
1. Alfetim
2. Alfusozine
3. Alfuzosin Hydrochloride
4. Alphuzosine
5. Benestan
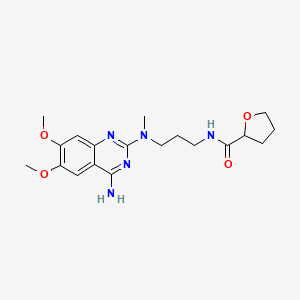
6. N-(3-((4-amino-6,7-dimethoxy-2-quinazolinyl)methylamino)propyl)tetrahydro-2-furancarboxamide
7. Urion
8. Uroxatral
9. Xatral
1. 81403-80-7
2. Alfuzosina
3. Alfuzosine
4. Alfuzosinum
5. Sl 77499
6. Alfluzocin
7. Alfluzosin
8. Alfuzosin (inn)
9. N-[3-[(4-amino-6,7-dimethoxyquinazolin-2-yl)-methylamino]propyl]oxolane-2-carboxamide
10. Chembl709
11. Nsc-760065
12. Chebi:51141
13. 90347ytw5f
14. N-(3-((4-amino-6,7-dimethoxy-2-quinazolinyl)methylamino)propyl)tetrahydro-2-furancarboxamide
15. N-{3-[(4-amino-6,7-dimethoxyquinazolin-2-yl)(methyl)amino]propyl}tetrahydrofuran-2-carboxamide
16. Alfuzosine [french]
17. Alfuzosinum [latin]
18. Alfusosine
19. Alfuzosin [inn]
20. Alfuzosina [spanish]
21. Alfuzosin [inn:ban]
22. 2-furancarboxamide,n-[3-[(4-amino-6,7-dimethoxy-2-quinazolinyl)methylamino]propyl]tetrahydro-
23. Sl 77499-10
24. N-[3-[(4-amino-6,7-dimethoxy-2-quinazolinyl)methylamino]propyl]tetrahydro-2-furancarboxamide
25. N-(3-((4-amino-6,7-dimethoxyquinazolin-2-yl)(methyl)amino)propyl)tetrahydrofuran-2-carboxamide
26. Smr000466340
27. Xatral (tn)
28. Hsdb 7290
29. Sl 77-499
30. Sl-7749910
31. Unii-90347ytw5f
32. N-(3-((4-amino-6,7-dimethoxyquinazolin-2-yl)
33. Sr-01000759345
34. Alfuzosin- Bio-x
35. Alfuzosin-[d7]
36. Alfuzosin [mi]
37. Alfuzosin [hsdb]
38. Prestwick0_000322
39. Prestwick1_000322
40. Prestwick2_000322
41. Prestwick3_000322
42. Spectrum2_000505
43. Spectrum3_001063
44. Spectrum4_001208
45. Spectrum5_000817
46. Alfuzosin [vandf]
47. Alfuzosin [who-dd]
48. Schembl34477
49. Bspbio_000323
50. Bspbio_002646
51. Kbiogr_001616
52. Mls000759449
53. Mls001424027
54. Mls006011887
55. Spectrum1505263
56. Spbio_000429
57. Spbio_002244
58. Bpbio1_000357
59. Gtpl7109
60. Dtxsid6048549
61. Kbio3_001866
62. Hms1922p11
63. Hms2051i10
64. Hms2093o06
65. Hms2235o22
66. Hms3369k03
67. Hms3393i10
68. Hms3749i05
69. Hms3886o22
70. Pharmakon1600-01505263
71. Act03219
72. Hy-b0192
73. Bdbm50033110
74. Ccg-39585
75. Mfcd00865792
76. Nsc760065
77. S5766
78. Stk643675
79. Akos005574697
80. Ccg-100859
81. Db00346
82. Nc00109
83. Nsc 760065
84. Sdccgsbi-0206735.p002
85. N-{3-[(4-amino-6,7-dimethoxyquinazolin-2-yl)(methyl)amino]propyl}oxolane-2-carboxamide
86. Ncgc00095152-01
87. Ncgc00095152-02
88. Ncgc00095152-03
89. Ncgc00095152-04
90. Ncgc00095152-05
91. Ncgc00095152-07
92. Ncgc00095152-19
93. 2-furancarboxamide, N-(3-((4-amino-6,7-dimethoxy-2-quinazolinyl)methylamino)propyl)tetrahydro-
94. Ba164145
95. Sbi-0206735.p001
96. Db-056523
97. Ab00514667
98. Ft-0630891
99. D07124
100. Ab00514667-07
101. Ab00514667-09
102. Ab00514667-10
103. Ab00514667_11
104. Ab00514667_12
105. (methyl)amino)propyl)tetrahydrofuran-2-carboxamide
106. 403a807
107. A840122
108. L001317
109. Q2736873
110. Sr-01000759345-4
111. Brd-a09056319-001-02-2
112. Brd-a09056319-001-05-5
113. Brd-a09056319-003-03-6
114. (+/-)-n-(3-((4-amino-6,7-dimethoxyquinazolin-2-yl)(methyl)amino)propyl)tetrahydrofuran-2-carboxamide
115. (+/-)-n-[3-[(4-amino-6,7-dimethoxy-2-quinazolinyl)methylamino]propyl]tetrahydro-2-furamide
