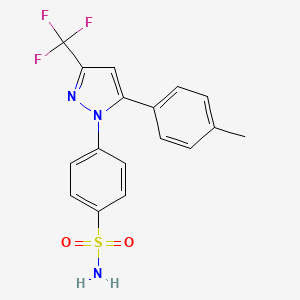

1. 4-(5-(4-methylphenyl)-3-(trifluoromethyl)-1h-pyrazol-1-yl)benzenesulfonamide
2. Celebrex
3. Sc 58635
4. Sc-58635
5. Sc58635
1. 169590-42-5
2. Celebrex
3. Celebra
4. Onsenal
5. Celocoxib
6. Celecox
7. Sc 58635
8. Sc-58635
9. 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1h-pyrazol-1-yl]benzenesulfonamide
10. 4-[5-(4-methylphenyl)-3-(trifluoromethyl)pyrazol-1-yl]benzenesulfonamide
11. Ym177
12. 184007-95-2
13. Ym 177
14. P-(5-p-tolyl-3-(trifluoromethyl)pyrazol-1-yl)benzenesulfonamide
15. Hsdb 7038
16. Mfcd00941298
17. 4-(5-(4-methylphenyl)-3-(trifluoromethyl)-1h-pyrazol-1-yl)benzenesulfonamide
18. Sc58635
19. 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1h-pyrazol-1-yl]benzene-1-sulfonamide
20. Chembl118
21. Nsc-719627
22. Nsc-758624
23. Dfn15
24. Jcx84q7j1l
25. Dfn-15
26. 194044-54-7
27. Chebi:41423
28. 4-(5-(p-tolyl)-3-(trifluoromethyl)-1h-pyrazol-1-yl)benzenesulfonamide
29. Benzenesulfonamide, 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1h-pyrazol-1-yl]-
30. Celecoxib [usan]
31. Ncgc00091455-01
32. Xilebao
33. Dsstox_cid_2777
34. 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1h-pyrazol-1-yl]-benzenesulfonamide
