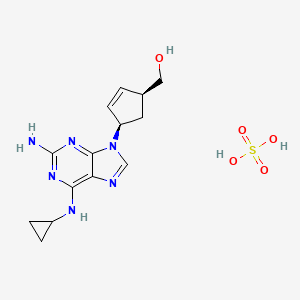

1. ((1r,4r)-4-(2-amino-6-(cyclopropylamino)-9h-purin-9-yl)-cyclopent-2-enyl)methanol
2. ((1r,4s)-4-(2-amino-6-(cyclopropylamino)-9h-purin-9-yl)cyclopent-2-enyl)methanol
3. (+)-abacavir
4. (+)-abacavir Sulfate
5. (+-)-abacavir Sulfate
6. (-)-cis-4-(2-amino-6-(cyclopropylamino)-9h-purin-9-yl)-2-cyclopentene-1-methanol
7. (1r,4r)-abacavir
8. (1r,4s)-abacavir Sulfate
9. (1s,4r)-4-(2-amino-6-(cyclopropylamino)-9h-purin-9-yl)-2-cyclopentene-1-methanol
10. (1s,4r)-4-(2-amino-6-(cyclopropylamino)-9h-purin-9-yl)-2-cyclopentene-1-methanol Succinate (1:1) (salt)
11. 1592u89
12. 2-cyclopentene-1-methanol, 4-(2-amino-6-(cyclopropylamino)-9h-purin-9-yl)-, (1r,4s)-, Sulfate (2:1)
13. 2-cyclopentene-1-methanol, 4-(2-amino-6-(cyclopropylamino)-9h-purin-9-yl)-, (1s,4r)-, Rel-, Sulfate (2:1)
14. 2-cyclopentene-1-methanol, 4-(2-amino-6-(cyclopropylamino)-9h-purin-9-yl)-, Hydrochloride, Hydrate (1:1:1), (1s,4r)-
15. Abacavir
16. Abacavir Enantiomer
17. Abacavir Hydrochloride Monohydrate
18. Abacavir Succinate
19. Abacavir Sulfate
20. Abacavir Sulfate Racemic
21. Abacavir Sulfate, (+)-
22. Abacavir Sulfate, (+-)-
23. Abacavir Sulfate, (1r,4s)-
24. Abacavir Sulphate
25. Abacavir, (+)-
26. Abacavir, (1r,4s)-
27. Abacavir, Trans-
28. Abacavir, Trans-, (+-)-
29. Abamune
30. Avacavir
31. Drg 0257
32. Drg-0257
33. Drg0257
34. Trans-abacavir
35. Trans-abacavir R,r-
1. Abacavir Sulfate
2. Abacavir (sulfate)
3. 216699-07-9
4. Abacavir (monosulfate)
5. 4-[2-amino-6-(cyclopropylamino)-9h-purin-9-yl]-(1s,4r)-2-cyclopentene-1-methanolsulfate
6. (1s,4r)-4-[2-amino-6-(cyclopropylamino)-9h-purin-9-yl]-2-cyclopentene-1-methanol, Monosulfate
7. Schembl40818
8. Chembl1200619
9. Hy-17423b
10. Akos015994684
11. Ks-1221
12. Cs-0083626
| Molecular Weight | 384.41 g/mol |
|---|---|
| Molecular Formula | C14H20N6O5S |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 4 |
| Exact Mass | 384.12158893 g/mol |
| Monoisotopic Mass | 384.12158893 g/mol |
| Topological Polar Surface Area | 185 Ų |
| Heavy Atom Count | 26 |
| Formal Charge | 0 |
| Complexity | 496 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Abacavir sulfate |
| Drug Label | Abacavir sulfate is a synthetic carbocyclic nucleoside analogue with inhibitory activity against HIV-1. The chemical name of abacavir sulfate is (1S,cis)-4-[2-amino-6-(cyclopropylamino)-9H-purin-9-yl]-2-cyclopentene-1-methanol sulfate (salt) (2:1). A... |
| Active Ingredient | Abacavir sulfate |
| Dosage Form | Solution; Tablet |
| Route | oral; Oral |
| Strength | 300mg; 60mg; 60 mg; 20mg; eq 300mg base |
| Market Status | Tentative Approval; Prescription |
| Company | Hetero Labs Ltd Iii; Mylan Pharms; Aurobindo Pharma; Cipla; Matrix Labs; Apotex; Strides Arcolab |
| 2 of 2 | |
|---|---|
| Drug Name | Abacavir sulfate |
| Drug Label | Abacavir sulfate is a synthetic carbocyclic nucleoside analogue with inhibitory activity against HIV-1. The chemical name of abacavir sulfate is (1S,cis)-4-[2-amino-6-(cyclopropylamino)-9H-purin-9-yl]-2-cyclopentene-1-methanol sulfate (salt) (2:1). A... |
| Active Ingredient | Abacavir sulfate |
| Dosage Form | Solution; Tablet |
| Route | oral; Oral |
| Strength | 300mg; 60mg; 60 mg; 20mg; eq 300mg base |
| Market Status | Tentative Approval; Prescription |
| Company | Hetero Labs Ltd Iii; Mylan Pharms; Aurobindo Pharma; Cipla; Matrix Labs; Apotex; Strides Arcolab |
Abacavir is indicated, in combination with other agents, for treatment of HIV-1 infection. /Included in US product labeling/
MICROMEDEX Thomson Health Care. USPDI - Drug Information for the Health Care Professional. 23rd ed. Volume 1. MICROMEDEX Thomson Health Care, Greenwood Village, CO. 2003. Content Reviewed and Approved by the U.S. Pharmacopeial Convention, Inc., p. 1
