
1. Bay Vp 2674
2. Bay-vp-2674
3. Baytril
4. Endrofloxicin
1. 93106-60-6
2. Baytril
3. Enrofloxacine
4. Cfpq
5. Bay Vp 2674
6. Enrofloxacino
7. Enrofloxacinum
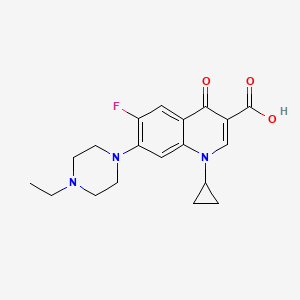
8. 1-cyclopropyl-7-(4-ethylpiperazin-1-yl)-6-fluoro-4-oxo-1,4-dihydroquinoline-3-carboxylic Acid
9. Enroxil
10. Baytril (tn)
11. N-ethylciprofloxacin
12. Erfx
13. 1-cyclopropyl-7-(4-ethyl-1-piperazinyl)-6-fluoro-1,4-dihydro-4-oxo-3-quinolinecarboxylic Acid
14. 3-quinolinecarboxylic Acid, 1-cyclopropyl-7-(4-ethyl-1-piperazinyl)-6-fluoro-1,4-dihydro-4-oxo-
15. Mfcd00792463
16. 3dx3xek1bn
17. Nsc-758616
18. Enrofloxacin For Veterinary Use
19. Mls000069441
20. Chebi:35720
21. Bay Vp 2674;pd160788
22. Endrofloxicin
23. Ncgc00018143-04
24. Cpd000059011
25. Pd160788
26. Smr000059011
27. 1-cyclopropyl-7-(4-ethylpiperazin-1-yl)-6-fluoro-4-oxoquinoline-3-carboxylic Acid
28. Dsstox_cid_25619
29. Dsstox_rid_81007
30. Dsstox_gsid_45619
31. Enrofloxacine [french]
32. Enrofloxacinum [latin]
33. Enrofloxacino [spanish]
34. Bay-vp-2674
35. Cas-93106-60-6
36. Enrofloxacin [usan:ban:inn]
37. Hsdb 6952
38. Unii-3dx3xek1bn
39. Brn 5307824
40. Ccris 8214
41. Enrofloxacin [usan:usp:inn:ban]
42. Enrofloxacin-[d5]
43. Opera_id_1106
44. Enrofloxacin (usp/inn)
45. Enrofloxacin [mi]
46. Enrofloxacin (usan/inn)
47. Enrofloxacin [inn]
48. Enrofloxacin [hsdb]
49. Enrofloxacin [usan]
50. 1,4-dihydro-1-cyclopropyl-7-(4-ethyl-1-piperazinyl)-6-fluoro-4-oxo-3-quinolinecarboxylic Acid
51. Bay-vp2674
52. Enrofloxacin [mart.]
53. Mls001076496
54. Mls001424169
55. Chembl15511
56. Enrofloxacin [usp-rs]
57. Schembl149150
58. Spectrum1503721
59. Dtxsid1045619
60. Enrofloxacin [green Book]
61. Hms2052o09
62. Hms2090e12
63. Hms2093i21
64. Hms2234m11
65. Hms3373p14
66. Hms3394o09
67. Hms3715b18
68. Pharmakon1600-01503721
69. Zinc597112
70. Enrofloxacin [usp Impurity]
71. Albb-030792
72. Bcp15457
73. Hy-b0502
74. Enrofloxacin [usp Monograph]
75. Enrofloxacin, >=98.0% (hplc)
76. Tox21_110831
77. Bbl009982
78. Dl-384
79. Nsc758616
80. S3059
81. Stk711109
82. Akos005530685
83. Bay-vp2674;pd160788
84. Tox21_110831_1
85. Ac-7614
86. Ccg-101102
87. Db11404
88. Ks-5010
89. Nc00352
90. Nsc 758616
91. 1-cyclopropyl-7-(4-ethylpiperazin-1-yl)-6-fluoro-4-oxo-quinoline-3-carboxylic Acid
92. 1-cyclopropyl-7-(4-ethylpiperazinyl)-6-fluoro-4-oxohydroquinoline-3-carboxylic Acid
93. Enrofloxacin 100 Microg/ml In Methanol
94. Ncgc00018143-01
95. Ncgc00018143-02
96. Ncgc00018143-03
97. Ncgc00018143-05
98. Ncgc00021632-03
99. 1-cyclopropyl-7-(4-ethyl-1-piperazinyl)-6-fluoro-1,4-dihydro-4-oxoquinoline-3-carboxylic Acid
100. 3-quinolinecarboxylic Acid, 1,4-dihydro-1-cyclopropyl-7-(4-ethyl-1-piperazinyl)-6-fluoro-4-oxo-
101. Enrofloxacin 1000 Microg/ml In Methanol
102. Sbi-0206725.p001
103. Db-057368
104. B1742
105. E0786
106. Ft-0625663
107. Ft-0667862
108. D02473
109. Ab00384269-16
110. Ab00384269_17
111. Ab00384269_18
112. Enrofloxacin, Vetranal(tm), Analytical Standard
113. 106e606
114. A844445
115. Q414413
116. Sr-01000000045
117. Sr-01000000045-3
118. Brd-k76534306-001-11-0
119. Enrofloxacin For Veterinary Use [ep Impurity]
120. Enrofloxacin For Veterinary Use [ep Monograph]
121. Enrofloxacin, European Pharmacopoeia (ep) Reference Standard
122. Enrofloxacin, United States Pharmacopeia (usp) Reference Standard
123. 1-cyclopropyl-7-(4-ethyl-1-piperazinyl)-6-fluoro-4-oxo-3-quinolinecarboxylic Acid
124. Enrofloxacin For System Suitability, European Pharmacopoeia (ep) Reference Standard