35. 4-[5-(4-methylphenyl)-3-(trifluoromethyl)pyrazol-1-yl]benzenesulfonami De
36. Dsstox_rid_76725
37. Dsstox_gsid_22777
38. Solexa
39. Benzenesulfonamide, 4-(5-(4-methylphenyl)-3-(trifluoromethyl)-1h-pyrazol-1-yl)-
40. Celebrex (tn)
41. Smr000550473
42. Cas-169590-42-5
43. Sr-01000837528
44. Ym-177
45. Unii-jcx84q7j1l
46. Celecoxibum
47. Celecoxib [usan:inn:ban]
48. Ccris 9330
49. Tpi-336
50. Celecoxib- Bio-x
51. Celecoxib-[d4]
52. Onsenal (tn)
53. Ai-525
54. Cep-33222
55. Celebcoxib
56. Elyxyb
57. Spectrum_000432
58. 1oq5
59. Celecoxib [inn]
60. Celecoxib [jan]
61. Celecoxib [mi]
62. Celecoxib [hsdb]
63. Spectrum2_001576
64. Spectrum3_001996
65. Spectrum4_000182
66. Spectrum5_001324
67. Celecoxib [vandf]
68. Celecoxib [mart.]
69. Celecoxib [usp-rs]
70. Celecoxib [who-dd]
71. Cid_2662
72. Schembl3708
73. Celecoxib (jan/usp/inn)
74. Bspbio_003596
75. Celecoxib [ema Epar]
76. Kbiogr_000723
77. Kbiogr_002351
78. Kbioss_000912
79. Kbioss_002354
80. Mls001165684
81. Mls001195656
82. Mls001304708
83. Mls006011862
84. Bidd:gt0408
85. Divk1c_000893
86. Spectrum1503678
87. Spbio_001512
88. Gtpl2892
89. Celecoxib [orange Book]
90. Celecoxib [ep Monograph]
91. Celecoxib, >=98% (hplc)
92. Dtxsid0022777
93. Bdbm11639
94. Hms502m15
95. Kbio1_000893
96. Kbio2_000912
97. Kbio2_002351
98. Kbio2_003480
99. Kbio2_004919
100. Kbio2_006048
101. Kbio2_007487
102. Kbio3_002830
103. Kbio3_003037
104. Celecoxib [usp Monograph]
105. Ex-a175
106. Cmap_000027
107. Ninds_000893
108. Bcpp000290
109. Elyxyb (dfn-15; Oral Solution)
110. Hms1922g14
111. Hms2089l18
112. Hms2093i07
113. Hms2234n18
114. Hms3259l08
115. Hms3261a14
116. Hms3373a09
117. Hms3654h09
118. Hms3715f11
119. Hms3867i03
120. Hms3884m07
121. Pharmakon1600-01503678
122. Consensi Component Celecoxib
123. Act02648
124. Albb-033772
125. Bcp02156
126. Zinc2570895
127. Tox21_111135
128. Tox21_201964
129. Tox21_300599
130. Tox21_500406
131. Us8741944, Comparative Compound
132. 4-[5-(p-tolyl)-3-(trifluoromethyl)pyrazol-1-yl]benzenesulfonamide
133. Bbl029086
134. Ccg-39354
135. Nsc719627
136. Nsc758624
137. S1261
138. Stl373576
139. Celecoxib 1.0 Mg/ml In Acetonitrile
140. Akos015842517
141. Celecoxib Component Of Consensi
142. Tox21_111135_1
143. Ac-4228
144. Am84588
145. Bcp9000507
146. Cs-0570
147. Db00482
148. Ks-1041
149. Nc00708
150. Nsc 719627
151. Nsc 758624
152. Sb19318
153. Idi1_000893
154. Celecoxib 100 Microg/ml In Acetonitrile
155. Ncgc00091455-02
156. Ncgc00091455-03
157. Ncgc00091455-04
158. Ncgc00091455-05
159. Ncgc00091455-06
160. Ncgc00091455-07
161. Ncgc00091455-08
162. Ncgc00091455-09
163. Ncgc00091455-13
164. Ncgc00254540-01
165. Ncgc00259513-01
166. Ncgc00261091-01
167. Bc164295
168. Bp-30217
169. Hy-14398
170. Nci60_041049
171. Sy064976
172. Sbi-0051875.p002
173. Unm-0000305813
174. Ft-0601628
175. Ft-0623536
176. Ft-0700357
177. Sw199611-3
178. A25046
179. C07589
180. D00567
181. Ab00052396-07
182. Ab00052396-08
183. Ab00052396-09
184. Ab00052396_10
185. Ab00052396_11
186. 590c425
187. Q408801
188. J-010566
189. J-520011
190. Q-200816
191. Sr-01000837528-2
192. Sr-01000837528-3
193. Brd-k02637541-001-02-4
194. Brd-k02637541-001-06-5
195. Z2210694606
196. Celecoxib, European Pharmacopoeia (ep) Reference Standard
197. Celecoxib, United States Pharmacopeia (usp) Reference Standard
198. 4-[5-(p-tolyl)-3-(trifluoromethyl)-1-pyrazolyl]benzenesulfonamide
199. 4-(5-p-tolyl-3-(trifluoromethyl)-1h-pyrazol-1-yl)benzenesulfonamide
200. 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-pyrazol-1-yl]benzenesulfonamide
201. 5-(4-methylphenyl)-1-(4-sulfamoylphenyl)-3-(trifluoromethyl)pyrazole
202. Celecoxib, Pharmaceutical Secondary Standard; Certified Reference Material
203. 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1h-pyazol-1-yl]benezenesulfonamide
204. Benzenesulfonamide,4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1h-pyrazol-1-yl]-
205. 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-pyrazol-1-yl]benzenesulfonamide;4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1h-pyrazol-1-yl]benzenesulfonamide
| Molecular Weight | 381.4 g/mol |
|---|---|