116. 1100050-87-0
117. 2-furancarboxamide, (+/-)-n-(3-((4-amino-6,7-dimethoxy-2-quinazolinyl)methylamino)propyl)tetrahydro-
118. N-[3-[(4-amino-6,7-dimethoxy-quinazolin-2-yl)- Methyl-amino]propyl] Tetrahydrofuran- 2-carboxamide
119. N-[3-[(4-amino-6,7-dimethoxy-quinazolin-2-yl)-methyl-amino]propyl]tetrahydrofuran-2-carboxamide;alfuzosin
120. N-{3-[(4-imino-6,7-dimethoxy-3,4-dihydroquinazolin-2-yl)(methyl)amino]propyl}oxolane-2-carboximidic Acid
| Molecular Weight | 389.4 g/mol |
|---|---|
| Molecular Formula | C19H27N5O4 |
| XLogP3 | 1.7 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 8 |
| Exact Mass | 389.20630436 g/mol |
| Monoisotopic Mass | 389.20630436 g/mol |
| Topological Polar Surface Area | 112 Ų |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 511 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Antihypertensive
O'Neil, M.J. (ed.). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals. 13th Edition, Whitehouse Station, NJ: Merck and Co., Inc., 2001., p. 46
Alfuzosin hydrochloride is used for the symptomatic management of benign prostatic hyperplasia (BPH, benign prostatic hypertrophy). Alfuzosin relieves moderate to severe irritative (e.g., frequency, urgency, nocturia) and obstructive (e.g., hesitancy, interrupted or weak stream, sensation of incomplete bladder emptying or straining) manifestations and improves urinary flow rates in a substantial proportion of patients.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2004. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2004 (Plus Supplements)., p. 3698
Uroxatal is not indicated for the treatment of hypertension. /Alfuzosin Hydrochloride/
Physicians Desk Reference. 58th ed. Thomson PDR. Montvale, NJ 2004., p. 3037
In a study of QT effect in 45 healthy males, the QT effect appeared less with alfuzosin 10 mg than with 40 mg, and the effect of alfuzosin 40 mg did not appear as large as that of the active control moxifloxacin at its therapeutic dose. This observation should be considered in clinical decisions to prescribe Uroxatral for patients with a known history of QT prolongation or patients who are taking medications known to prolong QT, although there has been no signal of Torsades de Pointe in the extensive postmarketing experience with alfuzosin outside the United States. There are no known PK/PD studies of the effects of other alpha blockers on cardiac repolarization. /Alfuzosin Hydrochloride/
Physicians Desk Reference. 58th ed. Thomson PDR. Montvale, NJ 2004., p. 3037
Postural hypotension with or without symptoms (e.g., dizziness) may develop within a few hours following administration of Uroxatral (alfuzosin HCl extended-release tablets). As with other alpha-blockers, there is a potential for syncope. Patients should be warned of the possible occurrence of such events and should avoid situations where injury could result should syncope occur. Care should be taken when Uroxatral is administered to patients with symptomatic hypotension or patients who have had a hypotensive response to other medications.