A unique and potentially fatal hypersensitivity reaction occurs in 2% to 5% of patients receiving abacavir. Symptoms typically occur within the first six weeks of therapy and include fever, rash, nausea, malaise, and respiratory complaints, in various combinations. Symptoms initially may be mild but increase in severity with continued administration. Discontinuation of the medication usually resolves all signs and symptoms, but rechallenge may cause rapid onset of severe reactions, hypotension, and death. Once an abacavir hypersensitivity reaction is suspected or confirmed, it is recommended that the patient never by rechallenged with abacavir.
Hardman, J.G., L.E. Limbird, P.B., A.G. Gilman. Goodman and Gilman's The Pharmacological Basis of Therapeutics. 10th ed. New York, NY: McGraw-Hill, 2001., p. 1359
The major toxicity associated with abacavir therapy is potentially life-threatening hypersensitivity reactions. In clinical studies, hypersensitivity reactions have been reported in approximately 5% of adult and pediatric patients receiving abacavir in conjunction with lamivudine and zidovudine. Fatalities related to hypersensitivity reactions to abacavir have been reported. Manifestations of hypersensitivity usually are apparent within the first 6 weeks of abacavir therapy, but may occur at any time during therapy. Severe hypersensitivity reactions are likely to recur within hours following rechallenge in patients with a prior history of hypersensitivity to the drug, and these reactions may include life-threatening hypotension and death. The most severe hypersensitivity reactions reported to date have been in individuals who were rechallenged with abacavir after a previous hypersensitivity reaction to the drug. There also have been reports of severe or fatal hypersensitivity reactions occurring after abacavir was reintroduced in patients with no identified history of abacavir hypersensitivity or with unrecognized manifestations of hypersensitivity to the drug. Although these patients had discontinued abacavir for reasons unrelated to hypersensitivity (e.g., interruption in drug supply, discontinuance of abacavir during treatment for other medical conditions), some may have had symptoms present before discontinuance of the drug that were consistent with hypersensitivity but were attributed to other medical conditions (e.g., acute onset respiratory disease, gastroenteritis, adverse reactions to other drugs). Most of the hypersensitivity reactions reported following reintroduction of abacavir in these patients were indistinguishable from hypersensitivity reactions associated with abacavir rechallenge (i.e., short time to onset, increased severity of symptoms, poor outcome including death).Hypersensitivity reactions can occur within hours after abacavir is reintroduced; however, in some cases, these reactions occurred days to weeks following reintroduction of the drug.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 619
Lactic acidosis and severe hepatomegaly with steatosis (sometimes fatal) have been reported rarely in patients receiving abacavir and also have been reported in patients receiving dideoxynucleoside reverse transcriptase inhibitors. Most reported cases have involved women; obesity and long-term therapy with a nucleoside reverse transcriptase inhibitor also may be risk factors. Increased serum concentrations of Gamma-glutamyltransferase (GGT, GGPT) have been reported in patients receiving abacavir.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 620