| Molecular Formula | C17H14F3N3O2S |
| XLogP3 | 3.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 3 |
| Exact Mass | 381.07588236 g/mol |
| Monoisotopic Mass | 381.07588236 g/mol |
| Topological Polar Surface Area | 86.4 Ų |
| Heavy Atom Count | 26 |
| Formal Charge | 0 |
| Complexity | 577 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 4 | |
|---|---|
| Drug Name | Celebrex |
| PubMed Health | Celecoxib (By mouth) |
| Drug Classes | Analgesic, Antirheumatic, Central Nervous System Agent, Musculoskeletal Agent |
| Drug Label | CELEBREX (celecoxib) is chemically designated as 4-[5-(4-methylphenyl)- 3-(trifluoromethyl)-1H-pyrazol-1-yl] benzenesulfonamide and is a diaryl-substituted pyrazole. The empirical formula is C17H14F3N3O2S, and the molecular weight is 381.38; the chem... |
| Active Ingredient | Celecoxib |
| Dosage Form | Capsule |
| Route | oral; Oral |
| Strength | 200mg; 100mg; 50mg; 400mg |
| Market Status | Prescription |
| Company | Gd Searle |
| 2 of 4 | |
|---|---|
| Drug Name | Celecoxib |
| PubMed Health | Celecoxib (By mouth) |
| Drug Classes | Analgesic, Antirheumatic, Central Nervous System Agent, Musculoskeletal Agent |
| Drug Label | CELEBREX (celecoxib) is chemically designated as 4-[5-(4-methylphenyl)- 3-(trifluoromethyl)-1H-pyrazol-1-yl] benzenesulfonamide and is a diaryl-substituted pyrazole. The empirical formula is C17H14F3N3O2S, and the molecular weight is 381.38; the chem... |
| Active Ingredient | Celecoxib |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 200mg; 100mg; 50mg; 400mg |
| Market Status | Tentative Approval; Prescription |
| Company | Watson Labs; Mylan Pharms; Teva |
| 3 of 4 | |
|---|---|
| Drug Name | Celebrex |
| PubMed Health | Celecoxib (By mouth) |
| Drug Classes | Analgesic, Antirheumatic, Central Nervous System Agent, Musculoskeletal Agent |
| Drug Label | CELEBREX (celecoxib) is chemically designated as 4-[5-(4-methylphenyl)- 3-(trifluoromethyl)-1H-pyrazol-1-yl] benzenesulfonamide and is a diaryl-substituted pyrazole. The empirical formula is C17H14F3N3O2S, and the molecular weight is 381.38; the chem... |
| Active Ingredient | Celecoxib |
| Dosage Form | Capsule |
| Route | oral; Oral |
| Strength | 200mg; 100mg; 50mg; 400mg |
| Market Status | Prescription |
| Company | Gd Searle |
| 4 of 4 | |
|---|---|
| Drug Name | Celecoxib |
| PubMed Health | Celecoxib (By mouth) |
| Drug Classes | Analgesic, Antirheumatic, Central Nervous System Agent, Musculoskeletal Agent |
| Drug Label | CELEBREX (celecoxib) is chemically designated as 4-[5-(4-methylphenyl)- 3-(trifluoromethyl)-1H-pyrazol-1-yl] benzenesulfonamide and is a diaryl-substituted pyrazole. The empirical formula is C17H14F3N3O2S, and the molecular weight is 381.38; the chem... |
| Active Ingredient | Celecoxib |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 200mg; 100mg; 50mg; 400mg |
| Market Status | Tentative Approval; Prescription |
| Company | Watson Labs; Mylan Pharms; Teva |
Cyclooxygenase 2 Inhibitors
National Library of Medicine's Medical Subject Headings. Celecoxib. Online file (MeSH, 2018). Available from, as of January 23, 2018: https://meshb.nlm.nih.gov/search
/CLINICAL TRIALS/ ClinicalTrials.gov is a registry and results database of publicly and privately supported clinical studies of human participants conducted around the world. The Web site is maintained by the National Library of Medicine (NLM) and the National Institutes of Health (NIH). Each ClinicalTrials.gov record presents summary information about a study protocol and includes the following: Disease or condition; Intervention (for example, the medical product, behavior, or procedure being studied); Title, description, and design of the study; Requirements for participation (eligibility criteria); Locations where the study is being conducted; Contact information for the study locations; and Links to relevant information on other health Web sites, such as NLM's MedlinePlus for patient health information and PubMed for citations and abstracts for scholarly articles in the field of medicine. Celecoxib is included in the database.