Physicians Desk Reference. 58th ed. Thomson PDR. Montvale, NJ 2004., p. 3037
Carcinoma of the prostate and benign prostatic hyperplasia cause many of the same symptoms. These two diseases frequently coexist. Therefore, patients thought to have benign prostatic hyperplasia should be examined prior to starting therapy with Uroxatral (alfuzosin HCl extended-release tablets) to rule out the presence of carcinoma of the prostate.
Physicians Desk Reference. 58th ed. Thomson PDR. Montvale, NJ 2004., p. 3037
If symptoms of angina pectoris should newly appear or worsen, Uroxatral should be discontinued. /Extended Release Alfuzosin Hydrochloride/
Physicians Desk Reference. 58th ed. Thomson PDR. Montvale, NJ 2004., p. 3037
For more Drug Warnings (Complete) data for ALFUZOSIN (16 total), please visit the HSDB record page.
Alfuzosin is used to treat the signs and symptoms of benign prostatic hyperplasia (BPH).
FDA Label
By selectively inhibiting alpha adrenergic receptors in the lower urinary tract, alfuzosin causes smooth muscle relaxation in the bladder neck and prostate, improving urine flow, thereby reducing BPH symptoms. Additionally, alfuzosin reduces the vasoconstrictor effect of catecholamines (epinephrine and norepinephrine), leading to peripheral vasodilation. This leads to a risk of postural hypotension/syncope, and prescribing information warns that caution should be exercised in patients who take nitrates, antihypertensives, or have experienced decreased blood pressure after using other medications.
Adrenergic alpha-1 Receptor Antagonists
Drugs that bind to and block the activation of ADRENERGIC ALPHA-1 RECEPTORS. (See all compounds classified as Adrenergic alpha-1 Receptor Antagonists.)
Urological Agents
Drugs used in the treatment of urological conditions and diseases such as URINARY INCONTINENCE and URINARY TRACT INFECTIONS. (See all compounds classified as Urological Agents.)
G04CA01
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
G - Genito urinary system and sex hormones
G04 - Urologicals
G04C - Drugs used in benign prostatic hypertrophy
G04CA - Alpha-adrenoreceptor antagonists
G04CA01 - Alfuzosin
Absorption
Alfuzosin is readily absorbed in the gastrointestinal tract and the absolute bioavailability under fed conditions is 49%. In patients over 75 years of age, alfuzosin is absorbed more rapidly and peak plasma levels are higher. One source mentions a bioavailability of 64%. After multiple doses under fed conditions, Cmax is achieved in 8 hours. Cmax and AUC0-24 values are about 13.6 ng/mL and 194 ngh/mL, respectively. Steady-state plasma concentrations are achieved after the second dose and are 1.2 to 1.6 times higher than after a single dose. With the extended-release formulation, alfuzosin release is sustained over 20 hours with a rate of dissolution ranging between 2 and 12 hours.
Route of Elimination
It is partially metabolised and excreted mainly in the bile and faeces. Following oral administration of a radiolabeled alfuzosin solution, the detection of radioactivity after one week was 69% in the feces and 24% in the urine.
Volume of Distribution
The volume of distribution of alfuzosin after intravenous administration in healthy volunteers is about 3.2 L/kg. Alfuzosin distributes heavily to the tissues of the prostate.
Clearance
Exercise caution if renal clearance is < 30 mL/min. The clearance of alfuzosin is increased in renal insufficiency (with or without dialysis), due to an increase in the free fraction.