Hypersensitivity reactions reported in patients receiving abacavir are characterized by the appearance of manifestations indicating involvement of multiple organ and body systems; these reactions have occurred in association with anaphylaxis, liver failure, renal failure, hypotension, and death. The most frequent manifestations of hypersensitivity reactions to abacavir include fever, rash, fatigue, GI symptoms such as nausea, vomiting, diarrhea, and abdominal pain, and respiratory symptoms such as pharyngitis, dyspnea, and cough. Other signs and symptoms include malaise, lethargy, myalgia, myolysis, headache, arthralgia, edema, paresthesia, lymphadenopathy, and mucous membrane lesions (e.g., conjunctivitis, mouth ulceration). Respiratory symptoms, including cough, dyspnea, and pharyngitis, have been reported in approximately 20% of patients with hypersensitivity reactions to abacavir. Fatalities have occurred in patients who developed hypersensitivity reactions in which the initial presentation included respiratory symptoms; some patients who experienced fatal hypersensitivity reactions were initially diagnosed as having an acute respiratory disease (pneumonia, bronchitis, flu-like illness). Hypersensitivity reactions can occur without rash; if rash occurs, it usually is maculopapular or urticarial, but may be variable in appearance. Laboratory abnormalities reported in patients experiencing a hypersensitivity reaction to abacavir include lymphopenia and increases in serum concentrations of liver enzymes, creatine kinase (CK, creatine phosphokinase, CPK), or creatinine.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 619
For more Drug Warnings (Complete) data for ABACAVIR SULFATE (17 total), please visit the HSDB record page.
Ziagen is indicated in antiretroviral combination therapy for the treatment of Human Immunodeficiency Virus (HIV) infection in adults, adolescents and children.
The demonstration of the benefit of Ziagen is mainly based on results of studies performed with a twice daily regimen, in treatment-nave adult patients on combination therapy.
Before initiating treatment with abacavir, screening for carriage of the HLA-B*5701 allele should be performed in any HIV-infected patient, irrespective of racial origin. Abacavir should not be used in patients known to carry the HLA-B*5701 allele.
Anti-HIV Agents
Agents used to treat AIDS and/or stop the spread of the HIV infection. These do not include drugs used to treat symptoms or opportunistic infections associated with AIDS. (See all compounds classified as Anti-HIV Agents.)
Reverse Transcriptase Inhibitors
Inhibitors of reverse transcriptase (RNA-DIRECTED DNA POLYMERASE), an enzyme that synthesizes DNA on an RNA template. (See all compounds classified as Reverse Transcriptase Inhibitors.)
J05AF06
Following oral administration of a 600-mg dose of radiolabeled abacavir, 82.2% of the dose is excreted in urine and 16% of the dose is excreted in feces. The 5-carboxylic acid metabolite, 5-glucuronide metabolite, and unchanged abacavir accounted for 30, 36, and 1.2%, respectively, of recovered radioactivity in urine; unidentified minor metabolites accounted for 15% of recovered radioactivity in urine.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 623
It is not known whether abacavir is distributed into human milk; the drug is distributed into milk in rats.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 621
Abacavir crosses the placenta in rats.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 621
The oral bioavailability of abacavir is high with or without food; the CSF-to-plasma AUC ratio is approximately 0.3.
Hardman, J.G., L.E. Limbird, P.B., A.G. Gilman. Goodman and Gilman's The Pharmacological Basis of Therapeutics. 10th ed. New York, NY: McGraw-Hill, 2001., p. 1359
For more Absorption, Distribution and Excretion (Complete) data for ABACAVIR SULFATE (7 total), please visit the HSDB record page.
Abacavir is partially metabolized by alcohol dehydrogenase (to form the 5'-carboxylic acid) and glucuronidation (to form the 5'-glucuronide).