NIH/NLM; ClinicalTrials.Gov. Available from, as of January 22, 2018: https://clinicaltrials.gov/
/Celebrex is indicated/ for the management of the signs and symptoms of osteoarthritis. /Included in US product labeling/
NIH; DailyMed. Current Medication Information for Celebrex (Celecoxib Capsule) (Updated: October 6, 2017). Available from, as of January 24, 2018: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=8d52185d-421f-4e34-8db7-f7676db2a226
/Celebrex is indicated/ for the management of the signs and symptoms of rheumatoid arthritis. /Included in US product labeling/
NIH; DailyMed. Current Medication Information for Celebrex (Celecoxib Capsule) (Updated: October 6, 2017). Available from, as of January 24, 2018: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=8d52185d-421f-4e34-8db7-f7676db2a226
For more Therapeutic Uses (Complete) data for Celecoxib (14 total), please visit the HSDB record page.
/BOXED WARNING/ WARNING: RISK OF SERIOUS CARDIOVASCULAR EVENTS. Cardiovascular Thrombotic Events: Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction, and stroke, which can be fatal. This risk may occur early in the treatment and may increase with duration of use. Celebrex is contraindicated in the setting of coronary artery bypass graft (CABG) surgery.
NIH; DailyMed. Current Medication Information for Celebrex (Celecoxib Capsule) (Updated: October 6, 2017). Available from, as of January 24, 2018: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=8d52185d-421f-4e34-8db7-f7676db2a226
/BOXED WARNING/ WARNING: RISK OF SERIOUS GASTROINTESTINAL EVENTS. Gastrointestinal Bleeding, Ulceration, and Perforation: Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events.
NIH; DailyMed. Current Medication Information for Celebrex (Celecoxib Capsule) (Updated: October 6, 2017). Available from, as of January 24, 2018: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=8d52185d-421f-4e34-8db7-f7676db2a226
A subpopulation of patients with asthma may have aspirin-sensitive asthma which may include chronic rhinosinusitis complicated by nasal polyps; severe, potentially fatal bronchospasm; and/or intolerance to aspirin and other nonsteroidal anti-inflammatory drugs (NSAIDs). Because cross-reactivity between aspirin and other NSAIDs has been reported in such aspirin-sensitive patients, celebrex is contraindicated in patients with this form of aspirin sensitivity. When celebrex is used in patients with preexisting asthma (without known aspirin sensitivity), monitor patients for changes in the signs and symptoms of asthma.
NIH; DailyMed. Current Medication Information for Celebrex (Celecoxib Capsule) (Updated: October 6, 2017). Available from, as of January 24, 2018: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=8d52185d-421f-4e34-8db7-f7676db2a226
Celebrex is contraindicated in patients with previous serious skin reactions to nonsteroidal anti-inflammatory drugs (NSAIDs).
NIH; DailyMed. Current Medication Information for Celebrex (Celecoxib Capsule) (Updated: October 6, 2017). Available from, as of January 24, 2018: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=8d52185d-421f-4e34-8db7-f7676db2a226
For more Drug Warnings (Complete) data for Celecoxib (33 total), please visit the HSDB record page.
Celecoxib is indicated for symptomatic treatment of adult osteoarthritis (OA) and adult rheumatoid arthritis (RA). Celecoxib is not a substitute for aspirin for cardiovascular event prophylaxis. It may be also be used to treat acute pain from various sources, juvenile rheumatoid arthritis in children over 2, ankylosing spondylitis, and primary dysmenorrhea. Celecoxib, in combination with [tramadol], is indicated for the management of acute pain in adults severe enough to require an opioid analgesic and in whom alternative treatments are inadequate.
Onsenal is indicated for the reduction of the number of adenomatous intestinal polyps in familial adenomatous polyposis (FAP), as an adjunct to surgery and further endoscopic surveillance (see section 4. 4).