Following oral administration of (14)C-labeled alfuzosin solution, the recovery of radioactivity after 7 days (expressed as a percentage of the administered dose) was 69% in feces and 24% in urine. /Alfuzosin Hydrochloride/
Physicians Desk Reference. 58th ed. Thomson PDR. Montvale, NJ 2004., p. 3036
The volume of distribution following intravenous administration in healthy male middle-aged volunteers was 3.2 L/kg. Results of in vitro studies indicate that alfuzosin is moderately bound to human plasma proteins (82% to 90%), with linear binding over a wide concentration range (5 to 5,000 ng/mL). /Alfuzosin Hydrochloride/
Physicians Desk Reference. 58th ed. Thomson PDR. Montvale, NJ 2004., p. 3036
The extent of absorption is 50% lower under fasting conditions. Therefore, Uroxatral should be taken immediately following a meal. /Alfuzosin Hydrochloride/
Physicians Desk Reference. 58th ed. Thomson PDR. Montvale, NJ 2004., p. 3036
The absolute bioavailability of Uroxatral 10 mg tablets under fed conditions is 49%. Following multiple dosing of 10 mg Uroxatral under fed conditions, the time to maximum concentration is 8 hours. Cmax and AUC0-24 are 13.6 (SD = 5.6) ng/mL and 194 (SD = 75) ng.h/mL, respectively.Uroxatral exhibits linear kinetics following single and multiple dosing up to 30 mg. Steady-state plasma levels are reached by with the second dose of Uroxatral administration. Steady-state alfuzosin plasma concentrations are 1.2- to 1.6-fold higher than those observed after a single administration. /Extended Release Alfuzosin Hydrochloride/
Physicians Desk Reference. 58th ed. Thomson PDR. Montvale, NJ 2004., p. 3036
For more Absorption, Distribution and Excretion (Complete) data for ALFUZOSIN (6 total), please visit the HSDB record page.
Alfuzosin undergoes extensive hepatic metabolism; only 11% of the administered dose is detected unchanged in the urine. Alfuzosin is metabolism occurs via three metabolic pathways: oxidation, O-demethylations, and N-dealkylation. Metabolites of alfuzosin are not pharmacologically active and CYP3A4 is main hepatic cytochrome enzyme responsible for its metabolism.
Alfuzosin undergoes extensive metabolism by the liver, with only 11% of the administered dose excreted unchanged in the urine. Alfuzosin is metabolized by three metabolic pathways: oxidation, O-demethylation, and N-dealkylation. The metabolites are not pharmacologically active. CYP3A4 is the principal hepatic enzyme isoform involved in its metabolism. /Alfuzosin Hydrochloride/
Physicians Desk Reference. 58th ed. Thomson PDR. Montvale, NJ 2004., p. 3036
The apparent elimination half-life of alfuzosin after oral administration is about 10 hours. The terminal half-life is 3-5 hours.
Following oral administration of Uroxatral 10 mg tablets, the apparent elimination half-life is 10 hours. /Extended Release Alfuzosin Hydrochloride/
Physicians Desk Reference. 58th ed. Thomson PDR. Montvale, NJ 2004., p. 3036
Alpha(1)-adrenoreceptors are found in the prostate, bladder base, bladder neck, prostatic capsule, and prostatic urethra; their activation may lead to contraction of smooth muscle and urinary symptoms in patients with BPH. Alfuzosin selectively binds to and inhibits alpha(1)-adrenergic receptors in the lower urinary tract. This leads to the relaxation of smooth muscle in both the prostate and bladder neck, resulting in the improvement in urine flow and a reduction of urinary symptoms.
Alfuzosin hydrochloride, a quinazoline-derivative alpha1-adrenergic blocking agent, is structurally and pharmacologically related to prazosin. Alfuzosin is a non-subtype-specific alpha1-adrenergic blocking agent that exhibits selectivity for alpha1-adrenergic receptors in the lower urinary tract (e.g., bladder base, bladder neck, prostate, prostatic capsule, prostatic urethra). Blockade of these adrenoreceptors can cause relaxation of smooth muscle in the bladder neck and prostate, resulting in improvement in urine flow and a reduction in symptoms of benign prostatic hyperplasia (BPH).
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2004. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2004 (Plus Supplements)., p. 3698