Hardman, J.G., L.E. Limbird, P.B., A.G. Gilman. Goodman and Gilman's The Pharmacological Basis of Therapeutics. 10th ed. New York, NY: McGraw-Hill, 2001., p. 1359
The metabolic fate of abacavir has not been fully determined, but the drug is metabolized in the liver. Abacavir is metabolized by alcohol dehydrogenase to form the 5-carboxylic acid and by glucuronyltransferase to form the 5-glucuronide; these metabolites do not appear to have any antiviral activity. Any involvement of cytochrome p450 isoenzymes in the metabolism of abacavir is limited.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 623
Intracellularly, abacavir is phosphorylated to abacavir monophosphate by adenosine phosphotransferase; abacavir monophosphate is then converted to carbovir monophosphate in a reaction catalyzed by cytosolic enzymes and then to carbovir triphosphate by cellular kinases. Intracellular (host cell) conversion of abacavir to carbovir triphosphate is necessary for the antiviral activity of the drug. The in vitro intracellular half-life of carbovir triphosphate in CD4+ CEM cells is 3.3 hours.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 623
The in vitro intracellular half-life of carbovir triphosphate /SRP: a metabolite of abacavir sulfate,/ in CD4+ CEM cells is 3.3 hours.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 623
The plasma elimination half-life of abacavir following a single oral dose (given as abacavir sulfate) is about 1.5 hours. In HIV-infected children 3 months to 13 years of age who received 8 mg/kg of abacavir every 12 hours (given as an oral solution containing abacavir sulfate), steady-state plasma elimination half-life averaged 1.3 hours and was essentially the same as that reported after a single dose. Following oral administration of a single 300-mg dose of abacavir to an individual with renal failure (glomerular filtration rate less than 10 mL/minute) undergoing peritoneal dialysis, the plasma elimination half-life of the drug was 1.33 hours.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 623
Like dideoxynucleoside reverse transcriptase inhibitors (e.g., didanosine, lamivudine, stavudine, zalcitabine, zidovudine), the antiviral activity of abacavir appears to depend on intracellular conversion of the drug to a 5-triphosphate metabolite; thus, carbovir triphosphate (carbocyclic guanosine triphosphate) and not unchanged abacavir appears to be the pharmacologically active form of the drug. Substantial differences exist in the rates at which human cells phosphorylate various nucleoside antiviral agents and in the enzymatic pathways involved.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 621
Enzymatic conversion of abacavir to carbovir triphosphate appears to be complex and involves certain steps and enzymes that differ from those involved in the enzymatic conversion of dideoxynucleoside reverse transcriptase inhibitors. Abacavir is phosphorylated by adenosine phosphotransferase to abacavir monophosphate, which is converted to carbovir monophosphate by a cytosolic enzyme. Subsequently, carbovir monophosphate is phosphorylated by cellular kinases to carbovir triphosphate. Abacavir is not a substrate for enzymes (i.e., thymidine kinase, deoxycytidine kinase, adenosine kinase, mitochondrial deoxyguanosine kinase) known to phosphorylate other nucleoside analogs. Because phosphorylation of abacavir depends on cellular rather than viral enzymes, conversion of the drug to the active triphosphate derivative occurs in both virus-infected and uninfected cells. Carbovir triphosphate is a structural analog of deoxyguanosine-5-triphosphate (dGTP), the usual substrate for viral RNA-directed DNA polymerase. Although other mechanisms may be involved in the antiretroviral activity of the drug, carbovir triphosphate appears to compete with deoxyguanosine-5-triphosphate for viral RNA-directed DNA polymerase and incorporation into viral DNA. Following incorporation of carbovir triphosphate into the viral DNA chain instead of deoxyguanosine-5-triphosphate, DNA synthesis is prematurely terminated because the absence of the 3-hydroxy group on the drug prevents further 5 to 3 phosphodiester linkages.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 621
The complete mechanism(s) of antiviral activity of abacavir has not been fully elucidated. Following conversion to a pharmacologically active metabolite, abacavir apparently inhibits replication of retroviruses, including human immunodeficiency virus type 1 (HIV-1) and type 2 (HIV-2), by interfering with viral RNA-directed DNA polymerase (reverse transcriptase). The drug, therefore, exerts a virustatic effect against retroviruses by acting as a reverse transcriptase inhibitor.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 621