
| Monoisotopic Mass | 132.078644241 g/mol |
| Topological Polar Surface Area | 27.7 Ų |
| Heavy Atom Count | 9 |
| Formal Charge | 0 |
| Complexity | 63.3 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anticonvulsants; Sedatives, Nonbarbiturate
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Paraldehyde has been used chiefly for the treatment of abstinence phenomena and other psychiatric sates characterized by excitement, and for the emergency treatment of convulsive episodes. Its most persisting use has been in the treatment of delirium tremens.
Gilman, A.G., T.W. Rall, A.S. Nies and P. Taylor (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 8th ed. New York, NY. Pergamon Press, 1990., p. 368
PARALDEHYDE IS EFFECTIVE IN STATUS EPILEPTICUS, BUT SHOULD BE RESERVED FOR PATIENTS WHO DO NOT RESPOND TO PHENOBARBITAL. ... DRUG IS ALSO FREQUENTLY USED IN DELIRIUM TREMENS, & IN PATIENTS UNDERGOING WITHDRAWAL THERAPY FOR ALCOHOLISM.
Osol, A. (ed.). Remington's Pharmaceutical Sciences. 16th ed. Easton, Pennsylvania: Mack Publishing Co., 1980., p. 1019
VET: HYPNOTIC. LIMITED TO INDUCE ... /CNS DEPRESSION/ AS PREANESTHETIC IN SOME ANIMALS & RARELY AS ANESTHETIC ITSELF.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 413
For more Therapeutic Uses (Complete) data for PARALDEHYDE (10 total), please visit the HSDB record page.
IN AT LEAST 8 CASES OF POISONING INVOLVING 4 FATALITIES, USE OF DETERIORATED PARALDEHYDE HAS BEEN IMPLICATED.
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 131
PREPARATIONS THAT HAVE BROWNISH COLOR OR SHARP PENETRATING ODOR OF ACETIC ACID SHOULD NOT BE USED.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 93. Bethesda, MD: American Society of Hospital Pharmacists, Inc., 1993 (Plus Supplements, 1993)., p. 1452
ALL CONTAINERS OF PARALDEHYDE, INCLUDING THOSE DISPENSED BY THE PHARMACIST, SHOULD CARRY STATEMENT DIRECTING USER TO DISCARD THE UNUSED CONTENTS OF ANY CONTAINER WHICH HAS BEEN OPENED FOR MORE THAN 24 HR.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 93. Bethesda, MD: American Society of Hospital Pharmacists, Inc., 1993 (Plus Supplements, 1993)., p. 1452
IN NO CASE SHOULD PARALDEHYDE BE TAKEN FROM OLD, PARTIALLY EMPTY CONTAINERS, SINCE AN APPRECIABLE PROPORTION OF THE DRUG MAY HAVE BEEN OXIDIZED TO ACETIC ACID & OTHER DECOMPOSITION PRODUCTS. BECAUSE PARALDEHYDE REACTS RAPIDLY WITH CERTAIN PLASTICS, IT SHOULD BE MEASURED WITH GLASS SYRINGES.
Gilman, A.G., L.S.Goodman, and A. Gilman. (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 7th ed. New York: Macmillan Publishing Co., Inc., 1985., p. 367
For more Drug Warnings (Complete) data for PARALDEHYDE (12 total), please visit the HSDB record page.
Paraldehyde was used historically as a sedative and hypnotic. It has been used in the treatment of seizures as an anticonvulsant.
Paraldehyde blocks neuromuscular transmission.
Hypnotics and Sedatives
Drugs used to induce drowsiness or sleep or to reduce psychological excitement or anxiety. (See all compounds classified as Hypnotics and Sedatives.)
Anticonvulsants
Drugs used to prevent SEIZURES or reduce their severity. (See all compounds classified as Anticonvulsants.)
N - Nervous system
N05 - Psycholeptics
N05C - Hypnotics and sedatives
N05CC - Aldehydes and derivatives
N05CC05 - Paraldehyde
Absorption
93% of orally administered paraldehyde is absorbed from the gastrointestinal tract.
Route of Elimination
70-80% is metabolized to carbon dioxide and subsequently exhaled. 11-28% is exhaled as the parent compound. 0.1-2.5% is excreted in the urine as the parent compound.