The effect of Onsenal-induced reduction of polyp burden on the risk of intestinal cancer has not been demonstrated (see sections 4. 4 and 5. 1)
Celecoxib inhibits cyclooxygenase 2 (COX-2) enzyme, reducing pain and inflammation. It is important to note that though the risk of bleeding with celecoxib is lower than with certain other NSAIDS, it exists nonetheless and caution must be observed when it is administered to those with a high risk of gastrointestinal bleeding. **A note on the risk of cardiovascular events** Significant concerns regarding the safety of COX-2 selective NSAIDs emerged in the early 2000s. [Rofecoxib], another member of the COX-2 inhibitor drug class, also known as Vioxx, was withdrawn from the market due to prothrombotic cardiovascular risks. Following an FDA Advisory Committee meeting in 2005, in which data from large clinical outcome trials were evaluated, the FDA concluded that the risk for cardiovascular thrombotic events for both COX-2 selective NSAIDs and nonselective NSAIDs was evident. It was determined that the benefits of celecoxib treatment, however, outweighed the risks. Postmarketing cardiovascular outcomes trial (PRECISION) revealed that the lowest possible dose of celecoxib was similar in cardiovascular safety to moderate strength doses of both naproxen and ibuprofen. Patients who had previous cardiovascular events including acute MI, coronary revascularization, or coronary stent insertion were not evaluated in the trial. It is not advisable to administer NSAIDS to these groups of patients.
Cyclooxygenase 2 Inhibitors
A subclass of cyclooxygenase inhibitors with specificity for CYCLOOXYGENASE-2. (See all compounds classified as Cyclooxygenase 2 Inhibitors.)
Anti-Inflammatory Agents, Non-Steroidal
Anti-inflammatory agents that are non-steroidal in nature. In addition to anti-inflammatory actions, they have analgesic, antipyretic, and platelet-inhibitory actions. They act by blocking the synthesis of prostaglandins by inhibiting cyclooxygenase, which converts arachidonic acid to cyclic endoperoxides, precursors of prostaglandins. Inhibition of prostaglandin synthesis accounts for their analgesic, antipyretic, and platelet-inhibitory actions; other mechanisms may contribute to their anti-inflammatory effects. (See all compounds classified as Anti-Inflammatory Agents, Non-Steroidal.)
L01XX33
M01AH01
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01X - Other antineoplastic agents
L01XX - Other antineoplastic agents
L01XX33 - Celecoxib
M - Musculo-skeletal system
M01 - Antiinflammatory and antirheumatic products
M01A - Antiinflammatory and antirheumatic products, non-steroids
M01AH - Coxibs
M01AH01 - Celecoxib
Absorption
Celecoxib is absorbed rapidly in the gastrointestinal tract. When a single oral dose of 200 mg was given to healthy research subjects, the peak plasma levels of celecoxib occurred within 3 hours. The Cmax is 705 ng/mL. When multiple doses are given, steady-state concentrations are reached on or before day 5. When taken with a high-fat meal, peak plasma levels are delayed for about 1 to 2 hours with an increase in total absorption (AUC) of 10% to 20%. The AUC of celecoxib has been shown to be significantly lower in patients with chronic renal impairment. A meta-analysis of pharmacokinetic studies has suggested an approximately 40% higher AUC (area under the curve) of celecoxib in black patients compared to Caucasians for unknown reasons.
Route of Elimination
Celecoxib is primarily eliminated by hepatic metabolism with small amounts (<3%) of the unchanged drug found in both the urine and feces. About 57% of an oral dose of celecoxib is excreted in the feces and 27% is found to be excreted into the urine in the form of metabolites. The main metabolite in urine and feces is identified as the carboxylic acid metabolite (73%). The amount of glucuronide in the urine is reported to be low.
Volume of Distribution
The apparent volume of distribution of celecoxib at steady state (Vss/F) is about 429 L, which suggests wide distribution into various tissues. Celecoxib is not preferentially bound to red blood cells. Another resource reports a volume of distribution of 455 166L.
Clearance
Apparent clearance (CL/F), single oral 200 mg dose, healthy subjects = 27.7 L/hr. Clearance may be decreased by about 47% in patients with chronic renal insufficiency, according to a pharmacokinetic study. Studies have not been performed in patients with severe renal impairment.
/MILK/ Limited data from 3 published reports that included a total of 12 breastfeeding women showed low levels of Celebrex in breast milk. The calculated average daily infant dose was 10-40 ug/kg/day, less than 1% of the weight-based therapeutic dose for a two-year old-child.