125. Enrofloxacin, Pharmaceutical Secondary Standard; Certified Reference Material
126. 1-cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-7-(4-ethyl-1-piperazinyl)-3-quinoline-carboxylic Acid
127. 1-cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-7-(4-ethyl-1-piperazinyl)-3-quinolinecarboxylic Acid
128. 1-cyclopropyl-7-(4-ethyl-1-piperazinyl)-6-fluoro-1,4-dihydro-4-oxo-3- Quinolonecarboxylic Acid
129. 1-cyclopropyl-7-(4-ethyl-1-piperazinyl)-6-fluoro-1,4-dihydro-4-oxo-3-quinolinecarboxylic Acid, 9ci
130. 1-cyclopropyl-7-(4-ethylpiperazin-1-yl)-6-fluoranyl-4-oxidanylidene-quinoline-3-carboxylic Acid
| Molecular Weight | 359.4 g/mol |
|---|---|
| Molecular Formula | C19H22FN3O3 |
| XLogP3 | -0.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 4 |
| Exact Mass | 359.16451973 g/mol |
| Monoisotopic Mass | 359.16451973 g/mol |
| Topological Polar Surface Area | 64.1 Ų |
| Heavy Atom Count | 26 |
| Formal Charge | 0 |
| Complexity | 613 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anti-Bacterial Agents
Substances that inhibit the growth or reproduction of BACTERIA. (See all compounds classified as Anti-Bacterial Agents.)
Pharmacokinetics and bioavailability of enrofloxacin were determined after single intravenous (IV) and intramuscular (IM) administrations of 5 mg/kg body weight (BW) to 5 healthy adult Angora goats. Plasma enrofloxacin concentrations were measured by high performance liquid chromatography. Pharmacokinetics were best described by a 2-compartment open model. The elimination half-life and volume of distribution after IV and IM administrations were similar (t1/2beta, 4.0 to 4.7 hr and Vd(ss),1.2 to 1.5 L/kg, respectively). Enrofloxacin was rapidly (t1/2a, 0.25 hr) and almost completely absorbed (F, 90%) after IM administration. Mean plasma concentrations of enrofloxacin at 24 hr after IV and IM administration (0.07 and 0.09 microg/mL, respectively) were higher than the minimal inhibitory concentration (MIC) values for most pathogens. In conclusion, once-daily IV and IM administration of enrofloxacin (5 mg/kg BW) in Angora goats may be useful in treatment of infectious diseases caused by sensitive pathogens.
PMID:11227198 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1189645 Elmas M et al; Can J Vet Res 65 (1): 64-7 (2001)
Plasma, urine, and skin drug concentrations were determined for dogs (n=12) given five daily oral doses of marbofloxacin (MAR) (2.75 mg/kg), enrofloxacin (ENR) (5.0 mg/kg) or difloxacin (DIF) (5.0 mg/kg). Concentrations of the active metabolite of ENR, ciprofloxacin (CIP), were also determined. The three-period, three-treatment crossover experimental design included a 21-day washout period between treatments. Area under the plasma drug concentration vs. time curve (AUC0-last, microg/mlxhr of MAR was greater than for ENR, CIP, ENR/CIP combined, and DIF. Maximum concentration (Cmax) of MAR was greater than ENR, CIP, and DIF. Time of maximum plasma concentration (Tmax) was similar for MAR and DIF; Tmax occurred earlier for ENR and later for CIP. Plasma half-life (t1/2) of MAR was longer than for ENR, CIP, and DIF. Urine concentrations of DIF were less than MAR or ENR/CIP combined, but urine concentrations of MAR and ENR/CIP combined did not differ. DIF skin concentrations were less than the concentrations of MAR or ENR/CIP combined 2 h after dosing, but skin concentrations of MAR and ENR/CIP combined did not differ.