Paraldehyde is rapidly absorbed from ... IM injection sites. Maximum serum concn, which may range from 34 to 150 ug/ml, are reached within 20-60 minutes following oral admin of 10 ml ... or IM admin of 0.25 ml/kg. ... Although tissue distribution ... has not been extensively studied, it is known that the concn ... in CSF is about 25 to 30% lower than that in the blood. Maximum paraldehyde concn are reached in the CSF 30-60 minutes after oral or IM admin ...
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 93. Bethesda, MD: American Society of Hospital Pharmacists, Inc., 1993 (Plus Supplements, 1993)., p. 1452
Oral paraldehyde is rapidly absorbed and widely distributed; the drug readily crosses the placental barrier. With hypnotic doses, 70 to 80% is metabolized in the liver, most of the remainder is exhaled, and a small amount is excreted in urine.
Gilman, A.G., T.W. Rall, A.S. Nies and P. Taylor (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 8th ed. New York, NY. Pergamon Press, 1990., p. 368
... PARALDEHYDE EQUILIBRATES RAPIDLY BETWEEN MATERNAL & FETAL BLOODS.
The Chemical Society. Foreign Compound Metabolism in Mammals Volume 3. London: The Chemical Society, 1975., p. 634
FETAL/MATERNAL CONCN RATIO IS 0.94. /FROM TABLE/
LaDu, B.N., H.G. Mandel, and E.L. Way. Fundamentals of Drug Metabolism and Disposition. Baltimore: Williams and Wilkins, 1971., p. 100
For more Absorption, Distribution and Excretion (Complete) data for PARALDEHYDE (9 total), please visit the HSDB record page.
Paraldehyde is believed to undergo depolymerization to acetaldehyde followed by oxidation by aldehyde dehydrogenase. It is thought to ultimately be metabolized to carbon dioxide and water.
It is believed that paraldehyde is depolymerized to acetaldehyde in the liver & then oxidized by aldehyde dehydrogenase to acetic acid, which is ultimately metabolized to carbon dioxide & water.
Gilman, A.G., T.W. Rall, A.S. Nies and P. Taylor (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 8th ed. New York, NY. Pergamon Press, 1990., p. 368
The mean half life is 7.5 hours in a range if 3.5-9.5 hours.
The biological half-life of paraldehyde /in humans/ has been reported to be 3.5 to 9.5 hours with an average of about 7.5 hours.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 93. Bethesda, MD: American Society of Hospital Pharmacists, Inc., 1993 (Plus Supplements, 1993)., p. 1452
Paraldehyde is believed to reduce the release of acetylcholine in response to neuronal depolarization. The exact mechanism of this effect is unknown.
BELIEVED TO DEPRESS MANY LEVELS OF CNS INCLUDING ASCENDING RETICULAR ACTIVATING SYSTEM TO CAUSE IMBALANCE BETWEEN INHIBITORY & FACILITATORY MECHANISMS.
American Society of Hospital Pharmacists. Data supplied on contract from American Hospital Formulary Service and other current ASHP sources., p. 1975
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
98
PharmaCompass offers a list of Paraldehyde API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Paraldehyde manufacturer or Paraldehyde supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Paraldehyde manufacturer or Paraldehyde supplier.
PharmaCompass also assists you with knowing the Paraldehyde API Price utilized in the formulation of products. Paraldehyde API Price is not always fixed or binding as the Paraldehyde Price is obtained through a variety of data sources. The Paraldehyde Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Paraldehyde manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Paraldehyde, including repackagers and relabelers. The FDA regulates Paraldehyde manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Paraldehyde API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Paraldehyde supplier is an individual or a company that provides Paraldehyde active pharmaceutical ingredient (API) or Paraldehyde finished formulations upon request. The Paraldehyde suppliers may include Paraldehyde API manufacturers, exporters, distributors and traders.
Paraldehyde Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Paraldehyde GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Paraldehyde GMP manufacturer or Paraldehyde GMP API supplier for your needs.
A Paraldehyde CoA (Certificate of Analysis) is a formal document that attests to Paraldehyde's compliance with Paraldehyde specifications and serves as a tool for batch-level quality control.
Paraldehyde CoA mostly includes findings from lab analyses of a specific batch. For each Paraldehyde CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Paraldehyde may be tested according to a variety of international standards, such as European Pharmacopoeia (Paraldehyde EP), Paraldehyde JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Paraldehyde USP).