NIH; DailyMed. Current Medication Information for Celebrex (Celecoxib Capsule) (Updated: October 6, 2017). Available from, as of January 24, 2018: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=8d52185d-421f-4e34-8db7-f7676db2a226
/MILK/ The aim of this study was to investigate the transfer of celecoxib into human milk. In one group of 3 breastfeeding patients on celecoxib at steady state, milk levels were determined at set intervals over 24 hours. Plasma levels were determined in 2 of their infants, age 17 and 22 months. In a second group of 2 subjects, intravenous lines were placed and a single 200-mg dose of celecoxib was followed by multiple paired plasma and milk samples over 8 hours. The mean milk-to-plasma ratio for celecoxib was 0.23 (95% confidence interval [CI]: 0.15-0.31). The average concentration of celecoxib in milk during the 8-hour dosing interval was 66 ug/L (95% CI: 41-89). The absolute infant dose averaged 9.8 ug/kg/d (95% CI: 6.2-13.4); the mean relative infant dose was 0.30%. Therefore, the average clinical dose transferred to the infant daily would be approximately 0.3% of the weight-adjusted maternal dose. ...
PMID:15479658 Hale TW et al; J Hum Lact 20 (4): 397-403 (2004)
/MILK/ A 40 year old woman who was breastfeeding her 5 month old daughter was admitted to the hospital for surgery. In the postoperative period, she received four doses of celecoxib (100 mg twice/day) in addition to other medications. Starting about 5 hours after her last dose, four milk samples were obtained by hand expression over a 24 hour interval. The elimination half life range was 4.0-6.5 hours. These data suggest that celecoxib would be eliminated from breast milk about 24 hours after the last dose. Although maternal plasma was not obtained, the estimated milk:plasma ratios (based on reported adult plasma levels) were 0.27-0.59. the infant did not resume breastfeeding until 48 hours after the last dose. If she had nursed, the estimated maximum infant dose would have have been about 40 ug/kg/day.
Briggs, G.G., Freeman, R.K., Yaffee, S.J.; Drugs in Pregancy and Lactation Tenth Edition. Wolters Kluwer/Lippincott Williams & Wilkins, Philadelphia, PA. 2015, p. 234
/MILK/ /The aim of this study was/ to determine the milk-to-plasma (M/P) concentration ratio of celecoxib, and estimate likely infant exposure. Blood and milk were sampled for 48 hr after oral administration of celecoxib 200 mg to six lactating volunteers. The M/P ratio was derived from the area under the concentration-time curves (0-infinity) and the infant 'dose' estimated from celecoxib concentrations in milk. The median (range) M/P ratio was 0.18 (0.15-0.26). The median (range) infant 'dose' was 0.23% (0.17-0.30%) of the maternal dose, adjusted for weight. ...
PMID:16390357 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1884977 Gardiner SJ et al; Br J Clin Pharmacol 61 (1): 101-4 (2006)
For more Absorption, Distribution and Excretion (Complete) data for Celecoxib (11 total), please visit the HSDB record page.
A large part of celecoxib metabolism is mediated by cytochrome P450 2C9 in the liver with some contribution from CYP3A4 and CYP2C8 and possible contributions from CYP2D6. It is metabolized by biotransformation to carboxylic acid and glucuronide metabolites. Three metabolites, a primary alcohol, a carboxylic acid, and a glucuronide conjugate, have been found in human plasma after celecoxib administration. These are considered inactive metabolites in regards to COX enzyme inhibition. Patients who are known or suspected to have decreased cytochrome P450 2C9 activity or function, based on their previous history, should be administered celecoxib with caution as they may have abnormally high serum concentrations resulting from decreased metabolism celecoxib.
Celecoxib metabolism is primarily mediated via CYP2C9. Three metabolites, a primary alcohol, the corresponding carboxylic acid and its glucuronide conjugate, have been identified in human plasma. These metabolites are inactive as COX-1 or COX-2 inhibitors.
NIH; DailyMed. Current Medication Information for Celebrex (Celecoxib Capsule) (Updated: October 6, 2017). Available from, as of January 24, 2018: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=8d52185d-421f-4e34-8db7-f7676db2a226
Celecoxib has known human metabolites that include Hydroxy celecoxib.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
The effective half-life of celecoxib is approximately 11 hours when a single 200 mg dose is given to healthy subjects. The terminal half-life of celecoxib varies because of its low solubility, which prolongs absorption.
A 40 year old woman who was breastfeeding her 5 month old daughter was admitted to the hospital for surgery. In the postoperative period, she received four doses of celecoxib (100 mg twice/day) in addition to other medications. Starting about 5 hours after her last dose, four milk samples were obtained by hand expression over a 24 hour interval. The elimination half life range was 4.0-6.5 hours. ...