PMID:11107003 Frazier DL et al; J Vet Pharmacol Ther 23 (5): 293-302 (2000)
Serum concentrations and pharmacokinetics of enrofloxacin were studied in 6 mares after intravenous (IV) and intragastric (IG) administration at a single dose rate of 7.5 mg/kg body weight. In experiment 1, an injectable formulation of enrofloxacin (100 mg/ml) was given IV. At 5 min after injection, mean serum concentration was 9.04 microg/mL and decreased to 0.09 microg/mL by 24 hr. Elimination half-life was 5.33 +/- 1.05 hr and the area under the serum concentration vs time curve (AUC) was 21.03 +/- 5.19 mg x hr/L. In experiment 2, the same injectable formulation was given IG. The mean peak serum concentration was 0.94 +/- 0.97 microg/ml at 4 hr after administration and declined to 0.29 +/- 0.12 microg/ml by 24 hr. Absorption of this enrofloxacin preparation after IG administration was highly variable, and for this reason, pharmacokinetic values for each mare could not be determined. In experiment 3, a poultry formulation (32.3 mg/ml) was given IG. The mean peak serum concentration was 1.85 +/- 1.47 microg/ml at 45 min after administration and declined to 0.19 +/- 0.06 microg/mL by 24 h. Elimination half-life was 10.62 +/- 5.33 h and AUC was 16.30 +/- 4.69 mg x h/L. Bioavailability was calculated at 78.29 +/- 16.55%. Minimum inhibitory concentrations of enrofloxacin were determined for equine bacterial culture specimens submitted to the microbiology laboratory over an 11-month period. The minimum inhibitory concentration of enrofloxacin required to inhibit 90% of isolates (MIC90) was 0.25 microg/ml for Staphylococcus aureus, Escherichia coli, Salmonella spp., Klebsiella spp., and Pasteurella spp. The poultry formulation was well tolerated and could be potentially useful in the treatment of susceptible bacterial infections in adult horses. The injectable enrofloxacin solution should not be used orally.
PMID:10935883 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1189609 Haines GR et al; Can J Vet Res 64 (3): 171-7 (2000)
Concentrations of enrofloxacin equivalent activity were determined by microbiological assay in the plasma of healthy and E. coli-infected broilers following single intravenous and oral administrations at 10 mg/kg. Tissue distribution and residue-depletion following multiple oral doses (10 mg/kg for 3 successive days) were investigated. Pharmacokinetic variables were determined using compartmental and non-compartmental analytical methods. Plasma enrofloxacin concentrations after intravenous dosing to healthy and infected birds were best described by a two-compartments model. Enrofloxacin concentrations in plasma of infected birds were lower than those of healthy ones. The disposition kinetics of intravenously administered drug in healthy and infected birds were somewhat different. The elimination half-life (t1/2 beta) was 4.75 vs. 3.63 hr; mean residence time (MRT) was 6.72 vs 4.90 hr; apparent volume of the central compartment (Vc) was 1.11 vs 1.57 l/kg; rate constant for transfer from peripheral to central compartment (k21) was 1.15 vs 1.41 hr-1 and total body clearance (ClB) was 0.35 vs 0.53 l/hr/kg in healthy and infected birds, respectively. After oral administration, the absorption half-life (t1/2abs) in the infected birds was significantly longer than in healthy birds, while elimination half-life (t1/2el) and MRT were significantly shorter. Bioavailability was higher in infected birds (72.50%) as compared to healthy ones (69.78%). Enrofloxacin was detected in the tissues of healthy and infected birds after daily oral dosing of 10 mg/kg for 3 days. It was more concentrated in liver, kidney, and breast muscle. The minimal inhibitory concentration (MIC) of enrofloxacin against E. coli was 0.064 microgram/ml. On the basis of maintaining enrofloxacin plasma concentrations over the MIC, a dose of 10 mg/kg given intravenously every 20.14 hr or orally every 20.86 hr should provide tissue concentrations effective against E. coli infection in chickens.
PMID:10689795 Soliman GA; Dtsch Tierarztl Wochenschr 107 (1): 23-7 (2000)
For more Absorption, Distribution and Excretion (Complete) data for ENROFLOXACIN (6 total), please visit the HSDB record page.
The pharmacokinetics of enrofloxacin and its active metabolite ciprofloxacin were investigated in goats after a single intramuscular administration of enrofloxacin at 2.5 mg/kg body weight. The plasma concentrations of enrofloxacin and ciprofloxacin were determined simultaneously by a HPLC method. The peak concentrations (Cmax) of enrofloxacin (1.13 microg/ml) and ciprofloxacin (0.24 microg/ml) were observed at 0.8 and 1.2 hr, respectively. The elimination half-life (t1/2beta), volume of distribution (Vd(area)), total body clearance (Cl(B)) and mean residence time (MRT) of enrofloxacin were 0.74 hr, 1.42 l/kg, 1329 ml/hr per kg and 1.54 hr, respectively. The t1/2beta, area under the plasma concentration-time curve (AUC) and the MRT of ciprofloxacin were 1.38 h, 0.74 microg h/ml and 2.73 h, respectively. The metabolic conversion of enrofloxacin to ciprofloxacin was appreciable (36%) and the sum of the plasma concentrations of enrofloxacin and ciprofloxacin was maintained at or above 0.1 microg/ml for up to 4 hr. Enrofloxacin appears to be useful for the treatment of goat diseases associated with pathogens sensitive to this drug.
PMID:11334149 Rao GS et al; Vet Res Commun 25 (3): 197-204 (2001)