Briggs, G.G., Freeman, R.K., Yaffee, S.J.; Drugs in Pregancy and Lactation Tenth Edition. Wolters Kluwer/Lippincott Williams & Wilkins, Philadelphia, PA. 2015, p. 234
The plasma elimination half-life of celecoxib following oral administration of a single 200-mg dose under fasting conditions is about 11 hours, and the apparent plasma clearance of the drug is about 500 mL/minute; these parameters exhibit wide intraindividual variability, presumably because the low aqueous solubility of celecoxib prolongs absorption. The half-life of celecoxib is prolonged in patients with renal or hepatic impairment and has been reported to be 13.1 hours in patients with chronic renal insufficiency and 11 or 13.1 hours in patients with mild or moderate hepatic impairment, respectively.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 2151
Unlike most NSAIDs, which inhibit both types of cyclooxygenases (COX-1 and COX-2), celecoxib is a selective noncompetitive inhibitor of cyclooxygenase-2 (COX-2) enzyme. COX-2 is expressed heavily in inflamed tissues where it is induced by inflammatory mediators. The inhibition of this enzyme reduces the synthesis of metabolites that include prostaglandin E2 (PGE2), prostacyclin (PGI2), thromboxane (TXA2), prostaglandin D2 (PGD2), and prostaglandin F2 (PGF2). Resultant inhibition of these mediators leads to the alleviation of pain and inflammation. By inhibiting prostaglandin synthesis, non-steroidal anti-inflammatory drugs (NSAIDs) cause mucosal damage, ulceration and ulcer complication throughout the gastrointestinal tract. Celecoxib poses less of an ulceration risk than other NSAIDS, owing to its decreased effect on gastric mucosal prostaglandin synthesis when compared to placebo. Celecoxib exerts anticancer effects by binding to the cadherin-11 (CDH11)protein, which is thought to be involved in the progression of tumors, and inhibiting the 3-phosphoinositide-dependent kinase-1 (PDK-1) signaling mechanism. In addition, celecoxib has been found to inhibit carbonic anhydrase enzymes 2 and 3, further enhancing its anticancer effects. As mentioned in the pharmacodynamics section of this drug entry, celecoxib may cause an increased risk of thrombotic events. The risk of thrombosis resulting from COX-2 inhibition is caused by the vasoconstricting actions of thromboxane A2, leading to enhanced platelet aggregation, which is uncontrolled when the actions of prostacyclin, a platelet aggregation inhibitor, are suppressed through the inhibition of COX-2.
Nonsteroidal anti-inflammatory drugs (NSAIDs) are well-known causes of acute renal insufficiency and gastropathy in patients with chronic inflammatory diseases. This action is presumed to result from nonselective inhibition of both constitutive and inducible forms of prostaglandin H synthases, also known as the cyclooxygenase enzymes (i.e., COX-1 amd COX-2). Celecoxib (Celebrex) is a COX-2 enzyme inhibitor and has emerged as a preferred therapeutic agent for the treatment of rheumatoid arthritis as compared to other NSAIDs. Celecoxib has recently been the subject of criticism for its side effects, mainly arterial thrombosis and renal hemorrhage, although it is considered a superior drug in protecting the gastrointestinal tract. In the present study, we report that celecoxib not only inhibited COX-2, but also exhibited the property of inhibiting adenylyl cyclase, an important enzyme forming the intracellular second messenger 3',5'-adenosine monophosphate (cAMP) from adenosine triphosphate (ATP). Celecoxib also inhibited cholera toxin-stimulated cAMP formation, which indicated its ability to permeate cell membranes in order to reach intracellular adenylyl cyclase. It inhibited in vitro adenylyl cyclase activity in both human colonic epithelial cells and purified adenylyl cyclase from Bordetella pertussis. The IC50 of celecoxib for B. pertussis adenylyl cyclase was calculated to be 0.375 mM. Lineweaver-Burk analysis showed that the type of enzyme inhibition was competitive. The apparent Km and Vm of adenylyl cyclase was calculated as 25.0 nM and 7.14 nmol/min/mg, respectively. Celecoxib changed the Km value to 66.6 nM without affecting the Vmax. The current study suggests that apart from inflammation, celecoxib therapy could be further extended to diseases involving cAMP upregulation either by endogenous reactions or exogenous agents. These new data showing inhibition of adenylyl cyclase should be considered in light of the drug's pathological effects or in patients specifically excluded from treatment (e.g., asthmatics).
PMID:12797547 Saini SS et al; Inflammation 27 (2): 79-88 (2003)
Cardiovascular disease is one of the leading causes of death worldwide, and evidence indicates a correlation between the inflammatory process and cardiac dysfunction. Selective inhibitors of cyclooxygenase-2 (COX-2) enzyme are not recommended for long-term use because of potentially severe side effects to the heart. Considering this and the frequent prescribing of commercial celecoxib, the present study analyzed cellular and molecular effects of 1 and 10 uM celecoxib in a cell culture model. After a 24-hr incubation, celecoxib reduced cell viability in a dose-dependent manner as also demonstrated in MTT assays. Furthermore, reverse transcription-polymerase chain reaction analysis showed that the drug modulated the expression level of genes related to death pathways, and Western blot analyses demonstrated a modulatory effect of the drug on COX-2 protein levels in cardiac cells. In addition, the results demonstrated a downregulation of prostaglandin E2 production by the cardiac cells incubated with celecoxib, in a dose-specific manner. These results are consistent with the decrease in cell viability and the presence of necrotic processes shown by Fourier transform infrared analysis, suggesting a direct correlation of prostanoids in cellular homeostasis and survival.
PMID:24519091 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3932973 Sakane KK et al; Braz J Med Biol Res 47 (1): 50-9 (2014)
Celecoxib, a selective cyclooxygenase-2 (COX-2) inhibitor, has recently been shown to affect the development of different types of cancer. The present study utilized a murine H22 hepatocarcinoma model to investigate the molecular mechanisms involved in celecoxib-induced inhibition of tumor angiogenesis. Tumor-bearing mice were randomly divided into five groups: i) control; ii) low-dose celecoxib (50 mg/kg); iii) high-dose celecoxib (200 mg/kg); iv) 5-fluorouracil (5-FU), (20 mg/kg) and v) combination of 5-FU and celecoxib (50 mg/kg). The antitumor effect of celecoxib was determined by measuring tumor volume. Tumor angiogenesis was evaluated by microvessel density (MVD). Tumor histology and immunostaining for CD34 in endothelial cells were performed to detect MVD. The expression levels of phosphatase and tensin homologue deleted from chromosome 10 (PTEN), phosphatidylinositol 3-kinase (PI3K), phospho-Akt (P-Akt), COX-2, hypoxia-inducible factor-1a (HIF-1a) and vascular endothelial growth factor-A (VEGF-A) were detected by ELISA, immunohistochemistry and western blotting, respectively. We discovered substantial growth delay in murine H22 hepatoma as a result of celecoxib treatment. The inhibition rate of tumor growth induced by high-dose and low-dose celecoxib was 49.3 and 37.0%, respectively (P<0.05). The expression of PI3K, P-Akt, COX-2, HIF-1a, VEGF-A and PTEN in tumor tissues treated with celecoxib was demonstrated by immunohistochemistry, and the MVD was decreased in a dose-dependent manner (P<0.05). Reduced PI3K and P-Akt was particularly apparent in the high-dose celecoxib group (P<0.05). ELISA and western blotting data showed that the expression of PI3K, P-Akt, COX-2, HIF-1a and VEGF-A were reduced and PTEN was increased after treatment with celecoxib. In conclusion, the impact of celecoxib-induced tumor growth delay of murine H22 hepatocarcinoma may correlate with the inhibition of angiogenesis by reducing PI3K, P-Akt, COX-2, HIF-1a and VEGF-A expression and increasing PTEN expression in tumor tissue.
PMID:24647425 Sui W et al; Oncol Rep 31 (5): 2252-60 (2014)
The mechanism of action of Celebrex is believed to be due to inhibition of prostaglandin synthesis, primarily via inhibition of cyclooxygenase-2 (COX-2).
NIH; DailyMed. Current Medication Information for Celebrex (Celecoxib Capsule) (Updated: October 6, 2017). Available from, as of January 24, 2018: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=8d52185d-421f-4e34-8db7-f7676db2a226